Atomic structure of elements
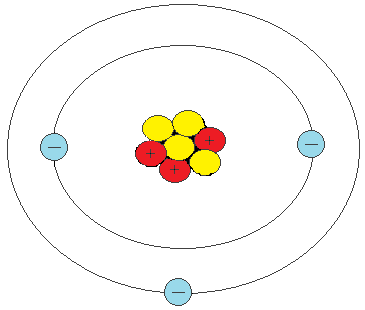
Lithium Atom
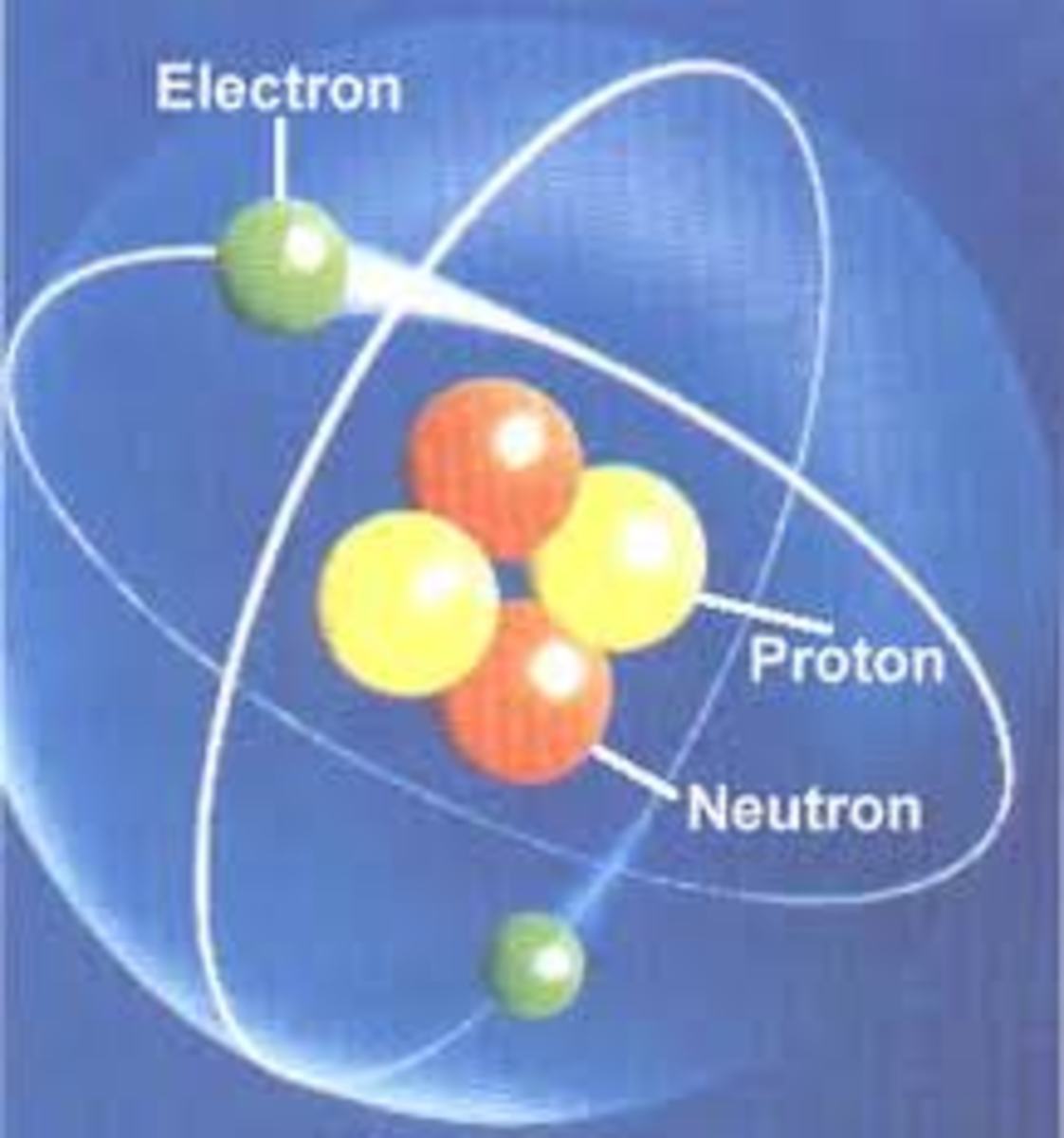
In my article What is an atom, I explained that all atoms are made up of protons, neutrons and electrons (except the hydrogen atom, which is just made up of protons and electrons - no neutrons). I showed a picture of a helium atom, with 2 protons, 2 neutrons and 2 electrons.
This is what happens if you get an atom with 3 protons, 3 neutrons, and 3 electrons - you get an element called Lithium. You can keep going with this idea, of more and more protons, neutrons and electrons, up past 100!
This would be a good place to say that sometimes, the number of protons, neutrons and electrons aren't always the same, and it is the number of protons which make the atom of a certain element what it is. An atom of lithium, for example, must have 3 protons, but it could have 3 neutrons, less than 3 neutrons, or more than 3 neutrons, and it would still be lithium.
These variations are called "isotopes" ( in this case, isotopes of lithium). These isotopes may sometimes be given their own names, but it is important to remember that they are still basically the same element. To stress again, it is the number of protons which makes the element what it is.
(If you think that the neutral charge of an atom would be affected if the proton and electron numbers didn't match - well done, you would be right. This can, and does, happen!)
Atomic number
This is so important, that the number of protons an atom has (the proton number), is sometimes called the atom's "atomic number". It also has a special scientific symbol "Z".
Z just refers to the number of protons in the atom's nucleus.
So you now know that the Z number of a hydrogen atom is 1, the Z number of a helium atom is 2, and the Z number of a lithium atom is 3!
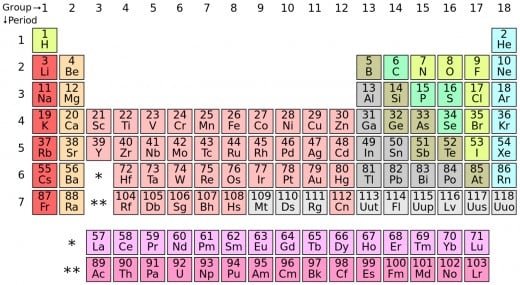
All known elements are listed, in order of their atomic number, in The Periodic Table. Look below, and find the 3 elements you've just learned about Hydrogen (H), Helium (He) and Lithium (Li).
The periodic table