Food Safety Ozone Molecules - What You Must Know for the New Water Technology
The terms "ozone" and "unstable" and "poisonous" are often used in the same sentence. Sadly I was even able to find them used in a formal definition of ozone (see the definitions provided below). And yet, I know that my hot tub uses ozone. Additionally, many commercial swimming pools use ozone. Not to mention that ozone was approved in June 2001 by the FDA for bottled water. So why all the negatives about ozone? Let's explore the real facts in more detail.
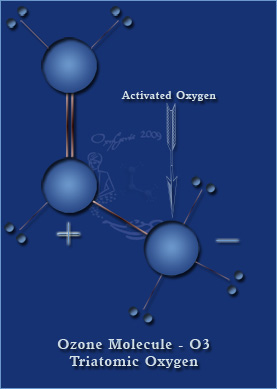
First what is ozone? Ozone is in chemistry "an allotrope of oxygen". Ozone is three atoms of oxygen rather than two. If you recall from chemistry oxygen is made of 2 atoms.

Ozone and Computer Technology - What It Means for Food Safety
In the late 1920s, early 1930s, Siemens introduced a portable ozone generator to the United States' military. Sadly, nothing more was ever mentioned about using ozone as a sanitizer for the military. Rumors bubbled about the un-stability of ozone. Confusion with atmospheric ozone and the measurement of ozone levels being reported in our urban centers. The report of ozone level is actually reporting the fighting power of ozone. You see, we cannot measure the millions of contaminants in our atmosphere, we can measure when there is a greater level of ozone. Ozone combats chemicals, ozone provides a protective level of UV protection that allows us to live on planet earth.
The negative rumors surrounding ozone, I can only surmise arose from the medical community and the testing on the military fields during the early part of the 20th century. What is wonderful, is the 21st century has married computer technology with ozone and now the level of the ozone can be effectively measured and regulated.
A well designed ozonator includes indicator lights that show when ozone is working.
Yes, ozone has been around as long as our earth, billions of years. It was discovered in the 1880's and it was harnessed with computer technology and now is safely available for the retail food industry to help prevent food illness and for home use. The best part of this new technology is the added safety and assurance that the smart internal chips provide in conjunction with the indicator lights.
Remember, the operation of the ozonator does not eliminate the need for proper food preparation and safety. Additionally, remember, ozone only works with cool water. This is contrary to what we were raise with - mom taught us to use warm water. Warm water with ozone lessens it effectiveness.
Understanding history is always helpful to decision makers. Understanding the migration of technology and its affects upon our food safety is an essential ingredient in living healthy lives and preventing food illness.

Food Safety - Ozone Molecule
Food Safety - Ozone Molecule

Ozone Definition in Chemistry
Ozone is an allotrope of oxygen.An allotrope refers to one or more forms of an elementary substance.
For example, graphite and diamond are both allotropes of carbon. O2 and ozone, O3, are allotropes of oxygen.
source: www.Chemistry.about.com
Ozone

