All About Cryosurgery
Cryosurgery sometimes is referred to as cryotherapy or cryoablation. It is a surgical technique in which freezing is used to destroy undesirable tissues. Although the prefix "cryo" is derived from the Greek word "kruos" for cold, usually refers to temperatures below 120K.
Cryosurgery deals with temperatures below the freezing temperature of tissue which is about 273K. It has been developed first in the middle of the 19th century and has now become a fast growing minimally invasive surgical technique for tumor treatments.
Mechanism of cell death
Liquid nitrogen, which boils at -196°C (-320.8°F), is the most effective cryogen for clinical use. It is particularly useful in the treatment of malignant lesions. Temperatures of -25°C to -50°C (-13°F to -58°F) can be achieved within 30 seconds if a sufficient amount of liquid nitrogen is applied by spray or probe. Generally, destruction of benign lesions requires temperatures of -20°C to -30°C (-4°F to -22°F). Effective removal of malignant tissue often requires temperatures of -40°C to -50°C (-40°F to -58°F).
The ultimate goal of the cryotherapeutics is to kill all cells in a diseased target area while causing minimum damage to the surrounding normal tissue. The mechanism of cell death is accomplished by a combination of both direct cellular destruction and anorexia secondary to vascular stasis. Three major phases categorise events at the cellular level during cryo-necrosis; an immediate phase, a delayed phase and a late phase.
Immediate phase
This phase occurs immediately during the freeze-thaw cycle. There are four ways in which the cell may be destroyed, all of them may occur simultaneously. These are ice crystal formation, dehydration and solute concentration, protein de-naturation and thermal shock.
Ice Crystal Formation
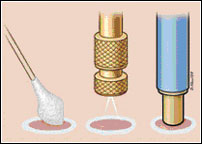
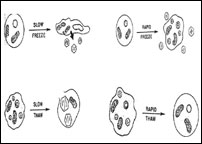
The formation of either intracellular or extra-cellular crystals depends upon the rate at which the target tissue is frozen and thawed. Slow freezing produces large crystals but the crystals are located outside the cell rather than inside where they are more lethal (Fig. A). If the cell is cooled rapidly, it will not be able to lose water so becomes super cooled and eventually forms intra cellular crystals (Fig. B). If the cells are warmed slowly, the small crystals are converted to large, more damaging crystals by re-crystallisation during slow warming than to their formation (Fig. C). If the cells are warmed rapidly, the unstable crystals melt before they have a chance to grow, resulting in the possible survival of the target cell (Fig. D). The maximum amount of cellular destruction results from a rapid-freeze-slow-thaw cycle.
Dehydration and solute concentration
During the freeze cycle the water contents of the tissue, especially the extra-cellular water used to form ice crystals. This results in a hyper-osmolar state with subsequent extraction of water from the intra-cellular space resulting in a toxic concentration of the electrolytes within the cell.
Protein denaturising
The cellular membrane, as well as the membranes of various organelles is made up of lipoprotein complexes. Freezing denature these sensitive lipoprotein complexes, thus damaging or destroying cellular membranes. Loss in phospholipids is a result of freezing renders the cell membrane permeable to ions and extra-cellular fluid so that the cell slowly swells and bursts. A rapid change in the temperature may damage the cell in some instances even before the freezing levels are reached.
Delayed phase
It occurs within few hours of final freeze-thaw cycle. Cellular destruction during this phase is caused by the effects of vascular stasis due to extreme clumping, packing and adherence or RBC to each other and to the vessel wall in the tissue area exposed to the cryogen.
Late immunological phase
There is theoretical possibility of this phase that the destruction of tumours by cryosurgery occasionally leads in increased tumor-specific transplantation immunity. The studies have indicated that cell-mediated cytotoxicity and humoral cyto-toxicity are augmented by cryosurgery. An induced immune response from cryosurgery has been found greater than the surgical excision causes.
Irreversible damage in treated tissue occurs because of intracellular ice formation. The degree of damage depends on the rate of cooling and the minimum temperature is achieved. And inflammation develops during 24 hours after the treatment, further contributing to destruction of the lesion through immunological mediated mechanisms.
Slow thaw times and repeated freeze-thaw cycle produces more tissue injury than a single freeze and thaw. Usually, several minutes are allowed between repeat freeze-thaw cycles. Repeat freeze-thaw cycles generally are employed only in the treatment of malignancy. But continuous freezing at one location for more than 30 seconds beyond when an adequate freeze ball is achieved around the target area can result in disruption of the collagen matrix of the skin and possible scarring.
