5 Ordinary Things with Intriguing Physics
Okay, this is a list for the nerds. Some people go through life just accepting the physics around them without truly appreciating the mechanisms behind it. If you are one of those people, I have other articles you can spend your time on. However, if you’re like me, then you are easily fascinated.

#1: The Cheerios Effect
The Cheerios effect is, in fact, a scientific name for a physics phenomenon. If you’re sharp you might be able to predict what it is.
Did you ever notice how your Cheerios stuck to each other in your cereal? How they stuck to the sides of a bowl? I remember how frustrating this was until I gave up chasing cheerios and just drank straight from the bowl. But why do they do that? Are they infatuated with each other?
The answer is pretty simple. At some point in a science class the word meniscus was/will be brought up. This is a small dip in the surface of water due to water molecules at the surface being pulled on by water molecules below them (stronger than air molecules above the surface water molecules). You can see this effect in a test tube.
Since an object that is floating in water is less dense than water, it will seek the highest point on the air/water interface. This causes floating objects to be attracted to the walls of the test tube. There are other objects or effects that increase or decrease the water level locally, and they also cause floating objects to attract/repel. Cheerios raise the water level around them slightly (because water is pulled by cheerio molecules more than other water molecules), and so adjacent cheerios seek the highest point by clumping together.
The clumping occurs generally with small macroscopic objects that float. A boat will not do this because surface tension is a negligible force at that scale.
Ironically, there is a theory that galaxies cling together for a similar reason.
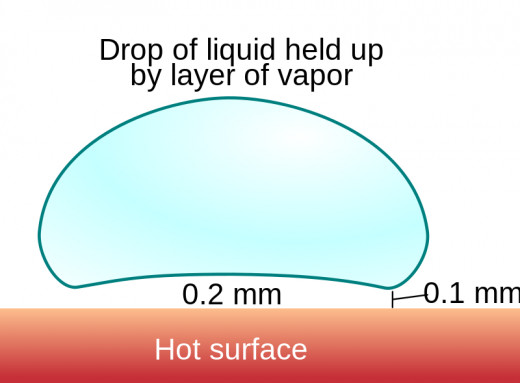
#2: The Leidenfrost Effect
The Leidenfrost Effect is more well-known and a fun one to demonstrate, although some demonstrations are particularly dangerous.
How come a water droplet dropped on an extremely hot pan will evaporate slower than one dropped on just a hot pan? How come you can quickly dip a wet finger into molten lead (don’t try it unless you know what you’re doing!)? How can someone blow out a mouthful of liquid nitrogen?
It all comes down to the Leidenfrost effect. Sometimes the surface a liquid is in contact with is so hot relative to that liquid that only part of it can turn to vapor. This is because the vapor that is immediately created becomes trapped under the rest of the liquid, insulating the rest of the liquid from the surface. It’s diagramed to the right.
Liquid nitrogen can be used in classroom demonstrations because room temperature objects are so hot relative to the liquid nitrogen that the Leidenfrost effect can occur. The temperature at which it begins to occur is known, shockingly, as the Leidenfrost point.
This effect can even be manipulated to make water droplets run uphill or navigate a maze:
#3: The Mpemba Effect
The Mpemba Effect is still out there to be proven, which is shocking. Even in ancient times it was theorized that hot water freezes faster than cold water. It wasn't until the 60's that a Tanzanian student, for which the effect is named, brought this odd quality of water to the attention of mainstream science. It’s been demonstrated in quite a few situations. You can demonstrate it, I can demonstrate it. My mother has maintained this as fact ever since I was a child. I was searching for it on the internet, fully expecting a definitive answer as to whether or not this is a real thing.
I’m disappointed to report that most still question the validity of this effect. They’ll acknowledge it, but we can’t predict it definitively given a set of parameters. Scientists say there are simply too many variables for an easy experimental setup. I doubt this is the case if they can find exoplanets and the Higgs boson. I see it as laziness.
I found an article from October 2013 entitled “Why Hot Water Freezes Faster Than Cold—Physicists Solve the Mpemba Effect,” from a reputable blog, and I was elated. However, it is casually conceded at the very end that the theory in the paper lacks predictive power. In my world, that is an incomplete theory, and an unsolved effect.
There are a couple of possible explanations, taken from this article:
- hot water evaporates faster, reducing the volume left to freeze (seems easily testable?)
- a frost layer forms on cold water, insulating it (seems easily to detect?)
- different concentrations of solutes in hot and cold water
My favorite quote is from a reviewer of scientific literature, who says "Even if the Mpemba effect is real — if hot water can sometimes freeze more quickly than cold — it is not clear whether the explanation would be trivial or illuminating."
This is odd to me because normally scientists jump on experiments that they are unsure whether they will be trivial or illuminating. This is because illuminating experiments win things like Nobel Prizes. Another thing we can laugh at scientists about? We’re still not sure about the physics of a bicycle.

#4: Hot Chocolate Effect
The Hot Chocolate effect was first documented in a physics journal in 1982, but it was one of those things that you’ve probably done. All you had to do was live before then and document the fact that adding powder to hot chocolate while stirring and tapping the side of the glass raises the pitch of the sound of the tap, and you could’ve had a decent paycheck. Or you could have named the effect after yourself and been remembered forever. Okay, I’ll admit, you might’ve had to come up with a couple of equations to describe it, like Newton did with gravity. He was genius by the way.
Anyway, I digress. Why does it happen? Because bubbles that form affect the way sound travels. Sound pitch depends on both density and compressibility of the substance it is travelling through. While the bubbles are in the hot chocolate, the density in the bubbly liquid is much less than normal and the compressibility is much higher (bubbles are compressible). This creates a difference in pitch that can be distinctly heard.

#5: Tea Leaves Float Upstream
This might be cheating, because it's also, like the cheerios effect, due to surface tension.
But it has a whole different story behind it.
Once again, this everyday effect had been failed to be documented by modern science. In 2008, a South American guy was preparing his friend some tea, and noticed the phenomenon. He called up his physicist buddy and they ran some experiments.
The experiment that was most demonstrative for them was also pretty cool. They set two tanks beside each other, and placed a bridge between them. They elevated one of the tanks about 1 cm, so that water began to flow to the bottom tank. When they placed tea leaves and contaminants that mimicked tea in the bottom tank, the tea leaves began to float to the elevated tank.
The secret is that the water in the top tank had a surface tension minutely different than the bottom tank with tea-like substance in it, and so the tea leaves seemingly defied gravity to travel to the top tank. When such a small force such as surface tension difference is exerted on a tea leaf, they still accelerated to 20 times the speed of gravity (since they have very little mass)!
Like I said in the beginning, you might have to be a nerd to care.
The interesting part is that they didn't immediately publish their findings because other physicists were so skeptical. Ironically, the experiment would have cost about $10 at most to run, and the calculations could have been done by a freshman undergrad.