Leptin, Fat Mice and The Science of Obesity
Causes of obesity are biological as well as sociological
As the obesity epidemic gathers momentum, with the resultant human misery and increasing costs to health services, unraveling the science behind appetite regulation becomes increasingly important. The view that overweight people are merely lacking in moral fibre and that more willpower is all that is necessary to solve the problem is no longer taken very seriously in most circles. The increasing incidence of obesity is certainly a combination of factors, including our increasingly sedentary lifestyles, the easy availability of fattening fast food, the increasing stress of life which leads to rising levels of mental distress and illness, which some people try to ameliorate by excessive eating. However understanding the biological pathways that regulate appetite, and how they fail in obese people, could provide important tips in how to help overweight individuals lose weight. Mutant mice have been an important tool in beginning to understand the basis of the physiological mechanism of controlling weight.
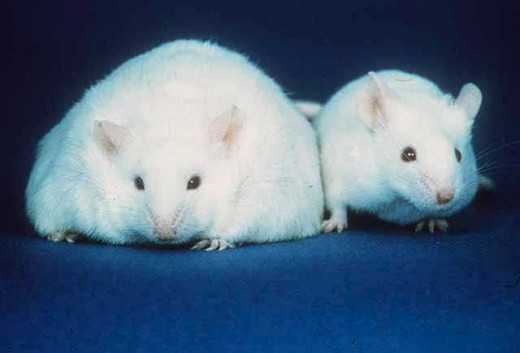
Fat Mutant Mice
The ob/ob (ob stands for obesity) mouse was the first mutation to cast light on the science behind appetite regulation. The mutation arose naturally in the mouse colony in Jackson laboratory in 1949. Although homozygous mice are identical to their wild type siblings at birth, they are always voraciously hungry, and gain weight at a much higher rate ending up 3x the body mass of a normal mouse and with 5x the amount of fat tissue.
Leptin, the Appetite Suppressing Hormone
It was not until 1994 that the mutated gene was cloned by a team led by Jeffrey M. Friedman at Rockefeller University. The gene was named leptin (from leptons the Greek for 'thin') and it codes for a hormone that is produced in adipose (fat) tissue. Leptin is released into the circulation and reaches the hypothalamus, where it exerts its effects in suppressing appetite by, among other mechanisms, inhibiting neuropeptide Y (NPY) neurones, which have a stimulatory appetite. Another mouse mutation db/db which is similar in behaviour and gross obesity was traced back to the genes for the receptors which bind leptin in the hypothalamus. Injections of exogenous leptin into ob/ob mice caused them to lose weight, but had no effect on db/db mice, since they have normal levels of the hormone, but cannot respond to it because they lack the receptors which bind it.
Leptin as a Cure for Human Obesity
The discovery of leptin as an appetite suppressor raised the possibility of developing therapeutics to control appetite in overweight people. A biotechnology company, Amgen, invested tens of millions to develop recombinant leptin as a cure for obesity. However, in trials, the drug was only effective in a small set of obese people, a mutation in the leptin gene is very rare. Since leptin is produced in fat tissue, and the serum levels are proportional to the amount of fat in the body, overweight people actually have higher leptin levels than thin people.
An important question is why, when leptin levels increase with fat, or after a big meal, does the appetite control system fail so easily? There are several theories, none of which have been fully accepted by the scientific community. One possibility is that certain ingredients in the modern diet, interfere with the mechanism. It has been suggested that lectins, which are present in high levels in grains, can bind leptin preventing it from signalling through its receptors in the hypothalamus. There is also some evidence that sleep deprivation depletes leptin levels. However it is also possible that the chronically elevated levels of the hormone in obese people, result in a desensitisation of the receptors, analogous to how people with typeII diabetes have a reduced response to insulin.
One interesting fact to emerge from the research is that very strict dieting leads to a reduction of leptin, which increases appetite. A reason why crash diets often fail. Willpower often looses to the hormone-powered physiological drive to eat when calorie input drops drastically.