The Light Reactions of Photosynthesis
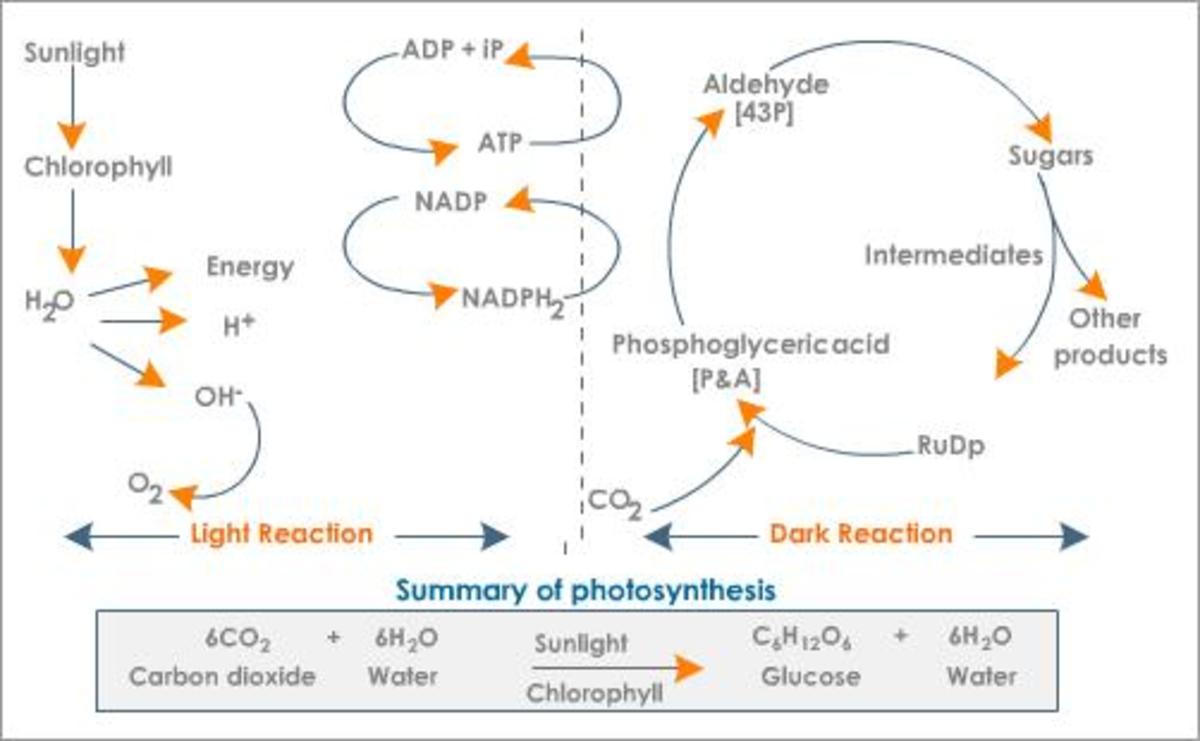
Photosynthesis is a very complex process and so can be different to define at times, as instead it must be described. To illustrate this point, the Oxford Dictionary of Biology (2008) outlines it as: “The chemical process by which green plants and other phototrophs synthesize organic compounds from carbon dioxide and water in the presence of sunlight.” The light reactions of photosynthesis occur in the thylakoid membrane and stroma of chloroplasts and produce NADPH, H+ and ATP [Figure 1].
