Common Pyrotechnic Powder Compositions

Common pyrotechnic powder compositions are a mixture of substances that allow the production of heat, light, sound, smoke or gas, or a combination of these effects. The effects are achieved through the combustion of the different pyrotechnic powder compositions. These compositions are often referred to as "low explosives".
The following are the common pyrotechnic powder compositions.

Black Powder
The most common composition for pyrotechnics include black powder. Basically, every sort of firework is made with black powder or the chemicals which black powder is made of.
The chemicals used to make black powder are:
- Potassium Nitrate
- Charcoal
- Sulfur
New pyrotechnic should focus on these three chemicals. Fireworks using only these three components are also the only full compositions that should be milled. All these are the most common components of fireworks. Black powder is used to lift the shells out of the mortar, as rocket fuel, then making them blow up in the sky.

Coloring Agent
The different colors that we see in fireworks are produced by different chemicals. There is a corresponding chemical used to attain a certain color.
The following are the chemicals and the colors they produce:
Strontium - Red
Copper - Blue
Sodium - Yellow or Orange
Barium - Green
Calcium - Orange
Iron - Gold
To achieve other colors such as purple, Strontium can be used in conjunction with Copper. The incandescence from the different chemicals happens when the solid particles are heated to extremely high temperatures. Excess energy is released in the form of light (hv) at the broad end of the spectrum. When the temperature gets higher, the wavelength of the light emitted is shorter. The color blue is produced when this happens. Fireworks that are blue colored are difficult to produce because it can only be achieved at very high temperatures.
Oxidizing Agent
Oxidizing
agents produce oxygen to burn the mixture.The usual oxidizers used for pyrotechnic powder compositions are:
- Nitrates
- Chlorates
- Perchlorates
Nitrates are the most commonly used oxidizer. Nitrates are composed of metal ion and nitrate ion. Nitrates only give out one third of their oxygen.
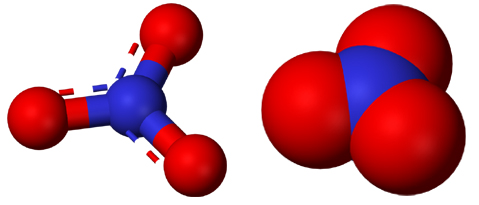
Chlorates are composed of metal ion and chlorate ion. Chlorates cause a more impressive reaction because they give out all of their oxygen. However, chlorates are also extremely explosive.
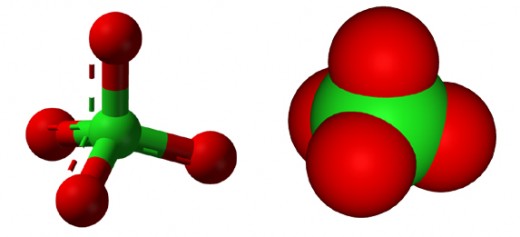
Perchlorates are made up of metal ion and perchlorate polyatomic ion. Even though perchlorates have more oxygen, they are less likely to explode compared to chlorates.
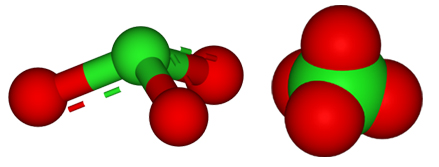


Reducing Agent
To produce hot gases, reducing agents burn the oxygen produced by the oxidizers.
The Sulfur and Charcoal or carbon are two examples of reducing agents.
These two react with oxygen, forming Sulfur Dioxide and Carbon Dioxide, respectively. Reducing agents control the speed of the reaction. The reaction can be faster or slower, depending on the reducing agents that are combined.
Regulator
To speed up the reaction, metals are usually added. Finer powder means a faster reaction. To slow down the reaction, cornmeal is used.
The two examples of firework regulators are:
- Cornmeal
- Metal
Binder
Finally, the binder is needed for most of the pyrotechnic powder compositions. A binder for pyrotechnic powder compositions is an organic glue that keeps the chemicals in a solid mass rather than as loose powder.

The first choice for this purpose is a basic water soluble binder, dextrin. Dextrin also known as cooked corn starch.

A substitute of dextrin is liquid laundry starch. This substitute works just as well at binding the pyrotechnic powder compositions together. The liquid laundry starch is made up of a solution of 50% liquid starch to 50% water by volume. This is then, used to dampen the pyrotechnic powder composition.
Another binder that can be used is paron. Paron, however, is less common and is used only for red and green fireworks because it improves their colors.



