The Science behind Beer


The saying goes ‘Beer is an acquired taste’ and it really is. I’ve had an evolving relationship with beer. First, when I was younger, thinking it was gross when adults would drink it around me based on the smell and a little taste. In college it became a staple but it is always the cheap stuff. Thankfully now I’m at the quality versus quantity stage where I enjoy a beer with dinner every so often- the way others drink a nice glass of wine. Beer itself has been evolving quite a bit lately, we no longer need to rely on the big boys- like Budweiser and Heineken- there are plenty of microbrewers out there. Small-scale breweries are taking risks and coming up with some delicious, unusual concoctions. And of course behind beer is my focus topic- science. So let’s get down to the nitty-gritty and see how it all happens.
Beer brewing ingredients and the overall process are interconnected. Let’s look at a brief overview of the process, discuss the major ingredients in detail and then look at the process in more detail. Overall sugars from grain are used by yeast, for energy, and the by-product of this conversion is alcohol and carbon dioxide. This process is known as fermentation, where energy is derived from the oxidation of organic compounds. In the case of alcohol fermentation, carbohydrates (sugars) are oxidized topermityeast energy. As far as yeast is concerned ethanol and carbon dioxide are just side products. Lucky for beer lovers, brewers and scientists over the years have manipulated this little organism’s quest for energy into our favorite buzzy brew. (Ethanol is also known as ethyl alcohol or commonly alcohol even though science-wise it is just one type of alcohol.) First up in the overall process, the grain needs to be prepped to make the sugars available for when yeast it is introduced into the system. The grain, usually barley, stores it sugar in long chains of insoluble starch. Most of the steps occur before brewing and involve the process of working on the grain to get the starch exposed and broken down for accessibility. Once all this is done the fermentation can occur, as mentioned yeast utilizes the newly available sugar for energy and conversion of one molecule of sugar, glucose (C6H1206), gives two molecules of ethanol (C2H5OH) and two molecules of carbon dioxide (CO2). Once the yeast have converted as much sugar as they can they either settle to the bottom of the system or float to the top, depending on the type of yeast. The beer is generally separated from the yeast and other things, such as the spent grain and any degradation products from the grain, and it is further conditioned, packaged, and ready for consumption.


Barley, as mentioned is the most common grain for brewing. Barley contains spikes, which are the flowering component of this grain. Each spike is situated on the central axis of the plant, the rachis. The rachis contains nodes, where each node in turn contains three spikelets. Each fertile spikelet contains both the male (stamen) and female (pistil) reproductive components since barley is a self-pollinating plant. These spikelets give rise to a kernel and the bracts. Bracts are specialized leaves that surround the kernel of the immature plant and become the husk of the mature kernel. Directly under the husks is a multi-layered tissue-the aleurone-which surrounds the endosperm, another layer of tissue which provides nutrients to the developing kernel. Aleurone is the major site of enzyme synthesis of the barley. Barley is a domesticated version of the wild Hordeum Vulgare. It comes in two major varieties, genetic modification allowing for an alternative to the wild version. The natural scenario is two-row barley, where only the center spikelet on each side is fertile. Six-rowed barley has a mutation in the vrs1 gene, allowing for the lateral spikelets to be fertile in addition to the two center fertile spikelets. The differences between two-row and six-row have been looked at in great detail, in terms of their capacity for malting, the principal step in beer brewing. For example, the non-fertile lateral spikelets in two-row barley have a lower protein content, which means a higher fermentable sugar content. Typically barley contains 67% carbohydrates, sugars which can ultimately fuel yeast for alcohol fermentation, and 12.8% protein. There is a trade-off between carbohydrate and protein content, as one increases the other decreases.
Yeast is a single-celled organism responsible for the fermentation, converting sugar into energy and as a by-product releasing both alcohol and carbon dioxide, in beer production. Yeast ferments in an anaerobic environment which means without oxygen. The presence or absence of oxygen is the difference between what will happen when yeast and sugar are mixed together in water. In the presence of oxygen, yeast will use the energy from sugar to replicate- making more copies. Then when all the oxygen is consumed, but sugar is still available, yeast will then convert the sugar for energy and make alcohol. There are many different strains of yeast, each requiring different amounts and varieties of nutrients and a specific temperature at which to make alcohol. Large-scale brewers tend to have their own particular strain of yeast. The strain dictates the ideal amount of sugar for the given yeast to be exposed to and thus to be able to consume and a maximum tolerance level in terms of alcohol. Water is another critical component to the overall brewing process; it is used to prepare the barley for fermentation. As well it is the solvent for fermentation to occur, added to the grain to pull the sugars out into the open for the yeast to access them. Hops are the other major ingredient and I will discuss them in more detail in the second part of this article.
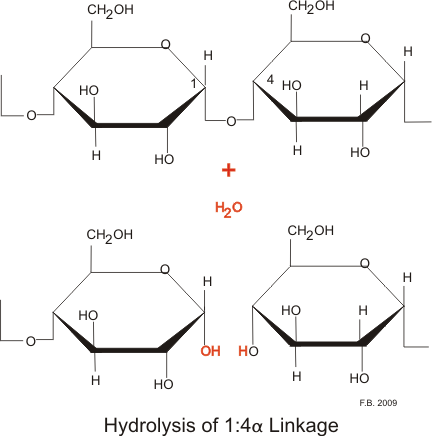
The process of brewing starts with exposing the sugars in the grain to allow yeast to utilize them. The grain is steeped, soaked, in water and then the plant is left to germinate for several days. The germination will allow for the all the necessary enzymes to be formed and released. Enzymes are required to breakdown cellular components as well as the starch itself.Starch is an insoluble storage form of glucose. Glucose is a simple sugar, a monosaccharide, the simplest sugar in fact and it is the major energy source of the cell. It comes from the organism’s energy source-food for animals, photosynthesis for plants, organic compounds for yeast. When in excess glucose is stored as starch, within the organism, which is made up of many glucose monomers bound together. The bonds holding the monomers together are glycosidic bonds, which link up a specific carbon from one glucose to the next via its specific carbon. Glucose contains six carbons that form a ring (with oxygen and hydrogen- recall the chemical composition is C6H1206).When glucose is needed in the organism, it requires one of two enzymes to break the bonds between the glucose units. There are two amylases, alpha and beta, both are capable of breaking glycosidic bonds. The difference in these two enzymes takes into consideration the two types of starches.There is amylose, which is made up of linear chains of several hundred glucose units.The bond between glucose monomers in amylose is between the one carbon of the first glucose and the fourth carbon on the second glucose, and thus the bond is referred to as a 1, 4 glycosidic bond. The other type of starch is amylopectin which has 1, 4 bonds in a linear fashion like amylose but also has 1, 6 bonds which allow for branching at approximately every 20th glucose residue. There are several thousand glucose units in each amylopectin. Alpha amylase has access to more bonds since it is not restricted to end bonds like beta amylase, and it can break down the branching of amylopectin as well as the linear amylose.
The germination stage must be stopped at a critical point; you want starch to begin to break down but not completely. The intermediate between starch, a polysaccharide, and glucose, a monosaccharide, is the disaccharide maltose which consists of two glucose molecules bound together.It is important to allow germination long enough for the starch disrupting enzymes to be released but not used.The vast majority of the glucose should remain bundled up in its storage form of starch when germinated ceases. The germination is stopped by drying, or kilning, between 50 and 85°C, where the temperature depends on what type of malt you are going for. The temperature must be gradually increased, because enzymes have a strict structure-function policy, and too high temps will disrupt the structure and render them unable to function. Next up is milling, where the grain will crack. This will allow the water added in the next step to extract the sugar. There is a delicate balance is this stage because the husks serve as a filter in the subsequent steps. Due to this brewing role, it is important not to break them down completely during this stage.

