Angiosperm Reproduction
The Oxford Dictionary of Biology (2008) defines angiosperms in a very simplistic way, stating only that they are “flowering plants”. However to properly describe them, their method of reproduction must be properly explained. Thus, a more accurate outline would be that of the Medical-Dictionary (2011), which not only states that they are flowering plants but goes into more detail about the structures and tissues which only they have.
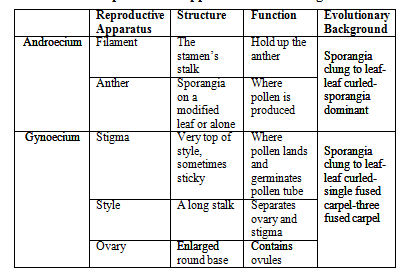
Table 1: Shows the Reproductive Structures, Functions and Evolutionary Background in Flowering Plants
Click thumbnail to view full-size
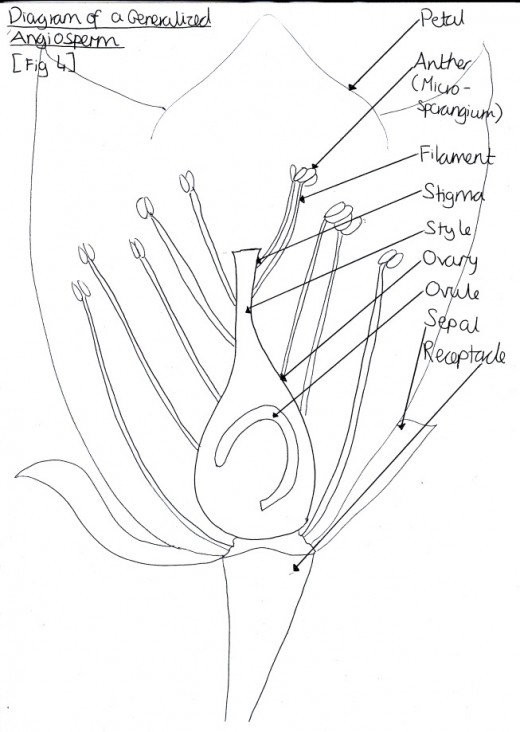
Figure 1: Reproductive Structures
Click thumbnail to view full-size
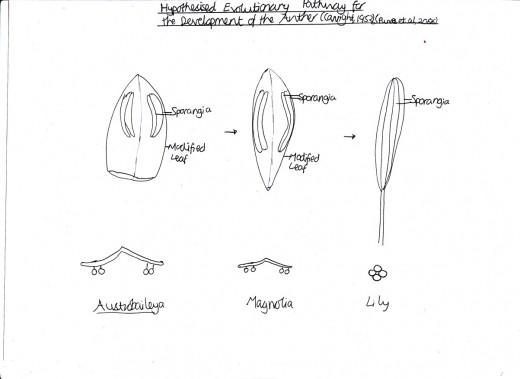
Figure 2: Anther Evolution
Click thumbnail to view full-size
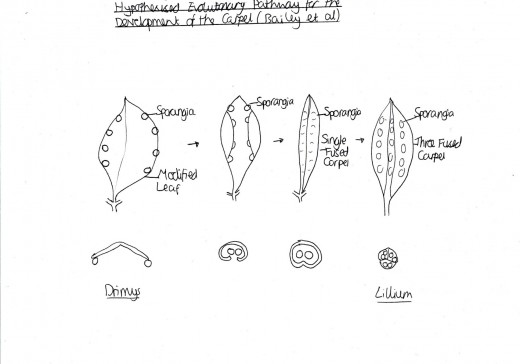
Figure 3: Carpel Evolution
Click thumbnail to view full-size
The Structure, Function and Evolution of the Reproductive Apparatus of Flowering Plants
The reproductive apparatus of flowering plants consists of the male reproductive organs, otherwise known as the androecium, and the female reproductive parts, called the gynoecium [Figure 1].
As shown by Table 1, the androecium is a collective term for the stamen, including the filament and the anther.
The filament holds up the anther to help prevent self-fertilisation. A part of the stamen, called the anther, is where pollen (or microspores) is produced, which itself produces the male gametophytes.
The anther was thought to have evolved from a modified leaf structure where, originally, the long sporangia clung to the leaf e.g. in Austrobaileya (Wet Tropics Management Authority, 2010). In the magnolia, the modified leaf evolved to be less wide and slightly curled until eventually, in the most recently evolved Lilium, the modified leaf structure is hardly visible and instead the sporangia is dominant (Canright, 1952). [Figure 2]
The gynoecium, or carpel, consists of the stigma, the style and the ovary, as Table 1 shows. The stigma is where the microspores land and germinate the pollen tube. The style exists so that there is a sizable period of space between the ovary and the stigma. The ovary contains ovules which contain female gametophytes.