Figure 2 Light Reactions of Photosynthesis
Click thumbnail to view full-size
Table 1 The Chlorophyll Types Different Organisms Use
Chlorophyll Type
| Organism
|
|---|---|
A
| Bacteriochlorophyll, lower and higher plants
|
B
| Lower and higher plants
|
C
| Protists and cyanobacteria
|
D
| Protists and Cyanobacteria
|
Graph 1 Relative Absorption of Pigments (modification of Bukhman (2008))
Click thumbnail to view full-size
The Photosynthetic Apparatus’ Structure, in the Thylakoid Membrane
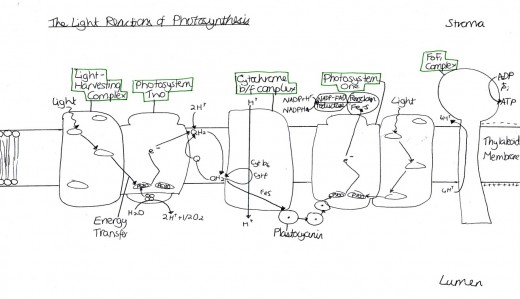
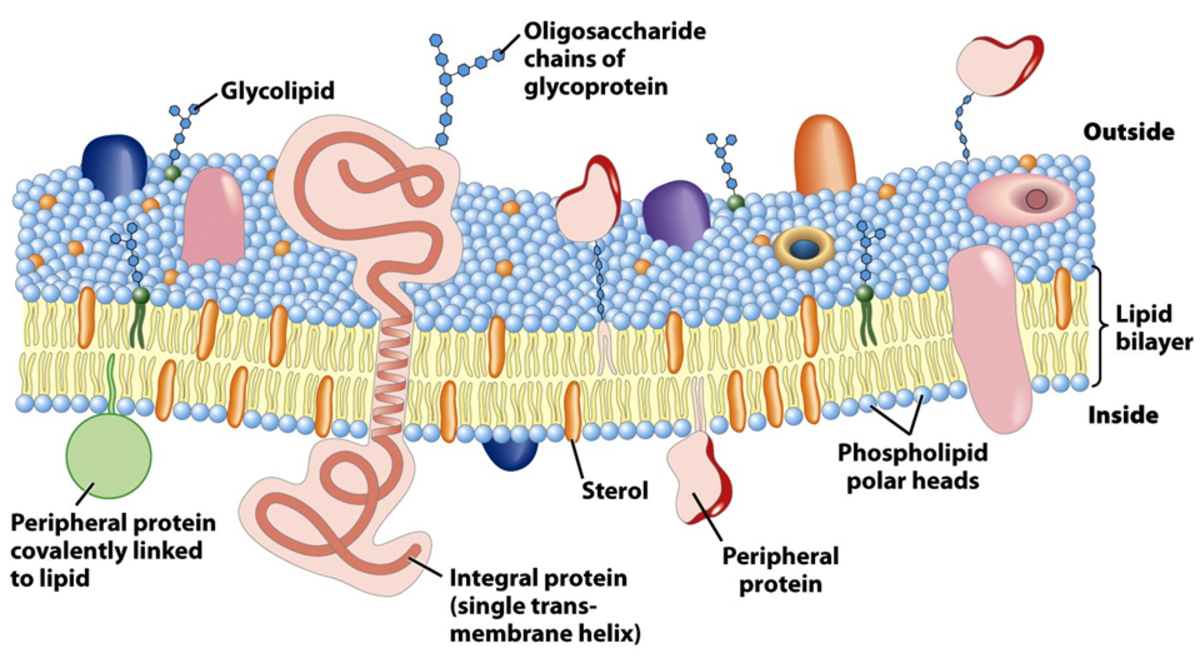
The thylakoid membrane are said to be “the photosynthetic membrane of chloroplasts” (Mauseth, 2008). Thus, the phospholipid bilayer contains some additional features, as well as the usual components (for example, the phosphate polar head and the hydrophobic lipid tail). They contain large proteins, found in both photosystems one and two, named pigment-protein complexes, within which two different types are categorised: the light-harvesting complexes and the reaction centre complexes. There are also cytochrome b6f complexes, which are proteins that are an element of the electron transport chain. The last major protein is that of the F0F1 complex which is necessary for ATP synthesis. They are all displayed by Figure 2.
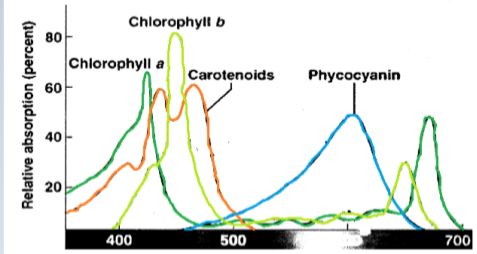
The main pigment associated with the thylakoid membrane’s photosynthetic apparatus is that of chlorophyll. There are several different types of chlorophyll which are utilised by different organisms [Table 1]. It is a green pigment which appears to be so because it tends to absorb other wavelengths of visible light but instead reflects green. This can be clearly seen if an absorption spectrum is plotted, such as the one below [Graph 1]. They are produced by using a spectrophotometer to measure the wavelength, which then displays a great deal of information about the light energy absorbed by a molecule.
There are other pigments that are also used called accessory pigments. They were discovered because the action spectrum (measured using the evolution rate of oxygen to show a biosystem’s maximum response to light), of photosynthesis does not match that of chlorophyll a. Thus, they absorb additional wavelengths of light and then pass on the energy to chlorophyll a. An example of these are carotenoids which have an absorption bandwidth of 400-500nm and so are red, orange or yellow in appearance. They also include all chlorophyll types excluding a. Another accessory pigment is that of phycocyanin which have an especially good absorption rate at 620nm and so are usually light blue [Graph 1].
