Gold - Mining, Chemistry, & Applications of Gold
Introduction
Once upon a time the metal gold was known by the name of the celestial object sun and was represented by the sign O [1] . The name gold originated from the English Anglo-Saxon word geolo, which means yellow [2]. The chemical symbol of gold, Au, originated from the Latin word Aurum , which means Glowing Dawn [3]
Gold is believed to be the first metal known to humans. Few thousand years back, a child observed a shiny rock in a creek. This is considered to be the first introduction of gold to humans [4]. The people of the Inka civilization in pre-Columbian America referred to this metal as The Tears of the Sun [4]. In Iliad and Odyssey, Homer mentions gold as the glory of immortals [4]. Plato and Aristotle considered that gold was a combination of water and sunlight [4] . Gold was first discovered in its natural state in streams.
Gold Mines
Some of the best known gold producing regions of the world are California, Colorado, Ontario, Mexico, Australia, Hungary, China, Russia and South Africa (the largest producer). Some famous American mines include the Empire Mine and Red Ledge Mine in Grass Valley (Nevada County), California. Very nice crystals and crusts have been found near Ouray, Colorado, and in Red Mountain Pass, San Juan County, Colorado. The Hollinger mine, along with other mines in the area of the Porcupine District, has produced large amounts of Gold [5].
Two thirds of the world's supply comes from South Africa and two thirds of the US production is from South Dakota and Nevada [6]. It is estimated that the total production of gold from mines is about 160,000 tonnes, since gold was first discovered [6].

Geology of Gold
Earth’s crust contains gold in a very small concentration of about 0.005 parts per million, i.e., 5 milligrams (mg) gold per tonne of the earth’s crust. Gold also occurs in sea water in concentrations of 0.1 to 2 mg per thousand liters water.
Gold ore is generally brown iron-tinged rock or massive white quartz. Gold is found in nature in the state of a free metal and a compound of tellurium known as telluride [7]. Gold occurs in different kinds of rocks. Load deposits which are the main deposits of gold, are formed by geological processes. These deposits can be commercially exploited. Gold deposits are generally found in volcanic and sedimentary rocks. These deposits are formed due to circulation of hot ground water. Water gets heated from the magma (molten rock) intruded into the Earth’s crust within about 2 to 5 miles of the surface. Heated water moves downward through fractures and permeable beds in cooler parts of the crust and is drawn laterally into areas heated by magma, where it is driven upward through fractures. The water gets superheated and carries with it metal dust from the surrounding rocks. When the heated waters reach cooler rocks at shallower depths, metallic minerals precipitate to form veins or blanket-like ore bodies [6].
A nugget of gold is a solid lump of gold containing about 70% - 90% gold. In nature, gold is rarely found as nuggets. A gold nugget mined from the Eagle's Nest mine in Placer County, California is shown here [8]. Nuggets are formed when gold is separated from gold containing rocks due to erosion and then carried away into a river or stream. Eventually, gold settles at the bottom of the water due to its heaviness. The gold deposits formed by this process are termed as placer deposits [5].
The Greeks had mined gold from Gibraltar (Pillars of Hercules) to Asia Minor and Egypt. The Romans made significant advancement in gold-mining technology. They mastered the technique of hydraulic mining by using streams of water and water-wheels. They were the first to introduce the roasting of gold-bearing ores to separate the gold from rock.
Extraction of Native Gold
Gold is the only metal that occurs in pure state in nature. This is termed as native gold. In the quest of native gold, even in days when gold had no value as money, prisoners of war, slaves, and criminals were being sent to work in gold mines by Indians, Chinese, Egyptians, Hittites, Phoenicians and others. In those days hydraulic mining was the primitive form of mining for the recovery of gold. This method was still being used by some miners as recently as the California gold rush of 1849. Water power was used to propel gold bearing sand over the hide of a sheep. This hide would absorb the gold bearing sand. This ‘golden sheep’s fleece’ was then hung up to dry and then beaten gently and collect the fallen gold dust [4].
