New Cancer Drug Kymriah

The New Wonder Drug
Science has come along way in the treatments of disease and seem to have opened the doors for a new drug Kymriah. This is the latest in fighting certain kinds of cancer. Many of the types of cancer Kymriah fights are found in children and young adults.
Kymriah was approved by the FDA back in August 2017. Every dose of Kymriah contains a patient's own immune cells, that are removed and then are sent to a lab to be genetically modified using a virus. The therapy , known as T-cell therapy, or CAR-T gives the cells the ability to recognize and kill the source of the cancer.
This is the first gene therapy drugs on the market right now, with more coming in the very near future.
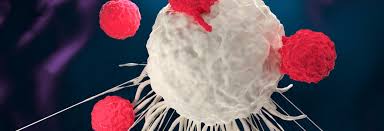
How it Works
Kymriah uses a patient's own cells to attack the cancer. Dr. Stephen Grupp stated,"We've never seen anything like this before and I believe this therapy may become the new standard of care for the patient population". Dr. Grupp is the director of cancer immunotherapy at Children's Hospital in Philadelphia.
They extract a patient's own cells and send them to the lab to be modified and when given back to the patient the drug attacks the cancer cells.
Kymriah comes with side affects, as do all drugs but this one has a life threatening one of possible death from low pressure. The name for this is cytokine release syndrome or CRS. The drug to fight CRS is tocilizumab, which is also a new drug.
Hospitals must be certified to administer Kymriah to patient's. At the moment there are only 20 centers certified to administer Kymriah but they are expecting more than 30 by the end of 2017.
For more information: http://www.cnn.com/2017/08/30/health/fda-first-gene-therapy-leukemia/index.html
Cost
Although the drug seems like a miracle the cost is phenomenal. Kymriah is a one time dose and its cost is over $400,000. which people are saying they thought it would be more expensive than that.
This price does not include hospitals generating the personal dose and the administration of the drug. The $475,000. is just for the drug itself.
Novartis, the company making the drug did make a deal that if a patient doesn't respond to the treatment within a month they will not make them pay.
A spokesman for CVS stated the company expects “CAR-T therapy will be covered under the medical benefit rather than the pharmacy benefit, which means that the medical health carrier and not the PBM will determine coverage for the drug.
All and all this seems to be a miracle drug and hopefully the science community continues and cancer in general one day will be a disease of the past..
You can read more here: http://www.fiercepharma.com/pharma/at-475-000-per-treatment-novartis-kymriah-a-bargain-or-just-another-example-skyrocketing
© 2017 Linda L Paquette