Figure 3 Diagram of a True Leaf
Click thumbnail to view full-size
Figure 4 Diagram of a Chloroplast
Click thumbnail to view full-size
How Light is Harvested
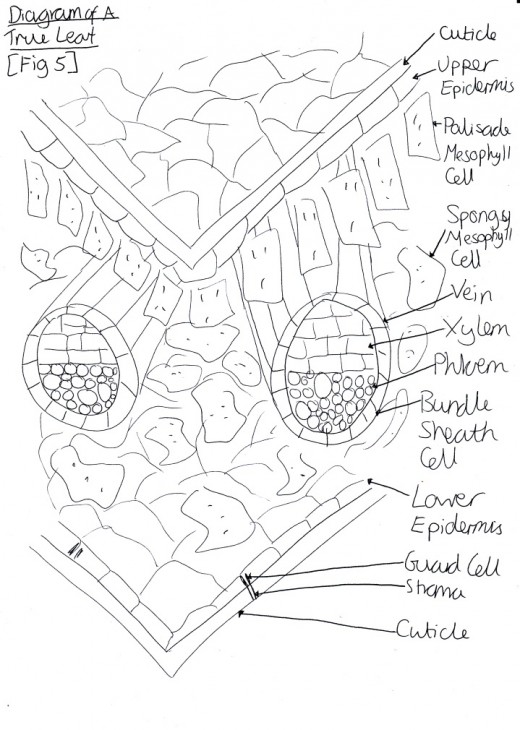
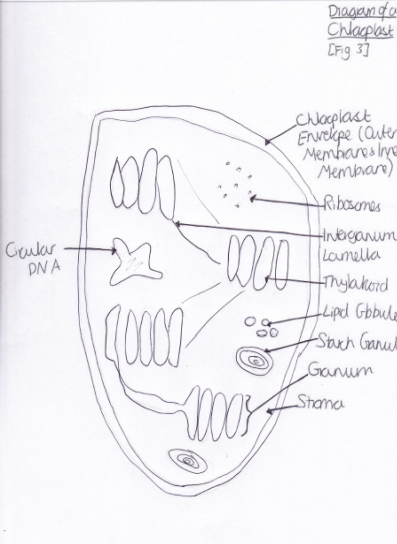
Light is harvested through a type of pigment-protein complex called the light-harvesting complex [Figure 2]. They contain chlorophyll molecules and accessory pigments (e.g. xanthophylls and carotenoids) which absorb light photons from a wide area and then pass it on in to a reaction center, through resonance energy transfer. Within this center there are a pair of chlorophyll a molecules (which, in photosystem one they are named P700 because they are best at absorbing 700nm wavelengths of light). Both chlorophyll molecules a and b are highly efficient at this energy transfer because they are tightly packed in the membrane, as a result of having hydrophobic phytol tails and virtually indistinguishable porphyrin ring structures which are able to lie parallel to each other. This means that the orbits of their electrons are very close and so interaction is easy, with very little energy being lost in the process, since these electrons are able to migrate so freely around. However other molecules, such as carotenoids, have a poor efficiency of resonance energy transfer because only 10% is successfully passed on. And so, plants have adapted to increase the amount of light which they are able to use through various methods. One such example is that the cuticle of the leaves are transparent to allow light to go through, because the epidermal cells have no chloroplasts (thus no pigments to colourise them) [Figure 3]. Also, chloroplasts have single sheets forming numerous small interconnected flattened stack or grana to increase the surface area for light absorption [Figure 4]. On the other hand, Chalker-Scott (2010) points out that plants can sometimes receive too much light to the extent that they ‘accumulate the light energy faster than they can transfer it to chemical energy’, which is known as photooxidation. When this occurs, the existing leaves become bleached and either adaptive strategies are used (e.g. producing small, thick leaves) or the plant dies.
How Water is Split Using Light Energy at Photosystem Two
As the processes of photosynthesis occur, electrons are lost from photosynthesis two and so they need to be quickly replaced. This is done by breaking down water into electrons, protons (H+) and O2, through a reaction called photolysis. The electrons take the place of the previously used electrons while the protons are used in the synthesis of NADPH. The leftover oxygen atoms pair up and then are discarded through the stomata during transpiration. This happens when the P680 chlorophyll molecules within photosystem two have enough excitation from energy being passed to them from other chlorophyll molecules or accessory pigments. However the exact workings behind this is not entirely known yet. In 2004 there was a breakthrough in the understanding of its workings when Iwata et al explained current research “lower resolution X-ray crystallography… have shown that the splitting of water occurs at a catalytic centre that consists of four manganese atoms”. This could greatly help our understading of this pathway in the future, because we have yet to understand how these elements play a part in the photolysis. It is especially an area of interest because, not only will it help scientists understand how photosynthesis works better but it could also provide information about how to use hydrogen as an efficient fuel source for the future.