Next up is mashing, where the mash is separated out to give a sweetened liquid.The starch breakdown will be complete and all will be in the simplest sugar forms, mostly maltose and glucose. Three-glucose chains, maltotrioses, are fermentable as well.The milled grain, known as the grist, is added to water and the mixture is heated, again gradually, to 75°C in the mash-lauter tun. This increase in temperature will render the enzymes inactive.This step is also referred to as lautering, separating the wort from the spent grain named for the false bottom on the tun that filters out the liquid wort from the spent grain. This overall step contains sparging, first the liquid is drained from the lauter tun bottom and then set to recirculate from the top and here it will pass through the husks from the kernels which serve as a filter.Additional water is then added to the spent grain, this is sparging, in order to make sure to get all the fermentable sugars.The wort, also called the extract, is the sugar-rich water strained out of the mash, leaving behind proteins, fats, and the spent grain. The wort is boiled during the brewing phase, which sterilizes and concentrates it. At the beginning of the boil, hops are added; contributing to the aroma and bitterness but also can act as a preserver of the brew.
After boiling the wort, it needs to be cooled and aerated before the yeast is added. Yeast will not be able to function at higher temperatures. Hops are removed before the next stage- fermentation. As previously described the yeast will use the now accessible and broken down simple sugars and in turn alcohol and carbon dioxide are generated. This process takes about ten days, but varies depending on the type of beer desired. There is an exhaust pipe to let out most of the carbon dioxide. Towards the end of the fermentation the vent is capped and the carbon dioxide remains in the brew. This adds carbonation to the beer. The strain of yeast dictates if it will sink to the bottom or float to the top when it has consumed all the sugar that it will. The beer is transferred from the fermentation tank to a conditioning tank, leaving the yeast and any remaining solids behind. At this time the beer with be saturated with additional carbon dioxide to complete carbonation. This conditioning tank will allow for the flavor to continue developing. It will also be filtered, although not always depending on the type of beer, and packaged.
As usual with complex scientific processes this is just an overview, fermentation is more complicated than I’ve described but the overall equation is glucose in and alcohol and carbon dioxide out, this is carried out by enzymes in the yeast. Speaking of enzymes, there are more enzymes involved in breaking down starch to simply sugars than the amylases I mentioned. But overall the process is preparing grain to expose its stored sugar; this includes creating a mash of the grain and water, straining out the fermentable sugars and introducing yeast for fermentation, and later hops to ultimately produce beer.
Part Two
- The Science behind Beer Ingredients
In-depth look into Hops, a major beer ingredient, also beer adjuncts, such as grains and fruit, a brief description of various types of beers, and alcohol content determination.