Medical Importance And General Considerations In Coagulation And Fibrinolysis
Coagulation Cascade
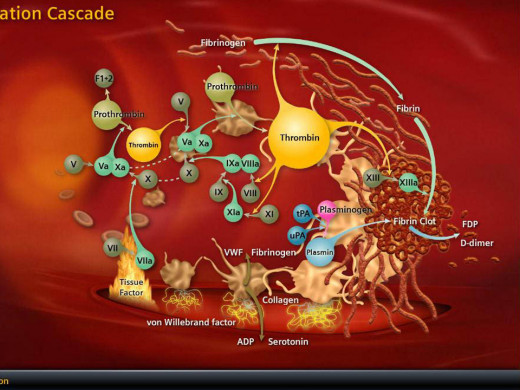
Coagulation
Nomenclature of the coagulation proteins has been accepted internationally and they are as follows:
- Factor I: Fibrinogen
- Factor II: Prothrombin
- Factor III: Tissue thromboplastin
- Factor IV: Calcium ions
- Factor V: Proaccelerin-labile factor
- Factor VII: Stable factor- Proconvertin
- Factor VIII: Antihemophilic globulin (AHG, AHF)
- Factor IX: Plasma thromboplastin component (PTC), Christmas factor
- Factor X: Stuart-Prower factor
- Factor XI: Plasma thromboplastin antecedent PTA
- Factor XII: Hageman factor
- Factor XIII: Fibrin stabilizing factor.
The numerical order does not represent the sequence of activation. In addition to the above, there are other recently described factors such as prekallikrein (PK) and high molecular weight kininogen (Kin) which are involved in the activation of factor XII and XI respectively, in the intrinsic pathway. The activated factors are represented with the suffix ‘a’.
All the coagulation proteins except factor VIII: C are synthesized mainly in the liver. Endothelial cells including those of hepatic blood vessels secrete factor VIII:vWF polymers. Factor VIII:C molecules form ionic bonds with factor VIII:vWF polymers which provide a carrier for them in the circulation and also prevents their destruction. Factors II, VII, IX and X are produced in the liver and these are termed vitamin- K dependent factors, since this vitamin is necessary for making them functional. Thrombin is produced from prothrombin and it converts soluble fibrinogen to fibrin which consists of insoluble polymers. Thrombin can be produced through two different reaction sequence- the intrinsic and extrinsic pathways.
The Intrinsic Pathway
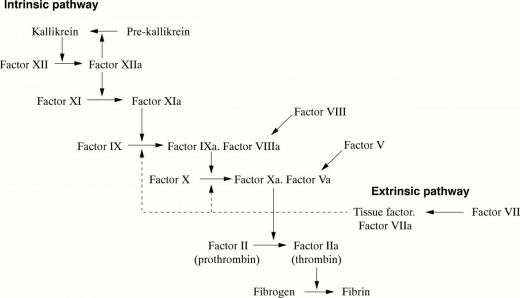
Intrinsic pathways
All necessary components for this pathway are present in circulating blood. This pathway is initiated by the absorption of factor XII (Hageman factor) and kininogen with bound prekallikrein and factor XI to negatively charged subendothelial surfaces, exposed during vascular injury. There after successive activation of factors occurs in a sequential manner. Factors XIIa activates IX to IXa. Activated form of factor VIII:C interacts with factor IXa bound to platelets on their surface. In association with activated factor VIII:C, factor IXa optimally activated and cleaves platelet bound factor X to Xa. Factor Va molecules that have been either adsorbed from plasma and then activated by a platelet protease or released in activated form from platelet alpha granules. This factor Xa-Va complex is adjacent to platelet bound prothrombin molecules. Factor Xa in this complex cleaves the prothrombin molecule into two portions, one part of which is thrombin. Thrombin causes local platelet aggregation and also leads to formation of fibrin monomers from fibrinogen. It also cleaves and activates factor XIII, and this covalently links fibrin monomers into polymers enxymically. Calcium (factor IV) is required at various steps of the whole process.