Bing. com Ozone Definition
"ozone Definitiono·zone[ ṓ zn ]To hear the pronunciation, install SilverlightNOUN 1. form of oxygen: a gaseous form of oxygen with three oxygen atoms per molecule, formed by electrical discharge in oxygen. Use: water purification. O32. fresh air: fresh pure air ( informal )[ Mid-19th century. Via German Ozon < Greek ozon, neuter present participle of ozein"smell"; from its pungent smell ]"
Ozone Health Standards
Food Drug Administration(FDA) requires ozone output of indoor medical devices to be no more than 0.05 ppm.
The Occupational Safety and Health Administration (OSHA) requires that workers not be exposed to an average concentration of more than 0.10 ppm for 8 hours.
The National Institute of Occupational Safety and Health (NIOSH) recommends an upper limit of 0.10 ppm, not to be exceeded at any time.
EPA’s National Ambient Air Quality Standard for ozone is a maximum 8 hour average outdoor concentration of 0.08 ppm (see - the Clean Air Act - www.epa.gov/air/caa/title1.html#ib)
Oxygen and Ozone
Oxygen is the very air that we breathe and essential to life. The third atom when added to oxygen become ozone. It is this third atom that is often labelled "unstable" because it can alter other chemicals and attach itself. Ozone is said to be very reactive. Ozone is the second most powerful natural antimicrobial known to man. It is Mother Nature's method of eliminating bacteria.
Can ozone be harmful? Yes, as in many items, in high concentrations ozone can be harmful. Breathing high concentrations of ozone can be disruptive and harmful to the lungs. And the EPA has set specific guidelines for our safety.
Can ozone be a toxic gas? Yes, in high concentrations.
The terms used for ozone are misleading. Is ozone always a toxic gas? In very low dosage and the EPA has deemed low dosage as less then 3 parts per million.
Other misleading terms are energized oxygen, condensed oxygen and pure air. These are creative terms sadly often used to hide the term "ozone". Let's recognize the science, read the approval letters given by the EPA for the effective and safe use of ozone and quit camouflaging what the second strongest natural antimicrobial is - it is ozone.
In atmospheric ozone, ozone is not the same as smog. Smog is air pollutions created by combustion polluters. While smog contains small amounts of ozone, it is largely composed of harmful chemicals such as carbon monoxide. In fact, smog and other pollutants may contribute to the damage of the ozone in the upper atmosphere known as the stratosphere. In the stratophere, ozone is the antimicrobial that also serves to to sterilize or rid the atmosphere of bacteria but also ozone is the the shield for protecting the earth from harmful ultraviolet radiation. It is ozone that blocks harmful UV rays. Remember SPF protects us from UVB, but it is the UVA rays that cause skin cancer.
Friends of ozone often hurt ozone by claiming cures. Ozonated water and ozone therapy have no medical basis for any "cure". The cures purported are anecdotal - some one's opinion which is unsubstantiated. Ozone can disinfectant. Ozone can reduce bacteria. Ozone in high doses can be toxic. We need to learn and understand......
The full truth about ozone is that ozone is not always a toxic gas. The truth is ozone is the second strongest natural antimicrobial know to man! It is over 3,000 times more.powerful than chlorine. It has been clinically proven to attack even the difficult and very harmful bacteria MRSA, salmonella and listeria.
I love my hot tub! And I was thrilled to find out that it included ozone as a disinfectant. Technology has changed, microprocessors are now available to assist with the safe deliverance of highly effective ozone. Now is the time to recognize the term ozone for what it is - an allotrope of oxygen which can be unstable and can be toxic in high doses but also can be harness and used effectively for drinking water, bottled water and hot tubs too!
Ozone Approved for Bottled Water June 2001 by United States FDA
Ozone - Breath of Fresh Air
The reference given to ozone as "fresh air" is more accurate to the avid gardener in me®. Leave it to the British to have the proper reference! As an avid gardener, I do love the smell of ozonated water. It really does smell like a thunderstorm. Yes, it is pungent, very distinctive but a delightful smell IF we understand the reason for the smell - it is Mother Nature's way of providing an all natural disinfectant.

