Biologics for Psoriasis
What are BIOLOGICS?
Before we dive right into discuss the available biologics (aka "biologic response modifiers) for psoriasis, we first need to quickly define a "biologic drug."
Biologic drugs are somewhat difficult to define. Biologics differ from ordinary chemical prescription drugs. They are more complex molecules which require a far more involved manufacturing process to prepare. They include things like antibodies used to treat certain types of cancers, vaccines used to prevent diseases, interleukins used to treat a variety of conditions from rheumatoid arthritis to ulcerative colitis and TNF (tumor necrosis factor) inhibitors.
Biologic drugs have a high degree of specificity with respect to their target and mode of activity. For example, in the treatment of plaque psoriasis, biologic drugs have been designed to target T lymphocytes, interleukin (IL) -12 and IL-23 and TNF-alpha (tumor necrosis factor).
Biologics are very expensive and highly sensitive drugs which most often are delivered by injection, sometimes by the patient or sometimes by the practitioner.
What is PSORIASIS?
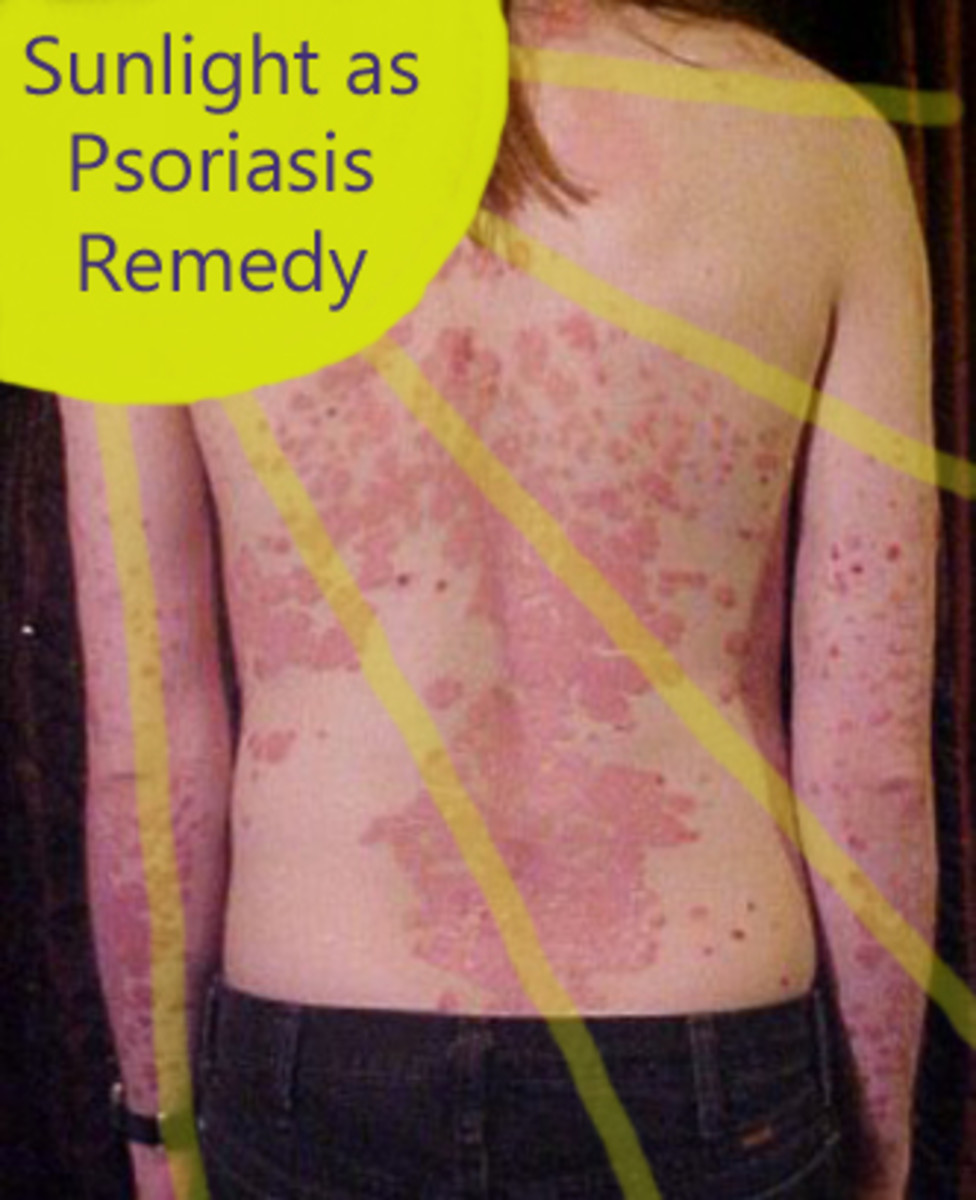
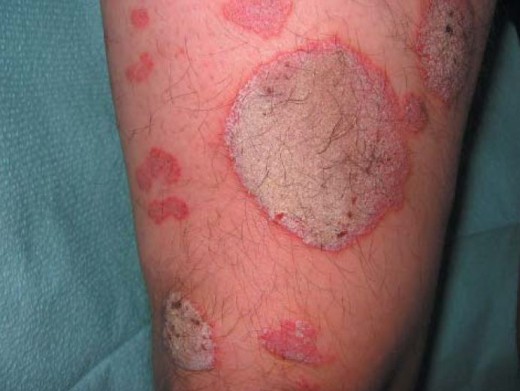
Psoriasis is currently understood to be an autoimmune disorder. The body mistakenly thinks our skin cells are "enemies" (to put it in simple terms) resulting in signals that speed up the process of skin growth. The condition often results in areas of reddened or flaky patches of skin - most commonly on the elbows, knees or trunk of the body. However, psoriasis can be more than a skin condition, often effecting the joints and resulting in arthritis-like symptoms. Anyone can develop psoriasis, though it most commonly arises between the ages of 15-35 and tends to run in families.