Water, in particular, is used because of a great many useful properties it possesses. It is available in large quantities (it consists of almost 70% of the Earth’s surface [USGS 2011]) and so can be collected for photosynthesis without wasting too much energy. This an especially important point when you remember that all plant life evolved from completely aquatic organisms e.g. the photosynthetic bacteria 2700-3000 million years ago (Metcalf and Codd, 2004). It is also non-toxic which is especially useful because this means it can be stored without great damage, which is useful for when there is little water available at times but an excess at others. A great extent of this is succulent plants, for example, Ferocactus piosus which is adapted to store water because of its’ harsh environment, since it is a xerophyte. Another stable is that it is suited for photosynthesis because it is a very stable molecule and so can carry much energy.
Figure 5 Explanationary Diagram of PS1 Reactions
Click thumbnail to view full-size
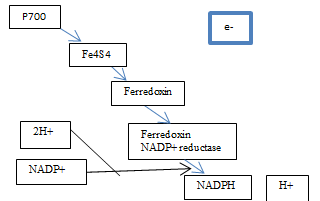
How NADPH is Generated via the Functionings of Photosystem One
The excited electrons from the cytochrome complex travel to photosystem one via the soluble electron carrier phatocyanin [Figure 2]. Once there they excite the pair of P700 chlorophyll molecules at photosystem one and the energy is absorbed by Fe4S4 which is an electron acceptor that is bound to the membrane. An electron acceptor allows electrons to be bound to it and so has the potential to become reduced. When Fe4S4 absorbs the energy, it develops into a strong but unstable reducing agent that quickly reduces the protein ferrodoxin. According to the Oxford Dictionary of Biology (2008), reducing agents are ‘compounds that tend to undergo … [oxidation] readily.
The same (this passing of electrons and so reduction) then happens between the ferrodoxin and ferrodoxin-NADP+ reductase (Mauseth, 2008). The previous reactions are known as redox reactions, which is when something has a change in its oxygen state. The ferredoxin-NADP+ reductase is an enzyme that is part of the oxidoreductases family, which works as a catalyst to aid the synthesis of NADPH+. Thus, it gets combined with 2H+ from the photolysis of water in photosystem two and the co-enzyme NADP+ (an abbreviation for nicotinamide adenine dinucleotide phosphate) that gets regenerated from the Calvin cycle in the dark reactions of photosynthesis and acts as a hydrogen carrier. However, since two electrons are needed, the pathway between P700 and ferredoxin-NADP+ reductase has to be repeated twice. This reaction then results in the formation of NADPH and H+, through the reduction of the NADP+ [Figure 5]. Then, the electron lost during this process get replaced by the electrons produced from the photolysis of water in photosystem two and the cycle is able to begin once more.
The reaction is made more efficient because there is little wastage. For example, any excess NAPH is converted back into NADP during the Calvin cycle. This means that the two processes are able to work in harmony to provide the necessary components for their individual reaction types. Regardless of this fact, it is still said that the most energy lost in photosynthesis is lost in photosystem one as light, making the maximum overall efficiency of photosynthesis to be just 33%.
How ATP is formed
ATP is shorthand for adenosine triphosphate. It is a nucleotide that can go through hydrolysis to become ADP (adenosine diphosphate) or AMP (adenosine monophosphate). ATP is important because it is thermodynamically unstable, meaning that it is susceptible to being broken down, however it is also kinetically stable, so it needs a catalyst otherwise it would take a long time for it to break down (University of Arizona 2003). This means that the energy within its phosphanhydride bond can be released with control (since the catalyst can be controlled). Thus, this highlights the great importance of the complex which synthesises it so this energy can actually be released.
Within and above the membrane (in the stroma), is something called a F0F1 complex, otherwise known as ATPsynthetase [Figure 2]. The F0 part of the complex is inside the membrane while the F1 part is the part in the stroma. It is composed of five different sub units, named: alpha, beta, gamma, delta and epsilon. F0F1 contains proton channels in the membrane that contain enzymes for ATP synthesis from ADP and Pi (inorganic phosphate). The ADP and Pi for this reaction come from the Calvin cycle, thus meaning that they are recycled by it.
The energy to run this reaction comes from the flow of H+ from the lumen, through the complex and then out to the stroma, down an electrochemical gradient. To enable one molecule of ATP to be produced, three protons have to flow through the F0F1 complex. This H+ comes from the photolysis of water and then is the end product, after NADPH is produced in photosystem one. However this path is not always necessary. In cyclic electron transport, chloroplasts are able to produce extra ATP without synthesising extra NADPH.