Atomic Emission and Absorption Spectroscopy and Flame Photometry: The Test for Sodium and Potassium

A Complete Explanation To Analytical Chemistry By The Leading Author
Introduction
Ever looked at a bottle of water and saw how much sodium or potassium is in it? Ever wondered how these elements were measured? Ever wondered how the doctor knows how much sodium or potassium is in your blood? This article aims to find out.
Atomic Emission Spectroscopy is an analytical technique to determine a quantitative analysis of cation metal salts such as potassium, lithium or sodium in any given sample. These salts are easily excited to a higher energy level. This technique is designed to cause atomic excitation of a sample and then to measure the the intensity of the emitted radiation. It can then determine the level of a salt in a substance. For example, this method could determine the amount of sodium in water. The instrument used to do this technique is known as a flame photometer. This is one of three atomic spectroscopy techniques. The other two branches are:
- Atomic Absorption Spectroscopy-where the flame photometer measures the intensity of light absorbed by the electrons so they can move up to the 'excited' higher level.
- Inductively Coupled Plasma- an expensive, relatively new technique.
Atomic Emission Spectroscopy is a relatively inexpensive and simple method.
Atomic Theory of AES
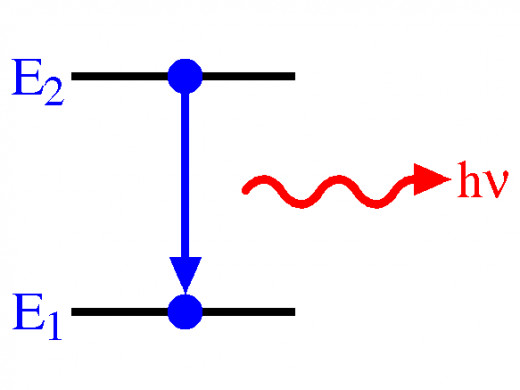
The electrons become excited by a flame in the photometer. When the electrons that are further away from the nucleus (known as free electrons) get excited by the flame, they absorb a certain amount of energy from a specific frequency of the light spectrum and move out to a higher shell of the nucleus. Soon after, they emit the same energy and move back down to their normal energy level or the ground state. This can be shown on the diagram below where E2 is the electron in the excited, higher state and E1 is the electron in the normal or ground state. Hv is the radiation given out by the electron when it moves back down to its ground state.
The Path of the Sample
When you place the sample in the small cup, the sample is passed through a small, narrow tube and is brought to the atomization chamber where the liquid becomes atomized (turned into a fine, misty spray). The spray is then brought to the flame and the electrons become excited. The photometer then measures the intensity of the light that is emitted by the electrons as they return to the ground state. Depending on which flame photometer you use, you might get a spectral line or a direct recording. The spectrum line is produced. The photometer measures the intensity of the line and a recording is shown (usually in mmol/L).

How to Use a Flame Photometer
- Turn on power switch and leave to warm up for about 20 minutes.
- Transfer your samples to 0.3ml plastic cups by injecting the sample of water into the 0.3ml plastic cup. Then, get rid of the nib of the pipette and add a new one so as not to contaminate the next sample.
- When the flame photometer has warmed up, select which element you want to test for. Place empty sampling cups into all the rings of the Auto Sampler. Fill the calibration ring, labelled 'Cal', with electrolyte solution of Internal Standard Solution 1.5mmol/l Cs.
- Make sure the flame photometer is calibrating itself.
- Press 'zero' and it will then zero itself. It will test all the empty sampling cups.
- After it has done this, you can place your samples in the auto sampler.
- The flame photometer displays the amount of potassium in mmol/L. To get mg/L, multiply your reading by the element's molecular weight. For example, if you got a reading of 0.05 mmol/l for potassium, it would be 1.95mg/l.
Uses
Flame photometer are widely used in laboratories all over the world as they are cheap and compact enough. There are some laboratories which especially have flame photometers
- Biochemistry laboratories use a flame photometer to measure key elements such as sodium or potassium in a person's blood or urine.
- Analytical chemistry laboratories use flame photometers to measure the amount of specific elements such as sodium or potassium in drinking water and ground water.
Why are you reading this article?
Summary
- Atomic Emission Spectroscopy is a simple and inexpensive method.
- It is used to measure the amount of metal salts such as sodium or potassium in an aqueous solution such as blood, water or urine.
- There are two other branches of atomic spectroscopy known as atomic absorption spectroscopy and inductively coupled plasma.
- The sample is atomized and brought to the flame. The electrons in the sample get 'excited' and absorb energy. They go back down to the ground state and emit back out the energy that they absorbed.
- A spectral line is produced and the flame photometer can then determine how much of that element is in each sample.
- This technique is used in biochemistry laboratories to determine the amount of an element is in blood or urine.
- Analytical chemists use this techniques to measure the amount of sodium, potassium or lithium in water samples.
Sources
Holme D. and Peck H. (1998), Analytical Biochemistry Third Edition, Prentice Hall
Give Your Say!
Is this a good article?
Conclusion
Please feel free to post any comments, suggestions or questions you may have.
I'll be delighted to answer you!