Fluoride, how much is too much?

What is Fluoride?
Fluoride is a chemical ion, it has an extra electron that gives it a negative charge, it is found naturally in water, foods, soil and several other minerals, but it is also created in laboratories where it may be added to water and other dental products such as toothpaste and mouthwash.
Fluoride does a good job of protecting teeth from decay and cavities in several ways. When sugars combine with the bacteria in the mouth, acid is created which erodes tooth enamel and damages teeth. Fluoride demineralizes the acids, thus protecting the teeth. If the teeth are already damaged, the opposite occurs, a process called re-mineralizing occurs by the fluoride which begins to strengthen the enamel.

non-fluoride children toothpaste
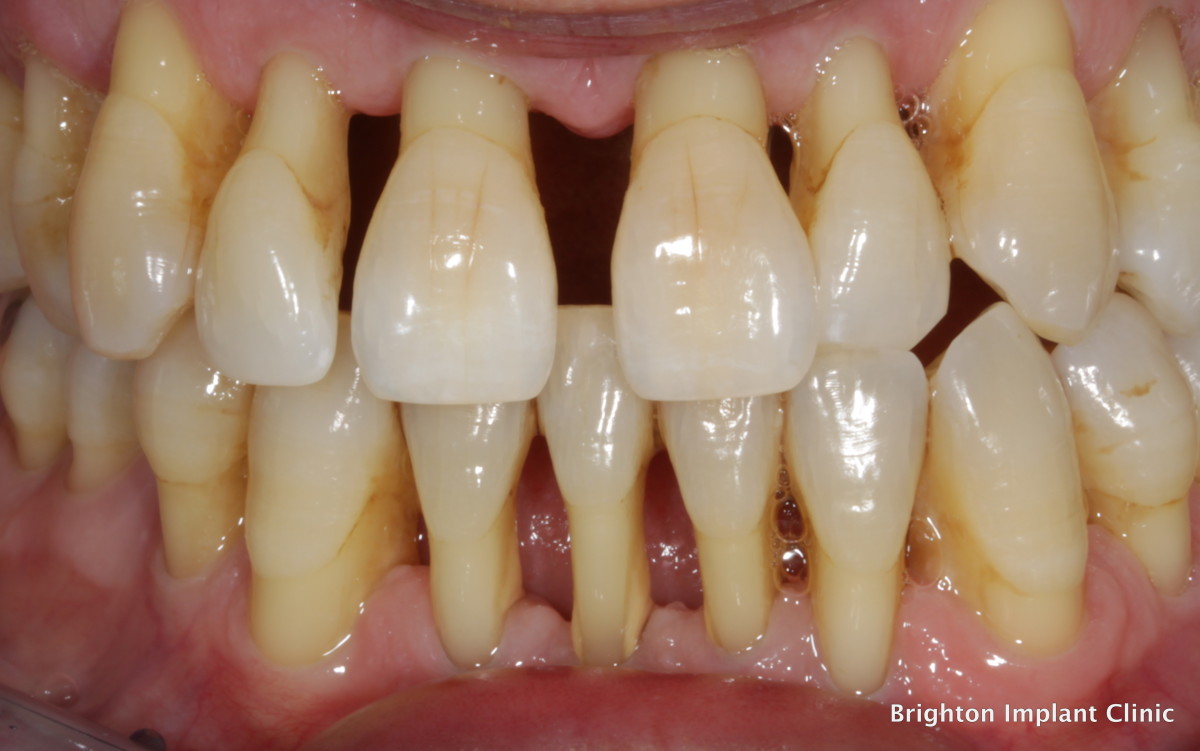
Too much fluoride causes fluorosis...
Consuming too much fluoride while the teeth are being formed can lead to fluorosis. I thought it was interesting when the permanent molars on my triplet’s teeth came out stained, just as they were being formed. They hadn’t even started eating sugars yet, I was concerned as to why their teeth were rotten, and I decided to do some research.
I found that fluorosis causes white or brown discoloration or spots on the enamel or the tooth surface. The effects can range from minor discolorations to surface irregularities of the teeth. Fluorosis does not develop after the teeth have already erupted. It is mostly a cosmetic concern.
My children were pre-mature and I made sure they never drank the faucet water which contains fluoride. I purchased bottled water that had fluoride added in it. Every day, I would only give them the allowed amount of this fluoride water along with their normal meals, milk, vegetables, juice, etc…
By recently doing all this research, I have found that everyday foods and drinks already contain fluoride. Some items do not have labels, like tomatoes, cucumbers and apples. What was even more concerning are the labeled food do not even mention the word fluoride on the label, although they do have it. A common can of Coca Cola has fluoride in it, milk and cereals.
Fluorosis is a cosmetic condition, not a disease. Often, it is so mild that only a dental professional can detect it. Most cases of fluorosis result from young children taking fluoride supplements or swallowing fluoride toothpaste when the water they drink is already fluoridated.
If you happen to see this discoloration in your children’s teeth, the best thing to do is seek a dental professional and inform them of your child’s fluoride intake. This will help them determine if the discoloration seen is a result of fluorosis. Only a dentist can determine the difference between fluorosis and tooth decay, through proper examination of the teeth and gums including X-Rays.
Keep in mind that the spots left behind from fluorosis are permanent and can only be cosmetically repaired by resin composite, they may also darken over time.
What can I do to avoid this?
If you have children at home, make sure to teach them how to properly brush and you only use a small pea-sized amount of toothpaste on their tooth brush. Make sure and teach them to spit and not swallow after brushing. What I did is made sure I purchased non-fluoride toothpaste, just in case they swallowed, I also made sure to avoid sweet tasting toothpaste when I decided to teach them to spit.
Although adding fluoride to drinking water has been one of the greatest preventive measures of the 20th century, it’s important to make sure your child doesn’t over fluoride since fluoride has also been added to many other everyday consumables. If you believe your child’s teeth are stained and this is a concern you were not aware of, consult with your dentist and pediatrician immediately, if you don't have a dentist, make sure you select a good one, I am so glad I did.