Recovery of Gold from Ore
Gold is usually mined from the ore containing very small amount of gold. Generally the recovery of gold is considered to be economical when it is in the range of 13.7 to 17.1 grams (g) per tonne with cut-off around 8.6 g per tonne [9]. However, with the passage of time and advancement in methods of mining and milling, the recovery of some deposits of lower grade but larger tonnage has become economical. Open pit methods of mining greatly reduces the mining costs. But this is possible only when veins occur near the surface [9].
Gold is recovered in the form of concentrates from its ores mainly by (a) amalgamation or (b) cyanidation processes associated with roasting. These processes yield gold with a concentration of between 70% - 90%. High purity gold (99.6% to 99.7%) is then obtained by chlorination (Miller’s Process). Maximum purity (99.99 %) is achieved by electrolysis. When silver is associated with gold in the native form it is also recovered by the amalgamation and cyanidation processes.
Amalgamation
When clean mercury is brought into contact with clean gold, a solution of gold in mercury (an alloy of gold and mercury) is formed. This is an amalgamation process. Mercury can then be removed from gold by dissolving it in nitric acid. Mercury can also be removed by heating the alloy and vaporizing it off. Gold will remain behind and can be refined to a desired high purity. However, for amalgamation process to work, gold should be in finely divided form.
When gold is encased in the gangue or surface coated, ultra-fine grinding of the material is necessary to liberate gold particles from the mineral. The presence of clay, iron, and other base metal sulfides results in flouring of mercury. Flouring is the division of the mercury into extremely small globules. This gives it a white flour-like appearance. Any gold that attached to the floured mercury is also lost along with mercury. To avoid flouring, grinding the material with alkali or lime is necessary. Flotation prior to amalgamation is required if the mineral contains carbon or graphite. The presence of sulfides of arsenic, antimony, and bismuth causes flouring and sickening of the mercury. If the mercury does not attach to gold due to the presence of impurities like oil, grease, clay, manganese, and iron sulfates, and base metal and iron sulfides present on the surface of the mercury, it is called the sickening of the mercury. In such a situation, usually gold can not be recovered by amalgamation process [10].
Amalgamation process for the extraction of gold was widely used till the end of the first millennium. In America mercury was introduced for the first time in the 16th century to amalgamate Mexican gold and silver. The amalgamation process was widely used in Canada from the 1860s to1890s [11]. Amalgamation process creates pollution. Mercury is a toxic metal. Mercury used in amalgamation process cannot be recovered as much as is necessary to ensure that the remaining mercury does not pollute the environment.
Cyanidation
Cyanidation is a process for the extraction of gold from ores and was first developed by MacArthur Forrest in 1887 [12]. Since then this is the principal method of extracting gold from ores. In this process, the ore is crushed to a very fine powder. Such powdered ore is heaped onto open-air leach pads put on a base of asphalt or impervious plastic sheeting. A dilute solution of cyanide, usually sodium cyanide, is sprayed through sprinklers on the heap. The cyanide solution percolates down through the heap for several weeks, forming cyano gold complexes. This solution, enriched with gold, gets collected at the bottom into the pond termed as pregnant pond, from which it is pumped to the recovery plant. In the recovery plant, the solution containing cyano gold complex is filtered off and the remaining rock pulp is separated. Zinc dust is then added to the solution containing cyano gold complex to reduce the gold (III) oxidation state to zero oxidation state (metallic state). Gold is thus precipitated out as a high grade concentrate. Gold precipitate is then refined to get high purity gold. The perfection of the cyanide process largely replaced amalgamation process [12].
This has proved to be an economical process even for the extraction of gold from low grade deposits in spite of the low recovery (60% - 70%) of gold. Since 1973 this process is used In United States for extraction of gold from low grade deposits at Placer Development's Cortez open pit in Nevada and at Pegasus Gold's Zortman Landusky mine in Montana.
It should be noted that cyanide is extremely toxic and must be handled with special care.