Food Safety Knowledge - Cell Lysis - Destruction of the Cell from Inside Out
Definitions from the Free Dictionary
"o·zone (zn)n.1. An unstable, poisonous allotrope of oxygen, O3, that is formed naturally in the ozone layer from atmospheric oxygen by electric discharge or exposure to ultraviolet radiation, also produced in the lower atmosphere by the photochemical reaction of certain pollutants. It is a highly reactive oxidizing agent used to deodorize air, purify water, and treat industrial wastes.2. Informal Fresh, pure air.[German Ozon, from Greek ozon, neuter present participle of ozein, to smell.]o·zonic (-znk, -znk), ozonous (zns) adj.
The American Heritage® Dictionary of the English Language, Fourth Edition copyright ©2000 by Houghton Mifflin Company. Updated in 2009. Published by Houghton Mifflin Company. All rights reserved.
ozone [ˈəʊzəʊn əʊˈzəʊn]n1. (Chemistry / Elements & Compounds) a colourless gas with a chlorine-like odour, formed by an electric discharge in oxygen: a strong oxidizing agent, used in bleaching, sterilizing water, purifying air, etc. Formula: O3; density: 2.14 kg/m3; melting pt.: -192°C; boiling pt.: -110.51°C Technical name trioxygen2.Informal clean bracing air, as found at the seaside[from German Ozon, from Greek: smell]ozonic [əʊˈzɒnɪk], ozonousadj
Collins English Dictionary – Complete and Unabridged © HarperCollins Publishers 1991, 1994, 1998, 2000, 2003
ozone (zn)An unstable, poisonous allotrope of oxygen having the chemical formula O3. Ozone forms in the atmosphere through the process of photolysis, when ultraviolet radiation from the Sun strikes oxygen molecules (O2), causing them to split apart. When freed oxygen atoms bump into and join other O2molecules, they form ozone. Although ozone is broken down naturally in the atmosphere through chemical reactions with other atmospheric gases (such as nitrogen, hydrogen, and chlorine), in an unpolluted atmosphere the formation and breakdown of ozone is generally balanced, and the total concentration of ozone is relatively constant. The formation and destruction rates of ozone vary with altitude in the atmosphere, and with latitude. Most ozone forms in the 15 to 30 km (10 to 19 mi) altitude range and in latitudes closest to the equator where sunshine strikes the Earth the most. The ozone is then transported northward and southward by wind and is generally most concentrated in areas above the Canadian Arctic and Siberia and above Antarctica. Ozone is used commercially in water purification, in air conditioning, and as a bleach.A Closer LookOzone is both beneficial for and threatening to all of Earth's organisms, including human beings, depending on how high in the atmosphere it is found. Ozone is naturally produced in the stratospheric portion of Earth's atmosphere (in the ozone layer) by the action of high-energy ultraviolet radiation on molecular oxygen (O2 ). By absorbing much of the Sun's ultraviolet radiation, the ozone layer serves as a sunscreen for organisms on Earth. In recent years the ozone has thinned or disappeared in parts of the ozone layer, creating an ozone hole that lets in dangerous amounts of ultraviolet radiation. Ozone holes are caused in part by the release into the atmosphere of industrial and commercial chemicals, in particular the chlorofluorocarbons (such as freon) used in aerosols, refrigerants, and certain cleaning solvents. Closer to Earth's surface, ozone is one of the so-called greenhouse gases that are produced by the burning of fossil fuels and cause the greenhouse effect. Ozone at ground level is also an air pollutant, contributing to respiratory diseases such as asthma.
The American Heritage® Science Dictionary Copyright © 2005 by Houghton Mifflin Company. Published by Houghton Mifflin Company. All rights reserved.
Thesaurus Legend: Synonyms Related Words Antonyms Noun1.ozone - a colorless gas (O3) soluble in alkalis and cold water; a strong oxidizing agent; can be produced by electric discharge in oxygen or by the action of ultraviolet radiation on oxygen in the stratosphere (where it acts as a screen for ultraviolet radiation)atomic number 8, O, oxygen - a nonmetallic bivalent element that is normally a colorless odorless tasteless nonflammable diatomic gas; constitutes 21 percent of the atmosphere by volume; the most abundant element in the earth's crustgas - a fluid in the gaseous state having neither independent shape nor volume and being able to expand indefinitely Based on WordNet 3.0, Farlex clipart collection. © 2003-2008 Princeton University, Farlex Inc."

Food Safety - Technology Has Changed - So Too the Paradigm of Ozone Needs to Change
Since the discovery of ozone in 1840s, technology has changed. We need to stop hiding the fact that ozone is the technology that keeps us free from bacteria in our hot tubs. We need to consider adopting mandates that commercial swimming pools use ozone or other less corrosive and less harmful antibacteria agents. We need to recognize that chlorine may not always be the right answer.
We need to stop the creative terms that camoflagued the technology that is rightfully and correctly called ozone. The EPA is correct in safeguarding the misrepresentation of "cures". And we as consumers need to take heed when we see a "cure" purported.
Ozone is a tool which like any other tool can be very helpful when used properly. Food safety today needs to look more closely at adding ozonated water to naturally destroy bacteria.
Articles About Water and Ozonated Water
- Tap Water - Corrosive Pipes, Lead Joints
Clean tap water is the foundation for health. All too often we forget the complexities that surround tap water. Yes, we turn on the faucet but behind the faucet is a distribution system and production... - 4 Myths About Ozone Water Exposed-Does Not Cure Cancer
Ozone water is a wonderful addition to the home, however, it frustrates me that there is so much misinformation permeating the Internet. In this brief article, I hope to dispel the myths and clarify what the new product can and cannot do. Ozonated water does not cure cancer.
Articles About Atmospheric Ozone and Ozonated Water
- Ozonated Water Uses Shopping Tips and More | CPAP Ma...
Ozonated water has some helpful uses - the difference is no chemicals. Who can be helped? Horses with foot fungus, dental care and maintenance of CPAP machines. - Atmospheric Ozone | Protective Nature of Ozone | Gre...
Mother nature kills pollutants with an action called ozone. It is a non-chemical purifier and leaves no residue. The sanitizing occurs in the upper atmosphere. It is ozone that is a friend to both air and water. It is ozone that you smell after a thu