Batteries- Works and Benefits
Baghdad Battery

Intro to Batteries-Ancient
The lights go off in your house, the electronics don’t work. What is the first thing that you do? You grab a radio and listen to the news. Well how does the radio work without the electricity? Simple. It uses batteries. What's the one thing that you need for an emergency kit. Batteries! Batteries are something that we all use. They power our cell phones, watches, and other electronic devices. There are many forms of batteries and each type is unique. After reading this you will inform you about the history, the science and the uses of three different types of batteries; the Baghdad battery, Nickel Cadmium Batteries and Lithium Ion Batteries. Not all batteries work the same.
How do batteries work you may ask? Well a battery generates electricity using two metals, a positively charged metal called a cathode and a negatively charged metal called an anode. Both metals are in contact with an electrolyte or acid. The oldest battery found dates to about 2000 years ago. It is called the Baghdad or Parthian Battery, because it was found near modern day Baghdad and dates to the Parthian period. The Baghdad battery is simple and easy to create. As you can see it is a clay pot, filled with vinegar (which is a weak acid) and has an iron rod as the anode and a copper cylinder as the cathode. This battery can generate about 1.5 to 2 volts of electricity. Why did people thousands of years ago needed a battery? Well according to Dr. Arne Eggebrecht, a German researcher, these batteries are believed to be used to electroplate items with metals such as gold. Baghdad batteries may not be practical for every day use, but the next battery I will talk about will be more familiar to you.
NiCd Batteries

Nickel Cadmium Batteries
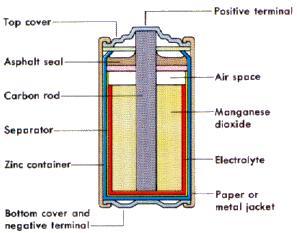
When we think of a battery we imagine a AA battery. A popular AA battery is the Nickel-Cadmium Battery. It was invented in 1899, by a Swedish scientist named Waldmar Jungner. The NiCd battery works similar to Baghdad battery but in a smaller form. They use a nickel hydroxide rod as the cathode in the center and Cadmium as the anode. In between these two metal is an electrolyte acid. Giving you the positive and negative ends. They come in many voltages you may know as AAA, AA, C, D and the square 9 Volt. We use NiCd batteries because of its benefits. They are lightweight, have a long shelf life and are fairly cheap. They are the most popular rechargeable battery because they charge fast. The downsides to this battery is that they contain toxic metals that harm the environment. NiCd batteries are used to power emergency medical equipment, cameras, and power tools. While these batteries were used in cell phones and computers, they have now been replaced with the next battery I will be talking about.
Li-Ion Batteries

Lithium Ion Batteries
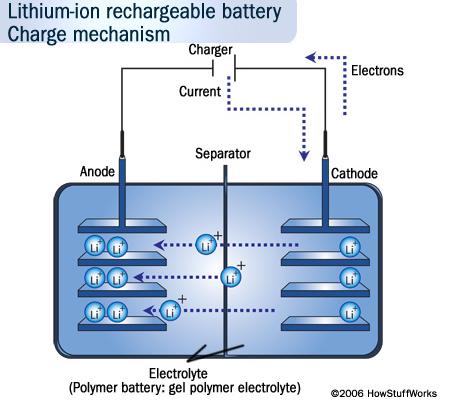
Lithium-Ion batteries were first commercialized in 1991 by the Sony Corporation. However many scientists had experimented with them since the 1970’s. These batteries generate electricity through the movement of lithium ions between the cathode and the anode (as you can see here). The voltage range of these batteries are 3.0 to 3.9 volts for the small types. The reason that lithium-ion batteries are popular is because they are very small and lightweight. They have a higher energy density which means they contain a lot of energy in a small size. The downside to this battery other than the price, is that they can catch on fire. In 2006 The Dell computer company had to recall it’s laptops because the batteries were catching on fire. However, this is a very small percentage of these batteries. Lithium-ion batteries are used in computers, iPods, and cell phones. This is the lithium battery that is in my cell phone, probably similar to the one in your phone. Lithium-Ion technology is still being researched for future uses. The Department of Energy says that “battery research is critical for achieving a clean, secure, and prosperous energy future.” Perhaps a cleaner way to power future vehicles. It may look like lithium is the battery of the future, but since we know batteries evolve this cannot be certain.
Other Batteries
Other types of batteries are Nickel-Metal Hydrides, Zinc-Carbon and Zinc-Manganese, and Silver-Oxide batteries. They all have benefits of their own. most are similar to the Nickel-Cadmium batteries. Silver-Oxides have a short shelflife and have a low energy density. They are used in button cell batteries. Nickel-Metal hydride batteries last a long time with a single charge and long shelf life. Zinc batteries are the cheapest of the popular batteries.
Well now you have just learned about the history, uses and inner workings of three different types of batteries. The Baghdad battery was simple, however it is not practical for modern uses. The Nickel Cadmium battery is much more practical because it can be small and recharged. The lithium ion battery is great because it has a high energy density and there is a promising future for the use of this battery. Hopefully you all have learned more about batteries then you had ever imagined. Now you can either make your own batteries or at the least check the type of battery you are using. If you have always wanted to know how a battery works, well now you know.