Some ores of gold contain sulfide minerals and/or organic matter. These ores become refractory and are resistant to the cyanidation process because sulfide minerals trap the gold particles and organic carbon absorbs dissolved gold cyanide after cyanidation . Hence, oxidation of such ores is necessary. Oxidation is achieved by one of the following three processes
(a) roasting the ore at high temperature using air and/or oxygen followed by ultra-fine grinding of the ore. When the minerals are roasted, the base metals, such as zinc, iron, lead, and copper, from the minerals get converted to their respective oxides
(b) bio-oxidation using bacteria that promote oxidation in an aqueous medium
(c) pressure oxidation in an autoclave at high pressure and somewhat high temperature.
Oxides of these minerals, can then be easily removed into a slag in a subsequent smelting process [13].
Smelting
If the ore has high concentration of gold (0.03% - 0.05%) and does not contain minerals such as sulfides, neither amalgamation nor cyanidation process is necessary. In such cases, the smelting process can be directly used to remove base metals and other impurities [14]. Generally the temperature required for smelting is between 1200 0C to 1400 0 C. This smelted product is suitable for further refining to get high purity gold.
Refining of Gold
There are two major processes for further refining the gold concentrate obtained from the above methods.
Miller Process
Dr. F. B. Miller developed a process of refining gold using chlorine gas at the Sydney Mint, Australia. This is based on the principle that chlorine combines with base metals and silver, the impurities generally present in gold concentrates and scrap gold, to form chlorides while gold is not affected. This gold concentrate is termed as Dore, which is unrefined gold consisting primarily of gold with smaller amounts of base metals and silver. This is then placed in clay pots in a furnace. On melting the dore (at temperatures around 1060 oC) chlorine is pumped in each pot. After two to three hours the pot is removed from the furnace and the molten chlorides skimmed off.
This process requires a high temperature of about 1060oC, which is expensive to maintain. Moreover, red-brown toxic copious fumes of gold chloride are generated. In this process gold of only 99.6% to 99.7% purity is produced, which is not suitable for a number of applications [15].
Wohlwill Process
Dr. Wohlwill developed the electrolytic process of gold refining in 1874 [16]. In this process the anodes are of impure gold suspended in porcelain cells and the cathodes are thin strips of pure gold. By passing an electric current from anode to cathode through the electrolytic solution of gold chloride (AuCl3) in hydrochloric acid the anodes are gradually dissolved and the gold therein is deposited on the cathode. Any silver which is insoluble in the electrolyte (as silver chloride) and any platinum group metals present in impure gold are precipitated to the bottom of the cell. Base metal impurities remain in the electrolytic solution.
The gold-coated cathodes are removed, melted and cast into bars. This process can produce gold up to 99.95 % purity initially. On repeating the electrolysis 99.99 % purity is achieved.
The main drawback of this process is that a large amount of gold, in the form of gold chloride, remains on site in electrolytic bath at all times. Moreover, a large amount of electricity is required for this process.
To the best of the knowledge of the author of this article, Miller's process and Wohlwill process are the only ones now used all over the world in commercial refining of gold.
A Pollution-free Novel Process for Refining
The author of this article has developed a solvent extraction process for industrial use for high purity gold (99.99%) from any gold concentrate including anode slime. Anode slime is a term used for the by-product of copper refining. It usually contains high concentrations of silver, gold, and some platinum group metals. This process neither involves smelting nor electrolysis and is practically pollution free. All the reactions are carried out at temperatures around 100oC. This process is far superior to Wohwill process or Miller process. The interested reader may contact the author.
Chemistry of Gold
Gold has an atomic number of 79 and atomic mass of 196.96657 amu (1 amu = 10-24g). It is placed in group 11 and period 6 in the periodic table. Its electron configuration is 1s2 2s22p6 3s23p63d10 4s24p64d10
4f14 5s25p65d10 6s1. Its oxidation states are 3 and 1.
