Why the Pressure Cooker?

Point for the No-Turkey-Cookers
When I was a freshman in college, our professor asked us at Thanksgiving time who all had ever cooked a turkey. Every girl in the class raised their hands. Not a single guy. Thus when we divided into teams, the women were the Turkey-Cookers, the men were the No-Turkey-Cookers. It was a genuine fun competition not intended in any way to be sexist and has little relevance to this hub.
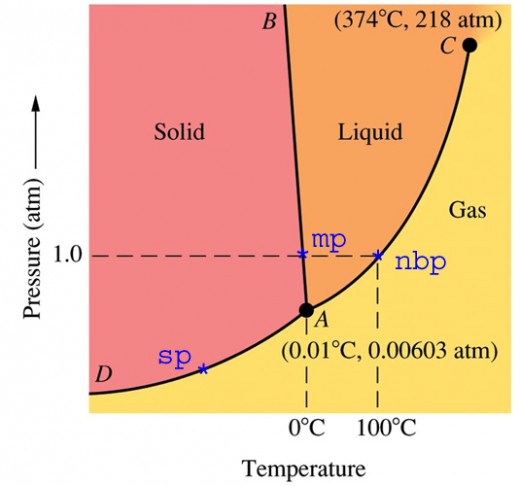
Anyway, I don't know if any of you have ever seen a phase diagram for water. I've put one to the side for your reference. Most people don't really understand how it works. On the vertical axis is pressure and on the horizontal is temperature. In the graph you see three zones (phases): solid, liquid, and vapor. On the lines is where two phases exist in equilibrium.
One point that I want to focus on is the equilibrium point between vapor and liquid at 212 fahrenheit - the boiling point of water. Many people use boiling water to cook things such as eggs and potatoes. Why then are pressure cookers sometimes used?
You see, boiling water at atmospheric pressure will never get hotter than 212 degrees. If you live in the mountains its even lower than that. A trick to raise the boiling point (and thus cook your food faster or more thuroughly) is to put the boiling water under pressure. By increasing the pressure even a couple pounds per square inch you can raise the boiling point of water dozens of degrees, depending on how high the pressure is. Many of you might have known a pressure cooker does this, but probably didn't know why.
Ta-dah. Simple explanation, right? You want to know another trick? Adding salt also lowers the vapor pressure (which means raises the boiling point). So if you add a lot of salt to your pressure cooker, the water inside will reach a much higher temperature than just 212.
Point for the No-Turkey-Cookers.
P.S. Note how the equilibrium line for the ice-water line is slanted backwards. This means that if you have ice and put great pressure on it, it will melt even though you didn't raise the temperature. How cool is that? Two points for the No-Turkey-Cookers.