There are several distinguishable "types" of psoriasis, each with certain particular manifestations:
- Plaque Psoriasis (the most common form)
- Guttate Psoriasis
- Flexural Psoriasis
- Erythroderma
- Generalized Pustular Psoriasis
- Palmoplantar Pustulosis
- Psoriatic Nail Disease
Approximately 7.5 million individuals in the U.S. have some form of psoriasis, 80% of which have plaque psoriasis.
Certain factors have been found to trigger cases of psoriasis such as trauma, stress, infections or alcohol. Many times psoriasis will develop without a specific trigger. The impact of psoriasis goes beyond the mere physcial effects it produces and also contributes, for many patients, to a decreased quality of life.
Treating psoriasis often begins with topical therapies such as corticosteroids, coal tar or moisturizing therapy. More severe cases will require treatments like phototherapy or oral medications such as methotrexate or immune suppressants like cyclosporine. Cases resistant these treatments may be advanced to consider biologic therapy for psoriasis.
BIOLOGICS for Psoriasis
Biologics are used to treat Plaque Psoriasis which has been resistant to other forms of treatment, or when other treatments cannot be tolerated or are inappropriate. There are currently several biologic drugs on the market which have been approved by the FDA to treat psoriasis. Biologic drugs all carry some significant potential side effects. They differ in their precise dosages and how often they must be administered. Depending on your insurance, they may require you to try a particular biologic for psoriasis first, before you can try a different one. Most biologic drugs will require your doctor to get special authorization (called "Prior Approval") before he/she can order it. Often the drug will be delivered to you directly from a special pharmacy through the mail.
