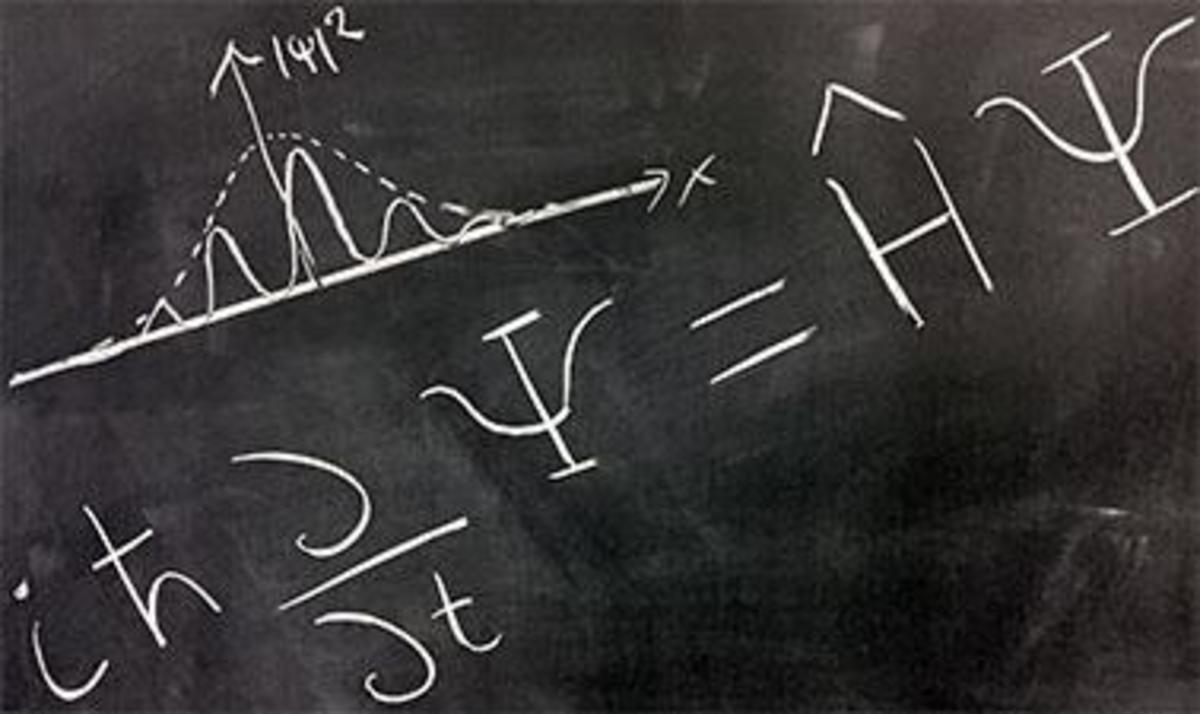Roles of Free Radicals, Intermediates and Signals in Health
Advances in medical research using free radical framework come up with categories
Look for disease
Molecular oxygen is a theory because it consists of concepts (atom, oxygen, electron, unpaired electrons) and relationships (joined covalently, spinning, comprise) between concepts. They fit the definition of theory given by Einstein. Molecular oxygen is a free radical with two unpaired electrons in its outermost orbital.
Actually, in “free radical theories of disease” there are two components. One consists of theory, the other consists of disease. There are several free radicals, each makes for a theory that is why we say ‘theories.’
“A theory by itself means nothing experimental... it is only when something is deduced from it that it is brought within the range of our material senses,” according to Norman Campbell (Campbell, N. What is Science? 1921:157). A theory is verified true if a statement that arise from it corresponds to experience. Seen as a context of our present discussion, a consequence that can be interpreted as disease is drawn from a theory of a free radical.
Sense-experience
Going back to the theory of molecular oxygen. One statement of fact derived from it is:
"An unpaired electron of molecular oxygen takes another electron from another molecule."
This statement of fact has been verified. It is true. We can have knowledge of it by means of inference mediated by instruments.
Molecules make up a tissue, or cytoplasm, or membrane, or DNA. From one of them, in one biological system, one or more electrons are taken. The molecule or tissue whose electrons had been grabbed is injured. This injury results in disease. This last sentence is an interpretation of disease.
“Free radicals can be caught in the act of creating cancer by using electron spin resonance (ESR) spectrometers. The unbalanced electron spin caused by the unpaired electrons in free radicals shows up in these tests. Organs with excessive free radical activity appear as hot spots” [Sharma, H., MD. Freedom from Disease (How to control free radicals, a major cause of aging and disease) 1993:90, author’s emphasis].
Categories of free radical theories of disease
Recently, there emerged categories in free radical theories. Molecular oxygen, superoxide, singlet oxygen and ozone belong in the oxidant category. Another category is intermediate. Still another category is signal (Eaton, S.S. et al., Editors. “Free Radicals and Medicine.” Biomedical EPR-Part A: Free Radicals, Metals, Medicine, and Physiology. 2005. 25-74).
We will use "free radical theories of disease" as a basket consisting of oxidant free radicals, intermediates and signals as these impinge on well being.
Examples of intermediate are hydrogen peroxide, hydroxyl radical, alkoxy radical, lipid peroxide, peroxynitrite, nitrous oxide, nitrosamine, nitrobenzene, carbon tetramethyl, trimethyl (mustard gas) and more.The intermediates act like free radicals in that they also grab electrons from molecules of tissues resulting in disease.