Gold is available in native state as alluvial gold and vein deposits. Only gaseous chlorine and fluorine will react with gold. Gold does not dissolve in sulfuric acid, nitric acid, or hydrochloric acid, but it dissolves in aqua regia (a mixture of HCl and HNO3) to form soluble chlorauric acid (HAuCl4). The HNO3 in HCl acts as an oxidizing agent and hence, gold gets dissolved in aqua regia. It is also possible to dissolve gold in hydrochloric acid when chrlorine gas is passed through it, as it oxidizes gold. Similarly gold can be dissolved in hydrochloric acid using hydrogen peroxide as an oxidizing agent. Gold dissolves in sodium or potassium cyanide which is used in the extraction of gold from ores. It also dissolves in solution of azoimide [17].
Physical Properties of Gold
The density of gold is 19.3 g/cc at 20°C. Its melting point is 1062oC and boiling point is 2000 oC. Gold is a good conductor of heat and electricity, and is highly resistant to corrosion. It is also resistant to bacterial colonization. Gold is a good reflector of heat without reducing light. It is highly opaque to X-rays.
Gold is extremely malleable, which is the ability to undergo deformation on compression before failure. It can be compressed to a thickness of 0.000018 cm. Gold is extremely ductile, which is the ability to extend on tension before failure. One gram of gold can be drawn into 3 km of thin gold wire of about 5 micron (1 micron = 10-6m.).
Gold has eighteen isotopes [17]. Two atoms are isotopes of each other if they have the same atomic number (same number of protons), but different atomic weight (different number of neutrons).
Applications of Gold
Applications in Commerce and Jewelry
Gold has been used as a medium of exchange since 3000 B.C. During the Classic period of Greek and Roman rule, gold and silver were traded to buy spices from India and silk from China [4]. In the late 1700's it became the world monetory standard [18]. Gold serves as a monetary reserve. The first gold coin was minted by Croesus of Lydia in circa 560 B.C.[6]. The coins were simply stamped lumps of a mixture of 63% gold and 27% silver. This mixture was termed as electrum. At the height of the Roman empire (A.D. 98-160), Roman gold and silver coins reigned from Britain to North Africa and Egypt [4].
Around half of the world's supply of gold is stored in the United States Treasury Department's gold depository in Fort Knox, Kentucky. This is considered to be one of the most secure buildings in the world [19].
As of 1991, more than 83% of gold is used for jewelry, 6% is used for medals and official coins, 6% is used in electronic equipment, 2.2% is used for dental materials, and 2.8% is consumed in a variety of industrial applications [20]. In jewelry generally metals like copper, silver etc. are added to gold to form gold alloys to increase the hardness and strength. However, some reduction in malleability and ductility has been observed in gold alloys compared to pure gold. In jewelry the purity of gold is measured in terms of karat. Twenty Four karat gold means 100 % pure gold which is intense yellow. Purity of gold is also expressed in terms of fineness. 100% pure gold is 1,000 fine. The weight of gold is generally measured in troy ounce (1 troy ounce = 31.1034768 grams).
One can get gold of different colors depending on the concentration of the alloying metals such as copper, silver etc. added to gold (See Table below). Alloying metals also affect physical properties of gold. [21].
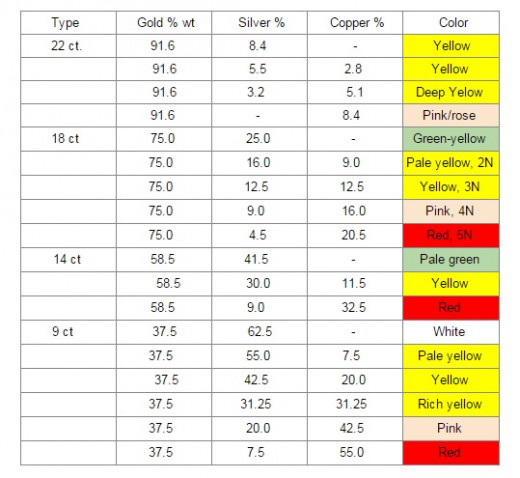
Applications in Electronics
Gold is used in ignition control electronics and in anti-lock brakes in automobiles. Gold contacts are indispensable in crash sensors for airbags [22]. Gold is an indispensable element for nanoscale electronic components because of its resistance to oxidation and its mechanical robustness [22].