Cryogenic agents: A cryogen is a substance (generally a gas or a liquid) which has the ability to remove the heat from a given target area. The most commonly used cryogens are liquid nitrogen, nitrous oxide and carbon dioxide. Table 1 shows the types of cryogens now available together with their surface temperature reductions:
Surface tissue temperatures attainable with various cryogens
Cryogens/Temperature (degree C)
Liquid/ 20 (cotton-wool bud)
nitrogen /180 (spray) /196 (probe)
Carbon dioxide snow /78
Nitrous oxide /89
Liquid nitrogen: is the most commonly used cryogen in the cryosurgery. It can be used by spray, swab pour and cryo-probe delivery system. Because of its low boiling point (-196oC), it causes maximum tissue destruction in a short period of time. It has deep penetration ability of available cryogens and is used on large as well as on small lesions.
Carbon dioxide: it is used in two forms: as a solid (dry ice) and as a gas. Both produce probe temperature of -78oC. Dry ice is used in the ophthalmic surgery for the extraction of sub-luxated lenses.
Nitrous oxide: it reaches a probe temperature of -89oC. It is readily available and storable and easily applied, either through a probe or as a spray.
Whilst -20oC is adequate for the treatment of benign and more superficial skin lesions, the subzero temperatures of -50oC are required for the treatment of malignant lesions. Clearly, liquid nitrogen with its boiling point of -196oC has the greatest "lethal" subzero temperature for tissue destruction and as it is now readily available and relatively cheap, it is the cryogen of choice in both primary and secondary care.
Procedure of the therapy
- Operator should wear disposable protective plastic gloves (avoid any possibility of cryogen contacting the skin of involved personnel)
- General anesthesia is usually necessary for any delicate cryotherapy (to allow accurate delivery of cryogen).
- Remove the hair from the area, wash and dry and apply a little paraffin jelly or vaseline for improved initial adhesion.
- If spray is to be used, mask off surrounding area with piece of plastic sheet, or apply white petroleum, Styrofoam cups or x-ray film to prevent freezing of underlying or adjacent tissue.
- Large tumors are debulked as much as possible, prior to freezing to produce a flat surface for easy contact freezing and to reduce the amount of tissue to be frozen.
- Select suitable probe needle for example, 10 or 20mm diameter or spray attachment depending on desirability.
Application techniques
Probe freezing: It is done by direct application of a probe tip to the lesion. The cryogen circulates through the probe tip and super cools it, when allowed to contact the target tissue. Probe freezing can be accomplished by one of the two mechanisms: contact freezing and penetration freezing. In contact freezing, firm contact is made between the cryoprobe and the target tissue. While in penetration freezing probe penetrates the target tissue, providing a large area of direct contact.
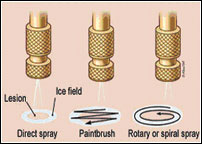
Spray freezing: It is done by direct application of the liquid nitrogen to the tissue and is the most destructive method. Liquid nitrogen is delivered to the target tissue at such a volume and velocity that it evaporates at the edge of the lesion. The spray orifice allows deeper and faster tissue penetration than the probe tip but the probe method is safer and more precise.
Methods of treatment
The dose of liquid nitrogen and the choice of delivery method depend on the size, tissue type and depth of the lesion. The area of the body on which the lesion is located and the required depth of freeze also should be considered. Also the additional factors should be considered which include the thickness of the tissue, the water content of the tissue and local blood flow.
Timed spot freeze technique
The timed spot freeze technique allows greater standardisation of liquid nitrogen delivery. It may be the most appropriate method for physicians who perform cryosurgery. The use of this technique maximizes the ability to destroy a lesion with minimal morbidity. The freezing time is adjusted according to variables such as tissue type and thickness, vascular and lesion characteristics.
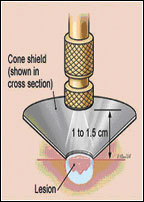
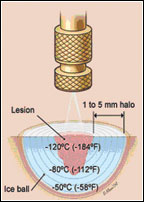
Timed spot freezing is performed with a small spray gun that typically holds 300-500mL of liquid nitrogen. For the standard spot freeze technique, the nozzle of the spray gun is positioned 1-1.5cm from the skin surface in case of skin lesions and aimed at the center of the target lesion (Figure 3). The spray gun trigger is depressed and liquid nitrogen is sprayed until an ice field (or ice ball) encompasses the lesion and the desired margin (Figure 4). The designated ice field may need to be delineated in advance with a skin marker pen because freezing may blur pretreatment lesion margins.
The margin size depends primarily on the thickness of the lesion and whether lesion is benign or malignant. Margins for most benign lesions can extend as little as 1-2mm beyond the visible pathologic border. Pre-malignant lesions need margins of 2-3mm while malignant lesions require margins of 5mm of clinically normal skin to ensure adequate removal. These margin sizes allow enough depth of freeze to ensure temperatures of -50°C to a depth of 4-5mm.