Overall, the carpel is thought to have evolved from filamentous leaves through various mutations over numerous years (Scott et al. 2004). In particular, Canright (1952) suggested that the modified leaf with sporangia of the primitive angiosperm stamen, as found in Austrobaileya, evolved to form a slightly folded modified leaf shape. This then evolved itself to produce a single fused carpel with the sporangia encased by the leaf. This, finally, formed the three fused carpel found in normal angiosperm stamen today e.g. lilium (Botany.com, 2010). [Figure 3]
How Plants Ensure Dispersal of their Progeny in the Environment
To enable plants to reproduce sexually, pollen must move from the anther of one plant to the stigma of the other. There are many ways of doing this.
One such way is through anemophyly, which is the transfer of pollen using the wind (Singh, 2011). The pollen itself is adapted to be smooth small and light so that it can be easily carried long distances by the wind and also so that it can be released from the anther with ease. There is also a great amount produced which, although it is energetically expensive, makes it more likely that the pollen will land where it is needed. Another adaption is that the stigmas are feathery and long. This means that they have a larger surface area to allow the pollen to be taken from the wind (Purves et al. 2004).
A second way of fertilisation is that of entomophyly, which is when pollen is transferred using insects. Figure 1 shows a good example of a flowering plant pollinated in this way. The pollen is sticky so that it is more likely to be picked up by the insect. The stigmas are also sticky so that it will be more likely for the pollen to attach to it. Also, plants are adapted to attract the insects initially. They have some sort of food reward e.g. nectar which is advertised by the plant with attractive scents. However this is not always required e.g., in Ophrys, wasps pollinate the plant because they believe it to be a female wasp and so this is the reward instead (Fay et al. 2008). Another adaptation is that the petals are large, coloured and of an attractive shape to further entice these insects. For example, Hemerocallis petals look like bulls eyes to insects flying above (British Hosta and Hemerocallis Society, 2012).
There are also a number of other pollinators e.g. through the use of birds, water, bats and humans.
An example of a plant specifically pollinated by birds in that of the columbine Aquilegia. Their flowers tilt towards the ground so the animal which pollinates it must be underneath this awkwardly positioned flower-and this is exactly how hummingbirds fly (Koning, 1994).
Plants which are pollinated by bats (e.g. the saguaro cactus, organ pipe cactus and agave plants) are specially adapted for this. They are open at night because that is when bats are mostly active. Also, the flower is large and has a wide shape to allow the face of the bat to be able to fit and so that it also produces a distinct echo when the bats use sonar. To allow the bats to be able to find them to an even greater extent, these flowers produce strong scents to attract them. The bats are also adapted by having a longer wingspan so that they are able to hover over the plant for the required amount of time. They are also able to use their sonar to distinguish between different plants (Eliasinski 2012).
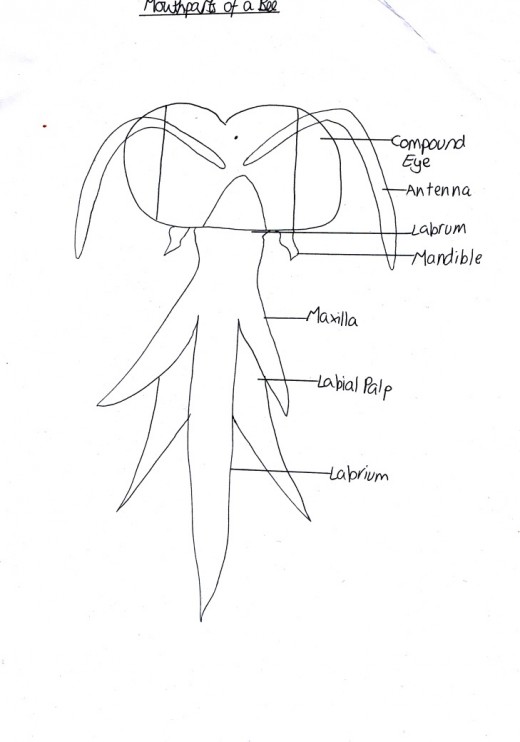
Figure 4: Mouthparts of Bees
Click thumbnail to view full-size
How Flowering Plants Co-evolved with Insects
Biology-Online (2011), defines co-evolution as being the close ecological relationship between two or more species which evolve and adapt to the changes of each other. It can either be mutualistic, so both species gain from the relationship, or predation, where one animal suffers because of it.