Gold has a good ability to wet silicon at 371°C. Hence, gold powder and gold sheets are used for soldering semiconductors [17]. Gold is used in electronic tubes as gold-plated grid wire. It gives high conductivity and suppresses secondary emissions[17].
The University of Delaware, USA has developed new, self-assembling, and repairing gold nanowires. This could find applications in the development of nanoelectronics [22]. Gold-gallium and gold-antimony alloys are used, primarily as wires, in electronic industries [17].
Applications in Chemical Industry
Gold can be used as a catalyst in chemical processing, pollution control and fuel cell applications. However, the particles of gold should be less than 5nm (nanoparticals) [22].
The most important aspect of gold as a catalyst is to achieve the light-off temperature. The light-off temperature is the temperature at which the catalyst becomes functional. Gold is already being used in the gold-palladium alloy catalyst in the production of vinyl acetate monomer. It is also possible to use gold catalyst to reduce odor and to remove nitrogen oxides from diesel engine exhaust gases [22]. Gold-based nanotechnology can be used for the development of sensors to detect species that have specific affinities for nanoparticles [22]. . Sodium gold cyanide is used as a gold plating solution. This plating has good chemical resistance and electrical properties. However, the plating lacks wear resistance [17].
Applications in Medical Sciences
An Isotope of gold, Gold-198 having half life of 2.7 days is used in some cancer treatments and also for treating some other diseases [23]. Scientists from National University of Singapore have patented novel gold complexes for use in pharmaceuticals for the treatment of cancer [22]. Gold wires are used for pacemakers and gold-plated stents to inflate and support arteries in the treatment of heart disease. This application is possible as gold is highly opaque to X-rays, which helps in positioning the stent [22].
Gold is resistant to bacterial colonization. Hence, it is used for implants in the inner ear. Gold is used to plate myringotomy tubes. Myringotomy is a surgical procedure in which a small incision is made in the ear-drum, the tympanic membrane and tubes are placed to drain and temporarily aerate the tympanic cavity [22].
Gold is used in drug delivery microchips. The microchip contains drug-filled reservoir which is covered, sealed and protected by thin gold membranes. By applying a small electric voltage to the gold reservoir cap a dose of the drug is administered to the patient. This causes the cap to dissolve and allows the drug to release from the reservoir [22].
Gold, alloyed with silver, platinum and sometimes with palladium and indium (for hardening), termed as dental gold, is used for dental applications [17]. In the seventh century BC, gold wires were being used to hold substitute teeth in place when original teeth were damaged [22].
Gold is used in the treatment of arthritis, generally being administered intramuscularly [24]. In India and Egypt people have been using gold-based medicinal preparations for the last few thousand years. In China, gold was used for the treatment of smallpox and measles. In Japan it is considered that thin gold foils placed into drinks or foods are beneficial to health [22].
Nanoparticles are particles having dimensions of the order of 100nm (1 nm =10-9 meter) or less. The properties of nanoparticles are different from the properties of the bulk material [25]. Gold nanoparticles are widely used in immunohistochemistry to identify protein-protein interaction. The properties of nanoparticles are different from the properties of the bulk material [26]. Gold nanoparticles absorb light very strongly in the near infrared, a spectral region that is barely absorbed by tissue. The absorbed light energy causes the gold particles to vibrate and is dissipated as heat into the surrounding area. The tiny nanoparticles of gold can be functionalized so that they specifically bind to tumor cells. Thus, only cells that contain gold particles are killed off as the cancer cells are relatively temperature sensitive [27].
Professor Leung Pak Hing and his team have discovered that phosphine-supported gold complexes have excellent anti-tumor activity and clinical trials are likely to begin shortly [22].