How are free radicals produced? For example, cyclooxygenase (CLOX) takes on arachidonic acid as substrate and produces prostaglandins, consisting of thromboxaneA2 and prostacyclin, with superoxide as by-product. ThromboxaneA2 promotes arterial constriction and aggregation of blood platelets that leads to thrombus that can result in heart attack (Sears, B. Ph.D. The Zone. 1995). Prostacyclin has an effect opposite that of thromboxaneA2. It promotes dilatation of arteries and allows more blood flow. Other enzymes produce superoxide: xinthase, NADhydrogenase, nitric oxidase.
Molecular oxygen is an oxidant free radical that we breathe. it is responsible for the metabolism of glucose to produce energy with superoxide as by-product. Molecular oxygen has two unpaired electrons in its outermost orbital with parallel spin around the nucleus. Each grabs an electron one at a time.
The metabolism of one molecule of glucose, in layman's language, goes as follows: One glucose molecule splits into two droplets, as it were, each droplet falls down like in a waterfall. Likewise two atoms of hydrogen split into electron and proton. Protons stand by. One electron falls down with each droplet. As it falls the electron emits energy in three stages. At the bottom of the waterfall molecular oxygen are waiting, in a way pulling down the falling electron and snap it. Thus one unpaired electron of molecular oxygen pairs up with one fallen electron. The other waiting electron is left behind that makes for superoxide. The fallen electron, one stand-by proton and one unpaired electron of molecular oxygen waiting at the bottom unite to make safe water. One glucose molecule produces 38 molecules of adenosine triphosphate (ATP) the currency of energy. Millions of this process go on per second to supply the energy needs of the body. For example, one brain cell consumes 10 million ATP per second. There are about three trillion cells of the body that consume energy; the brain cell uses 7.5 times more over other kinds of cell. That is why, when a person lacks molecular oxygen s/he becomes weak or dies. To repeat, the by-products of glucose metabolism are water and superoxide.
Singlet oxygen was formerly a molecular oxygen that was hit by ionizing radiation from the sun. The energy of radiation reversed the spin of its one unpaired electron. Thus two unpaired electrons in its outermost orbital spin in opposite directions. These two can grab electrons at the same time that is why the singlet is more destructive than molecular oxygen.
Superoxide is a master free radical that produces several siblings that are intermediates: hydrogen peroxide, peroxynitrite, hydroxyl radicals, alkoxy radical, lipid peroxide.
How are intermediates formed? The enzyme superoxide dismutase attaches one electron and one atom of hydrogen to superoxide. The mixture turns into hydrogen peroxide. Superoxide reacts with nitric oxide resulting in peroxynitrite. Nitrite, a meat preservative, reacts with an amine resulting in nitrosamine, a carcinogen in bacon. The energy of ultraviolet rays strikes the water in the skin and produces hydroxyl radical. There are more of these.
An example of signal category is nitric oxide (NO) produced by the endothelium nitric oxide synthase (eNOS). Three enzymes take on L-arginine as substrate and each produces NO and superoxide as by-product.
The enzyme endothelium nitric oxide synthase (eNOS) produces NO that serves as a signal for the endothelium to dilate, allow more blood flow and remedy angina or prevent heart attack. eNOS is like the drug nitroglycerin (Isordil, Imdur) that produces nitric oxide that alleviates hypoxia, angina and heart attack.
The enzyme neuron nitric oxide synthase (nNOS) produces another kind of NO. Most likely the NO of nNOS contributes to multiple sclerosis (destruction of nerve cells) and motor neuron disease.
The enzyme inducible nitric oxide synthase (iNOS) produces another NO mediated by the macrophage. Macrophage uses NO as bullets to shoot virus or bacteria like Streptococcus pyogenes, the cause of rheumatic fever. NO kills the bacteria but also does damage to healthy cells in the vicinity of inflamed infected cells. Hitherto healthy cells produce scars that later on graduate to stenosis thus rheumatic heart.
NO produced by nNOS and iNOs belong in oxidant free radicals that are responsible for most cancers.
However, the singlet oxygen can be put to good use. This is the free radical produced by chemotherapy drugs (Pressman, A. and S. Buff. Glutathione: The Ultimate Antioxidant). Singlet oxygen kills cancer cells and healthy cells when hit. That is why chemo has side effects.
Conducting experiments involving oxidant free radicals, signals and intermediates is difficult because they are very small (invisible), they deal with trapping agents, and their life span is very short. For example, hydroxyl radical (.OH) has a life span of 0.000000001 seconds. Free radicals and intermediates must be trapped to subject them to electron spin resonance spectroscopy. Trapping agents must be cleared (approved) for use in human beings. Good for research in nitric oxide because its trapping agent, dithiocarbamate, has been approved. Now we know that nitric oxide comes in three varieties owing to different enzymes. Other traps like nitrone spin are being evaluated.
New entries as of March 18,2013
Putting to good use oxidant free radicals
Nitric oxide (NO) produced by the eNOS is a useful free radical because it signals the endothelium to dilate and allow more blood flow.
NO produced by the iNOS (designated as NO/iNOS) is harmful because it mutates healthy cells resulting in scar, or stenosis, or tlumor or cancer.
However, NO/iNOS can be put to good use. It can be used to kill cancer cells in gene therapy that is a new method to cure cancer.
Chemotherapy uses free radicals because these are its active ingredients, particularly singlet oxygen.
In gene therapy, the human gene that controls production of NO/iNOS is carried by a protein coat that precisely delivers NO/iNOS to cancer cells. Once incorporated into the chromosomes of cancer cells, iNOS produces NO/iNOS that kills cancer cells. It is important that the delivery of NO/iNOS is specific to cancer cells so that it will not land on healthy cells.
Gene therapy for cancer is still in the development stage. I have more Hubs on gene therapy for cancer, like "Cancer cure: chemotherapy and gene therapy compared," .