The Extrinsic Coagulation Pathway
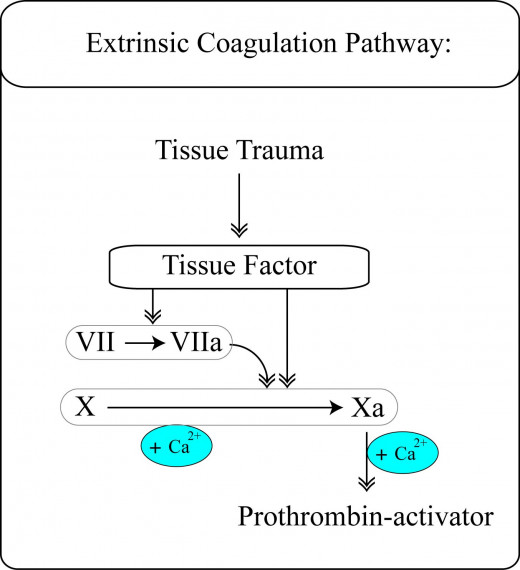
The extrinsic pathway
This is initiated by the entry of phospholipoprotein surface and organelle membranes from disrupted tissue cells. Factor VII is bound to these membranes and activated to factor VIIa. Factor VIIa activates factor X on the surface of the organelle membranes. Attachment of factor V and prothrombin and subsequent thrombin generation occur similar to that described for the intrinsic mechanism. Normally, the extrinsic and intrinsic mechanisms are complementary to each other, the former being more rapid in action. They are all confined to the local site of injury.
Thrombin cleaves fibrinogen molecule to release fibrinopeptides (fibrin monomers) which bind to form fragile polymers. Thrombin activates factor XIII to factor XIIIa which enzymatically converts fibrin monomers into stable polymers which form the insoluble fibrin clot. Polymerisation of fibrin is limited by the inhibition of factors XIIa, Xia, IXa and Xa and thrombin by anti-thrombin III (heparin cofactor) present in blood and endothelial cell surfaces. Another inhibitor is protein C which is a vitamin K dependent enzyme also present in blood and on endothelial cell surfaces. Protein C, after activation by thrombin, also cleaves and eliminates the activated forms of factor VIII:C and factor V. Deficiency of antithrombin III leads to thrombotic tendencies.
Fibrinolysis
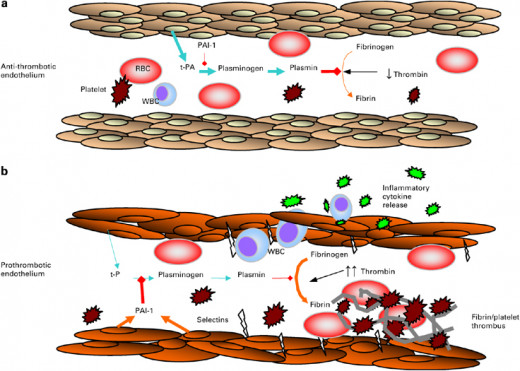
Fibrinolysis
This is the process by which fibrin polymers are lysed by the enzyme plasmin derived by partial proteolysis of the zymogen-plasminogen, which is adsorbed to fibrin polymers. Plasminogen activators are divided from the lysosomes of disrupted endothelial and tissue cells. These bind to fibrin polymers and converts firbin-bound plasminogen into plasmin. Plasmin remains bound to fibrinogen and degrades it into soluble fragments- the fibrin degradation products (FDP). In addition to splitting fibrin, plasmin is also capable of splitting fibrinogen and factor V and VIII:C. Activity of plasmin is inhibited by normal inhibitors. These are alpha, antiplasmin which is a rapid inactivator, alpha2-macroglobulin which is a slow inactivator and anti-thrombin. Antithrombin rapidly inactivated plasmin in the presence of heparin.
© 2014 Funom Theophilus Makama