Other Applications
Gold is an excellent reflector of heat and infra-red radiations. Hence, liquid gold is being used to reduce heat transmissions from aircraft engines. It is also used in the United States Apollo space program [6]. Nearly 41 kg. gold was used in the construction of the US Columbia space shuttle, especially in brazing alloys, fuel cell fabrication, coated plastic films and electrical contacts [28]. The Hubble space telescope is protected by gold coatings to provide corrosion resistance [28]. The cockpit windows of jet aircraft are coated with a wafer-thin film of gold. This deflects the harmful effects of the sun’s rays. and forms an invisible film to resist extreme temperatures [28]. Gold reflects heat without reducing light. Hence, it is used on some windows in the construction industry [28].
In this article few applications of gold are mentioned with a view to recognize the importance of gold besides its use in jewelry which is most commonly known. Many more applications exist and the potential for more applications is high. In future, nanoparticles of gold will play a miraculous role in the field of medicine and electronics as the physics of nanoscale particles is different from bulk gold metal. Intensive research for applications of nanoparticles of gold in all fields is being pursued. It will not be a surprise that in future nanoparticles of various metals will be prepared and will find use in various fields. Technology of preparation of nanopartcles of metals is well advanced.
It is pertinent to note that the properties of gold such as its shining yellow luster, inactivity to most chemicals, conduction of heat and electricity, malleability, ductility, resistance to corrosion and bacterial colonization and the specific properties of nanopraticles made this element unique.
References
- http://www.universetoday.com/guide-to-space/the-sun/symbol-for-the-sun/
- http://www.innvista.com/science/chemistry/elements/etymolo.htm
- http://www.babynames.com/name/AURUM
- http://www.onlygold.com/tutorialpages/HistoryFS.htm
- http://www.minerals.net/mineral/elements/gold/gold.htm
- http://www.usagold.com/reference/properties.html
- http://www.galleries.com/minerals/elements/gold/gold.htm
- http://nevada-outback-gems.com/prospect/gold_specimen/Natural_gold.htm
- http://earthsci.org/mineral/mindep/depfile/vei_dep.htm
- http://www.e-goldprospecting.com/html/gold_amalgamation.html
- http://www.jmeech.mining.ubc.ca/briefhi2.htm
- http://snobear.colorado.edu/Markw/Intro/Summitville/Cyanide_Leach/cyanide.html
- http://www.e-goldprospecting.com/html/roasting___smelting.html
- http://www.e-goldprospecting.com/html/smelting_refining.html
- http://info.goldavenue.com/Info_site/in_glos/in_glos_miller.html
- http://info.goldavenue.com/Info_site/in_glos/in_glos_wolhill.html
- http://www.azom.com/Details.asp?ArticleID=598
- http://nautilus.fis.uc.pt/st2.5/scenes-e/elem/e07930.html
- http://environmentalchemistry.com/yogi/periodic/Au.html#Overview
- http://serc.carleton.edu/research_education/nativelands/ftbelknap/golddeposits.html
- http://www.utilisegold.com/jewellery_technology/colours/colour_alloys/
- http://www.azom.com/Details.asp?ArticleID=1899
- Nanoscience and Nanotechnology in Nanomedicine: Hybrid Nanoparticles In Imaging and Therapy of Prostate Cancer - Radiopharmaceutical Sciences Institute, University of Missouri-Columbia
- http://nautilus.fis.uc.pt/st2.5/scenes-e/elem/e07940.html
- Applications of nanoparticles in biology and medicine by OV Salata, journal of Nanobiotechnology 2004, 2:3doi:10.1186/1477-3155-2-3
- http://www.malvern.co.uk/LabEng/industry/nanotechnology/nanoparticle_defiition.htm
- http://nanotechwire.com/news.asp?nid=6618
- http://www.bmnl.com.au/our_environment/why_we_mine_for_gold.htm
© 2015 Discover the World