The main mutualistic co-evolution is that of entomophyly, which is when insects transfer pollen between plants (Singh, 2011). At first, the earliest gymnosperms and angiosperms were pollinated by the wind and the pollen was held on to using sap. However, at around 65mya the first insects had evolved and they began to use this sap as a source of food, until eventually they became dependant on it (Carter, 2005). Plants benefited with this relationship because it meant they could reproduce over more areas then just those which could be reached by the wind. Therefore, plants evolved to be more attractive to the insects. They can now have special nectaries for the insects as well as large scented and specially coloured petals. For example, many yellow plants (such as the Black-eyes Susan Rudbeckia) give out UV wavelengths of light also because those are the wavelengths that bees can see most clearly and so it attracts them and then they use their modified mouthparts to suck up the nectar, as shown in Figure 4 (RHS 2012).
Another example of mutualistic co-evolution is that of defence, such as the relationship of the Acacias and the Acacia Ants. The ants live on the plant’s thorns and use the edges of the leaflets as their food. In return, the ants defend their plant by attacking other organisms which are near it and by reducing its competition by pruning away other plants nearby (Carter, 2005).
However, this co-evolution is not always mutually beneficial. An example of this co-evolution is the relationship between the Yucca moths and the Yucca plants. It could, at first observation, be seen as a mutualistic co-evolution because the Yucca plant is such a peculiar shape that only that specific moth may enter to pollinate it and in return, the moth is able to have a safe-haven in which to lay their eggs. However, the moth also eats the seeds of the plant thus even though the plant does benefit initially, it is not a mutualistic relationship (Glass, 2003).
Therefore, plants and insects have also co-evolved to enable themselves to deter or avoid each other to reduce the risk of damage or predation. An example of this is the carnivorous Pitcher plants, which are able to digest insects as a source of food. Insects protect themselves from this adaption if they are part of the inquiline community, since some insect larvae are able to live within the plant (where other insects would be digested) and survive.
Bibliography
Articles
Dunn (2009), Pollen, National Geographic Magazine.
Glass (2003), Yucca Flowers and Yucca Moths, A Moment of Science, Indiana Public Media.
Bergmen (2004), Insect Evolution: a Major Problem for Darwinism.
Fay et al. (2008), Ophrys: A Case of the Deceitful Origin of Species, Kew Scientist, Issue 33.
Singh (2011), Short Essay on Anemophily, Preserve Articles.
Books
Mauseth (2008), Botany: an introduction to plant biology, 4th edition, Jones and Bartlett.
Oxford Dictionary of Biology (2008), Sixth Edition, Oxford University Press.
Purves et al. (2004), Life the Science of Biology, Seventh Edition.
Ridge (2002), Plants, Oxford University Press.
Stewart (1993), Paleobotany and the Evolution of Plants.
Willis and McElwain (2002), The Evolution of Plants, OUP Oxford.
Journals
Scott et al. (2004), Stamen Structure and Function, The Plant Cell.
Canright (1952), The Comparative Morphology and Relationships of the Magnoliaceae. I. Trends of Specialization in the Stamens.
Reports
Eliasinski (2012), Bats and Some of their Amazing Adaptations.
Diamond (2002), Evolution, Consequences and Future of Plant and Animal Domestication, Nature Journal.
Websites
Medical-Dictionary (2011), Angiosperms, http://www.medical-dictionary.cc/what-does/angiosperms-mean.
Biology-Online (2011), Coevolution, http://www.biology-online.org/dictionary/Coevolution.
Wet Tropics Management Authority (2010), Plant Diversity - Flowering Plants, http://www.wettropics.gov.au/pa/pa_flowering_plants.html.
Botany.com (2010), Lilium-Lily, http://www.botany.com/lilium.html.
British Hosta and Hemerocallis Society, 2012). BHHS at RHS Hyde Hall, http://www.hostahem.org.uk/.
Koning, Ross E (1994), Pollination Adaptations, Plant Physiology Website, http://koning.ecsu.ctstateu.edu/plants_human/pollenadapt.html.
Carter (2005), Co-evolution and Pollination, http://biology.clc.uc.edu/courses/bio303/coevolution.htm.
RHS (Royal horticultural society) (2012), Rudbeckia occidentalis 'Green Wizard' http://www.rhsplants.co.uk/plants/_/perennials/rudbeckia-occidentalis-green-wizard/itemno.RH30000327/.