Once the ice field has filled the specified margin, the spray needs to be maintained with the spray canister trigger pressure and thus the liquid nitrogen spray flow adjusted to keep the target field frozen for an adequate time. This time may vary from 5-30sec beyond the initial time for formation of the ice field. If more than one freeze-thaw cycle is required for lesion destruction, complete thawing should be allowed before the next cycle (usually two to three minutes).
This technique also achieves temperatures that are adequate for tissue destruction in an ice field up to 2cm in diameter. The best approach for lesions larger than 2cm (including an adequate margin) is to use overlapping treatment fields.
The cryofreeze - spot freeze
During a cryofreeze, a white "Ice field" is formed on the skin surface. Histologically, the cryofreeze causes ischaemia and Necrosis by direct cellular damage and whilst the degree of tissue damage is partly related to the length of the freeze time employed, other factors play an important part. Different cells and tissues have their own sensitivity to cold.
Other techniques
Variations on the open spray technique include the rotary or spiral pattern and the paintbrush method. These techniques can be useful for treating larger benign lesions. They are not well standardised for ensuring the temperatures that are required for the destruction of malignant lesions.
Cryoprobes
While the open spray technique can be used for the most easily accessible lesions, a cryoprobe (Figure 1) attached to the liquid nitrogen spray gun can provide added versatility, depending on the site and type of the lesion. In this regard various sizes and types of cryoprobes are available. The cryoprobe is applied directly to the lesions. A gel interface medium often is used between the probe and the skin surface.
These are frequently used in the treatment of smaller facial lesions (for example, on the eyelids) where scatter of liquid nitrogen is undesirable. Probes also are useful in the management of vascular lesions where its pressure can be used to remove blood from the tissues thus allowing more adequate treatment.
Tissue temperature monitoring techniques: Target tissue must be frozen to a temperature of -20oC to -30oC. There are two basic techniques for temperature determination, by thermocouples and by visualisation and palpation.
Thermocouples: or pyrometers are the most accurate method of monitoring tissue temperature. Two thermocouple needles are used to monitor the temperature. The temperature sensitive device in the tip of each hypodermic needle is placed at the periphery of the lesion and at the depth of the lesion.
Visualisation and palpation: they make digital assessment of size and position of the ice ball. Only 75 per cent of the visible ice ball can be expected to slough.
Post operative tissue changes: Immediately after freezing, the target area becomes erythematous. Within the next three to four hours, there is considerable swelling and edema which persists for 24-48 hours; this is attributed to vascular stasis, thrombus and ischemia. There is also oozing of serum or blood. If the target tissue was ulcerated before surgery or if the freeze was performed on a biopsy site, hemorrhage may be a complication.
During the initial 24-48 hours, of post operative period, a scab is formed which demarches the necrotic tissue from the healthy tissue. Within the 10-14 days, the scab usually falls off, exposing granulation tissue. Epithelisation and wound contraction progress rapidly and usually are completed by post operative day 21. Following a healing, in to a de-pigmented area of alopecia or white hair (if hair follicles survive) may be observed.
Factors affecting tissue susceptibility to cryonecrosis: Tissue temperature and vascularity: if systemic body temperature and vascularity of the tissue is high; it takes more time to freeze and thaw more rapidly. Whereas, high water contents in the tissue facilitates freezing.
Indications
- Perianal diseases - adenoma, adenocarcinoma, fistula.
- Rectal - polyps, Aden carcinoma.
- Skin lesions:-benign and malignant tumors, exuberant granulation tissue.
- Oral cavity lesions - benign and malignant tumors, epulis, squamous cell carcinoma.
- Ophthalmic lesions - eyelid tumors, corneal ulcers, cataracts requiring cryo extraction of the lens.

Contra-indications
- Lesions located in pre-tibial areas, eyelid margins, nasolabial fold, ala nasi, and hair-bearing areas.
- Proven sensitivity or adverse reaction to cryosurgery.
- Lesion located in an area with compromised circulation.
- Concurrent treatment with immunosuppressive drugs.
- Collagen disease or autoimmune disease.
- Urinary bladder neoplasia.
- Pyoderma gangrenosum.
- Large mast cell tumors.
- Osteosarcoma.
- Nasal tumors.
- Prostatic neoplasis.
- Raynaud's disease.
Advantages of cryosurgery
- Little or no hemorrhage.
- Frequently faster than cryosurgery.
- Relatively free of postoperative complications.
- Phenomenon of cryo-immunity may delay or prevent neoplastic regrowth.
Disadvantages
- Objectionable odour resulting from tissue necrosis.
- Damage to adjacent structures such as joints and bones may lead to lameness and fracture.
- Risk of uncontrolled freezing - resulting in destruction of normal tissue.
Conclusion
Cryosurgery appears to be more effective than current standard treatment options. The complication rate was also lower in this series. It is also a promising modality in patients who are radiation therapy failures. Long term follow-up is still lacking with this procedure. Unfortunately, it will take about five to ten year before we know the true efficacy of this treatment. A long term study is now in progress.