The following are the biologic drugs1 which are currently approved for the treatment of psoriasis:
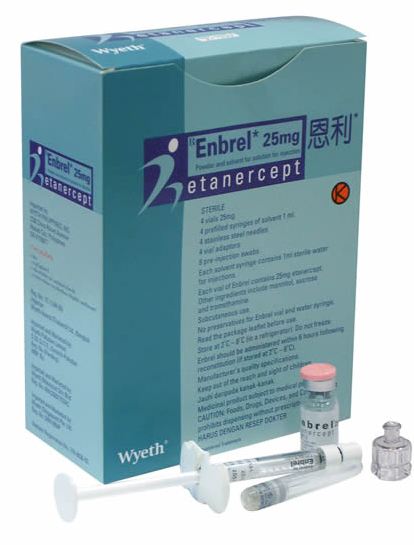
Enbrel (etanercept): Available as 25mg and 50mg dosages in prefilled syringes for subcutaneous injection once or twice weekly.
- For information on the use of Enbrel for psoriasis click HERE
Humira (adalimumab): Available as a 20mg/0.4ml and 40mg/0.8ml prefilled glass syringe for subcutaneous injection typically administered every other week.
- For information on the use of Humira for psoriasis click HERE
Remicade (infliximab): Available as a 100mg single-dose-vial and given exclusively as an IV injection by a doctor. The initial dose is usually given, followed by a dose at 2 wees, 6 weeks and then every 6-8 weeks thereafter.
- For information on the use of Remicade for psoriasis visit their website HERE
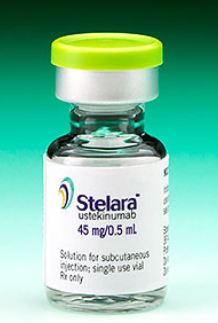
Stelara (Ustekinumab): Available in 45mg/0.5ml and 90mg/1ml vials and prefilled syringes given by an MD. The dosage is usually administered initially, followed by a dose at 4 weeks and then every 12 weeks thereafter.
- For information on the use of Stelara for psoriasis click HERE
Simponi (Golimumab): Available as a 50mg/0.5ml prefilled syringe is generally given once per month. Although not approved for "plaque psoriasis" it is approved for an often associated condition known as "psoriatic arthritis."
General considerations about the above Biologics for Psoriasis:
- These agents are generally approved only for patients over 18 years of age
- These agents are generally approved only for patients with significant (>10%) body surface area (BSA) involvement, or involvement of sensitive areas like the face or hands.
- Because of their effect upon the immune system, risks for serious infection exist. Patients will be monitored for signs of infection. Patients should be aware of the signs of infections like fever, chills, shortness of breath or other flu-like symptoms. Open cuts or wounds pose a risk of infection also. You may be tested for TB infection prior to using one of these biologics for psoriasis.
- Other, more rare, but serious side effects possible with one or more of these biologic drugs include: lowering of your blood count, heart failure, or certain types of cancers.
1. I have not included information about the drug Amevive (alefacept) for the treatment of psoriasis as the manufacturer has decided to stop production of this product.
FAQ - Frequently Asked Questions
But do the Biologics actually work for the treatment of plaque psoriasis?
Numerous studies and patient experience confirm the fact that Biologics do in fact work very well for the treatment of psoriasis. Although the side effects can be serious, the potential benefits and improvement in both symptoms and quality of life are quite impressive.
How fast will they work?
Patient response will vary. Many patients will see improvement within 3-4 months. Results such as a 75% clearing of the skin is not uncommon.
How much do Biologics cost?
Biologic drugs are expensive, costing up to several thousand dollars a month for treatment. Most insurances today will cover biologic drugs for approved patients. Also, the manufacturers of these products also have patient-assistant programs available for those who do not have the resources to afford them.
Do the injections hurt?
Some burning or stinging at the injection site may occur. Allowing the injection to reach room temperature before injection sometimes helps.
Have a Question or Comment to share?
If you have any questions about these medications, feel free to ask below. If you have a personal experience with Enbrel, Humira, Remicade or Stelara that you want to share, feel free to do so also. Thank you for readying and I hope the information about biologics for psoriasis has been helpful.