GCSE Chemistry - C3 - Alcohols

What Are Alcohols?
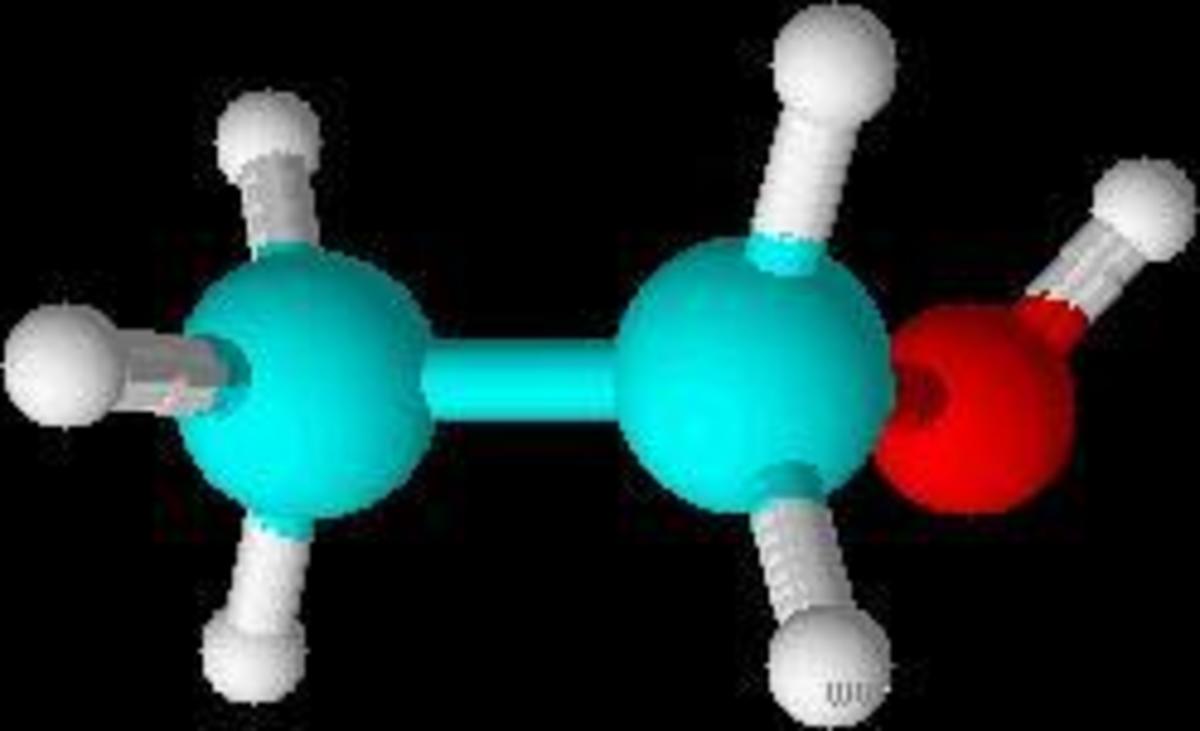
Alcohols are organic chemical compounds which form a homologous series.
They are compounds in which one or more hydrogen atoms in an alkane (saturated hydrocarbon) are replaced by hydroxyl (OH) groups.
The hydroxyl group (OH) is the part of the molecule that is responsible for the characteristic reactions and chemical properties of the alcohol. This is otherwise known as the 'functional group'.
Homologous Series
The Homologous Series is a series of compounds with a similar general formula. The compounds possess similar chemical properties due to the presence of the same functional group. Alcohols belong to a homologous series because they possess the same functional group but have different length hydrocarbon tails.
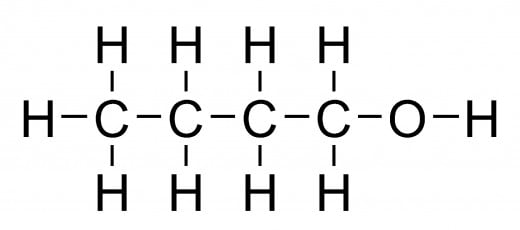
Different Alcohols And Their Formulae
Methanol: CH3OH
Ethanol: C2H5OH
Propanol: C3H7OH
Butanol: C4H9OH
Pentanol: C5H11OH
Hexanol: C6H13OH
Random Alcohol Related Facts
- Alcohol is flammable because it has a short hydrocarbon chain.
- The head of the alcohol molecule is hydrophilic (it's soluble in and has a strong affinity for water).
- The tail of the alcohol molecule is hydrophobic (it repels and fails to mix with water).
- Industrial alcohol is made by the hydration process which involves reacting ethene + water to produce ethanol.
- The conditions needed for optimum hydration is 700°C, 70 atm pressure and phosphoric acid as a catalyst.
- Ethanoic acid is used in vinegar and methanoic acid naturally occurs in wasps/nettles/ants stings.