Is Carbon the Enemy?
Background
Carbon has the chemical symbol ‘C’, and an atomic number of 6. It is classified as non-metallic. It is an element, meaning it is one of the basic building blocks of chemistry as it is composed of one type of atom. Carbon is tetravalent which means that it has 4 electrons available to form covalent chemical bonds. There are three naturally occurring isotopes of carbon: 12C, 13C and 14C. The latter is radioactive.
Although diamond (an allotrope of carbon) was known about as far back as 2,500 BC, it was the Egyptians and Sumerians who are recorded as first using charcoal (carbon made by heating wood) to reduce copper, zinc and tin to make bronze in about 3,750 BC. It wasn’t until the Eighteenth Century that any proper analysis of chemicals was carried out. In 1789 Antoine Lavoisier added carbon as an element.
Nano technology
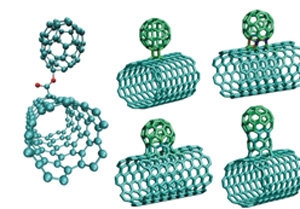
Allotropes of Carbon
An element can exist in a number of different atomic structures. These structures are called allotropes.
The most famous allotrope of carbon is diamond. Although famous for its gem properties, most mined diamonds and synthetic diamonds are used in industry. They are used because diamond is very hard and is extremely heat conducive. Diamonds in industry are typically used for cutting, grinding and polishing. Efforts are being made in the USA and Japan to use synthetic diamonds for building microchips and as heat sinks in electronics.
Graphite is regarded as the standard form of carbon because it is the most stable. Graphite is unusual in that it is non-metallic but nevertheless an electrical conductor. Graphite is also a lubricant: this is because of the water and air that gets trapped between the layers in its structure. The lubricity of graphite explains its name and most common use, namely as pencil ‘lead’. Layers peel off smoothly and leave a mark on paper. Graphite comes from the Greek word meaning to draw.
The conductivity of graphite has made it useful to make EDM electrodes. EDM is electrical discharge machining. It is a process where a high voltage is run between a graphite electrode and a ‘workpiece’ in a medium of a dielectric liquid. When the distance is short enough between the electrode and the workpiece an electrical spark is given off that erodes away a small piece of the workpiece, thus cutting it. Several hundred thousand sparks are given off per second that effectively cut even the hardest metals and alloys which are difficult to cut by conventional methods of grinding, milling etc.
Amorphous carbon does not have a crystalline structure. It is a glassy substance and is not found in a pure form as it usually contains traces of graphite.
Buckminsterfullerenes was named after its discoverer, Richard Buckminster "Bucky" Fuller. These are pure carbon in the shape of a hollow sphere, ellipsoid, or tube. These were discovered in 1985. It has led to the production of nano-tubes, which are the basis of nano-technology.
Carbon dioxide emissions

Carbon and the Environment
Carbon is the basic unit of organic molecules. 18% of the human body is composed of carbon. Carbon is part of our DNA. Carbon is the fourth most abundant element in the universe. Our atmosphere is regulated by plant life that breaks down carbon dioxide into sugars and oxygen by photosynthesis. This is where environmentalists are concerned with the reduction of tree cover on the planet. It means that less carbon dioxide (given out when animals breathe and from many industrial processes) is being ‘recycled’ into oxygen. The excess CO2 is being trapped instead in the atmosphere and stops the sun’s heat from escaping; thus warming up the planet. This is the greenhouse effect, and carbon dioxide is known as a ‘greenhouse gas’.
The notion of a person’s carbon footprint is a bit misleading as it is not connected to how much ‘carbon’ we use. Rather it refers to the goods, services and actions that we do and consume that give off carbon dioxide and that therefore contributes to global warming.
Typically, buying manufactured goods; driving a car; taking an airplane; using electricity that is made by burning fossil fuels all cause an increase in carbon dioxide levels. The business of being eco-friendly is concerned (but not exclusively) with finding ways of reducing our production of carbon dioxide. This can be by using public transport, solar energy, recycling and re-purposing and by being more efficient in energy use. Using better insulation in our homes, controlling heating and cooling with a programmable thermostat, installing low energy light bulbs are all ways to reduce electricity use and thus carbon dioxide production.
The notion of off-setting is about trying to make up for carbon dioxide emissions by doing actions that will absorb carbon dioxide. Typically this means planting trees, protecting trees and vegetative life and by paying developing countries to not cut down forests.
Conclusion
While the build-up of carbon dioxide in the atmosphere is of grave concern for humanity, the modern fad to label things as ‘carbon intensive’ etc. is misleading. Carbon is the basis of organic life. It is fundamental to engineering and design. Carbon is fundamental to the composition of the universe, and makes modern technologically advanced life possible. Carbon is not the villain: it is man’s out-of-control production of carbon dioxide that is to blame.








