What Is an Atom?
- Charges on Atoms
Atoms part 2
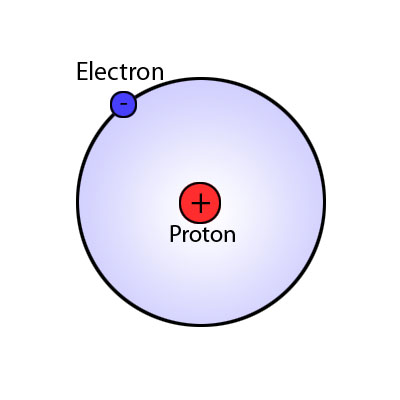
Hydrogen atom
An introduction to the atom
So just what is an atom?
If you're completely new to this subject, I want to try to explain this in such a way as you don't get put off after reading the first couple of lines. Please leave a comment at the end if I manage it!!!
An atom is simply the smallest "bit" of a chemical element that you can get. A chemical element is something that is listed in the periodic table, such as hydrogen, oxygen, copper or gold. You can't have an atom of water, because water isn't an element. Water's made up of two different elements - hydrogen and oxygen. So you can have an atom of hydrogen, and you can have an atom of oxygen, and they can join together, but you can't just get a atom of water.
Still with me? Good :-)
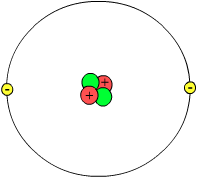
Helium atom
As you might expect, an atom of hydrogen is a bit different to an atom of gold. The atom of every element is a bit different to the atom of every other element. They're all still atoms though, and they are all similar to each other.
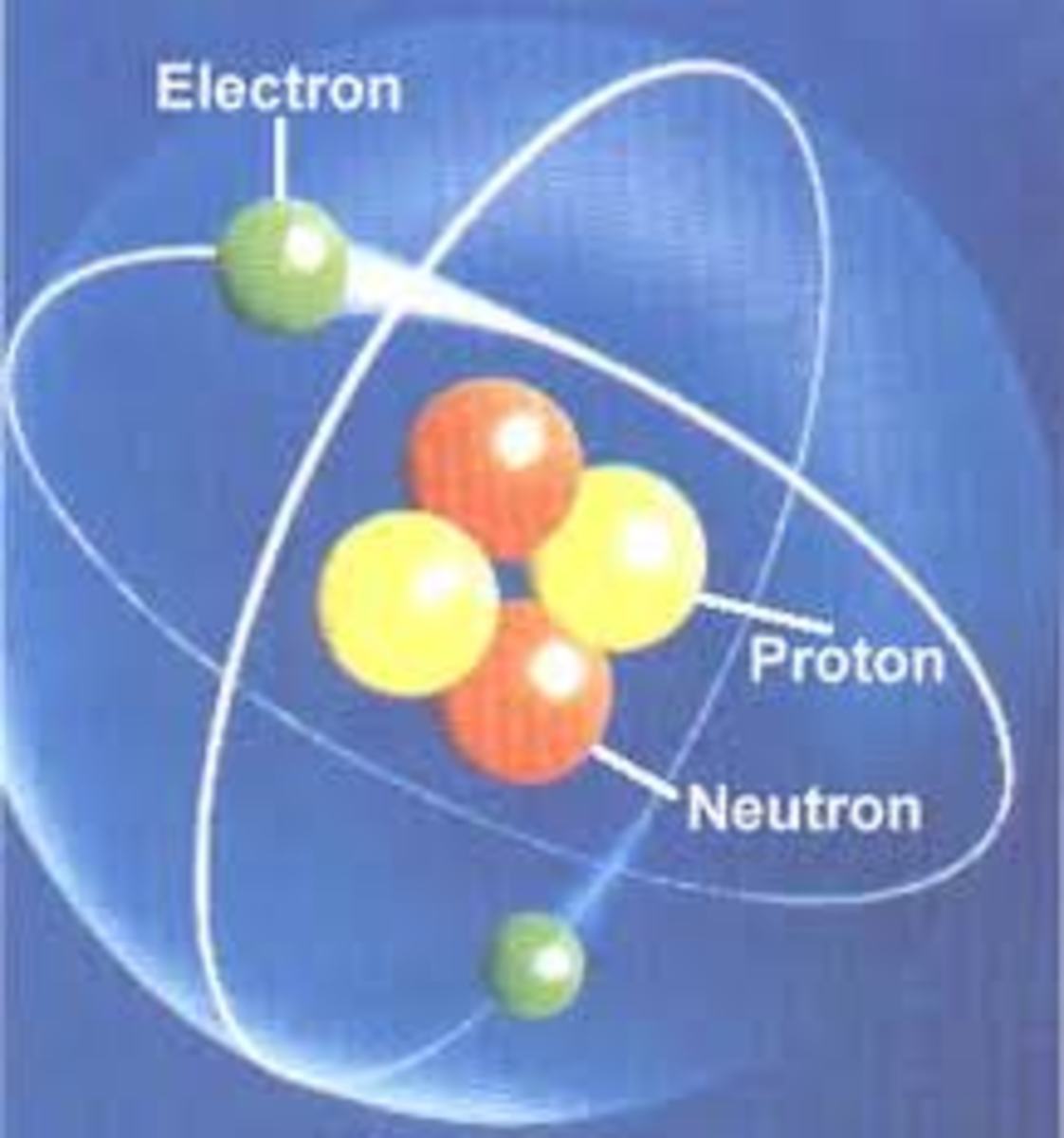
Protons, neutrons and electrons
Learn these three words; proton, neutron, and electron. These are some parts of an atom. The protons and neutrons are in the middle, and the electrons go round the outside. The protons and neutrons are both called "nucleons", because they are in the "nucleus" (the centre, or the middle). If this sounds a bit confusing, just think of people - a boy is a child, and a girl is a child. A man is an adult and a woman is an adult. A proton is a nucleon and a neutron is a nucleon.
Almost all atoms have protons and neutrons in the nucleus - there is just one exception; the hydrogen atom. A normal (most common) hydrogen atom has just 1 proton and no neutrons in it's nucleus, like you can see in the hydrogen atom diagram. It has 1 electron which goes around the outside.
Carbon atom
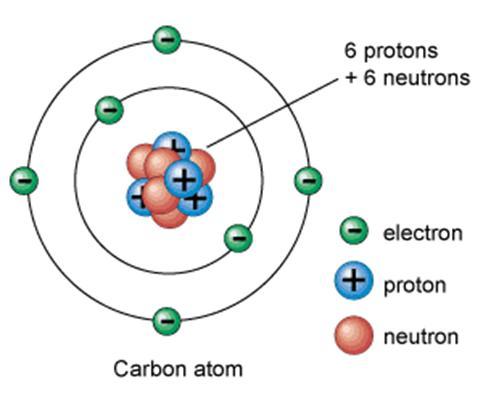
Isotopes
Atoms with a different number of protons to neutrons are called isotopes.
For example, carbon atoms have same number of protons (i.e. same atomic number - 6) but they may have different number of neutrons (which results in a different mass number). These different carbon atoms are called isotopes of carbon.
So is that it - all there is to know about atoms? No - that's just the beginning! Atoms are a basic (but not simple!) fact which comes into all kinds of science - physics, chemistry and biology. I have tried to make this understandable for complete beginners, and I do hope I've managed it. I know it is simplified and far from complete - if it turns out to be helpful, I'll try another hub, taking it one stage further.
Thanks for reading!