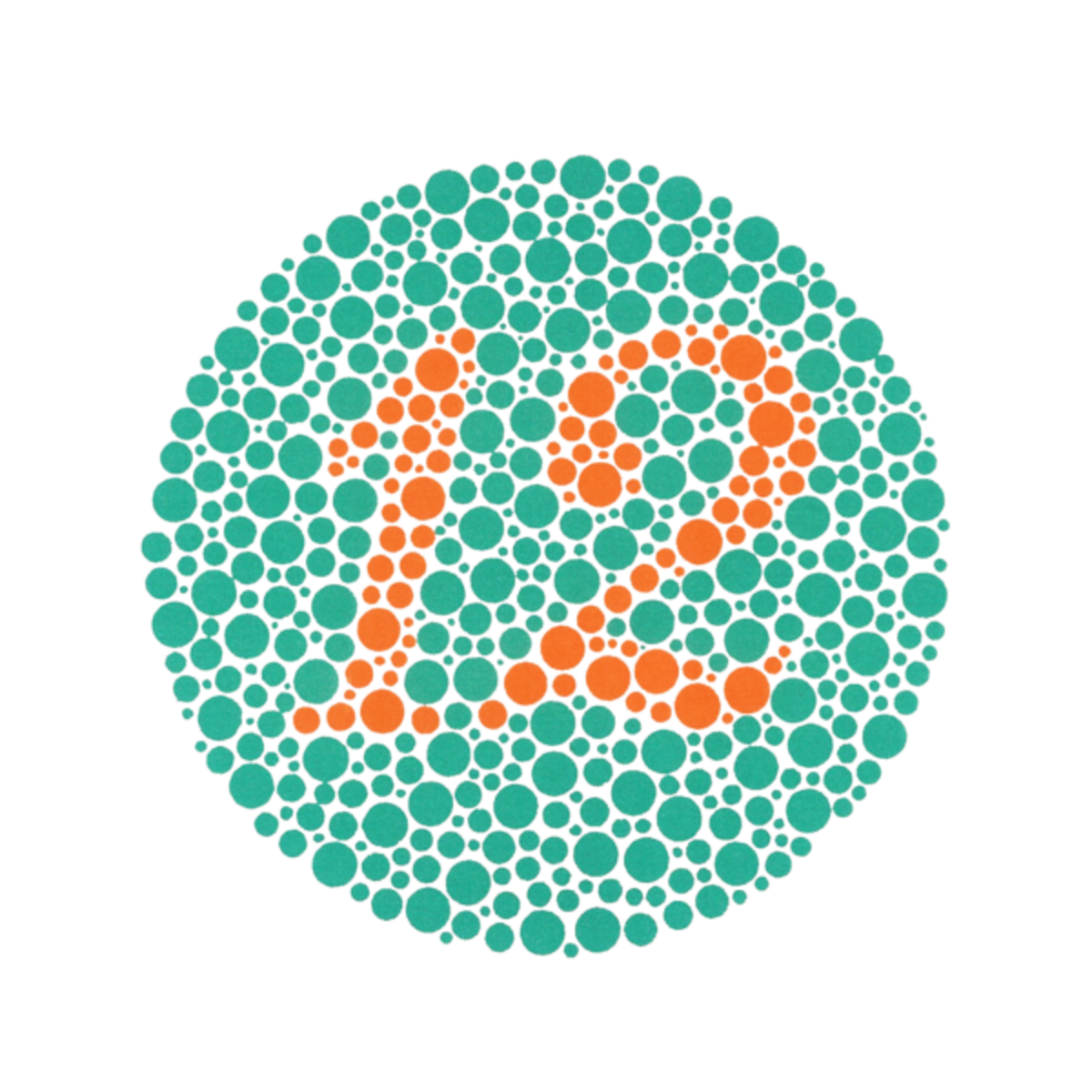John Dalton
Early Life And Education
John Dalton was born in a Quaker (a religious society) family in Eaglefield, Cumberland County. His mother, Deborah, lived by the motto "for God and husband". His father earned his living at the handloom. Such was the environment of the Dalton household. The puny infant grew to be a stubborn conscientious lad. Once put to a task, he could grapple with it against all odds with the tenacity of a bulldog. Mr Robinson, Dalton's schoolmaster was satisfied with him because he was very good at mathematics. Often, Dalton would request his teacher not to provide the solution of a problem as he wanted to solve it himself. Dalton always tried to find the most novel way to solve problems.
John Dalton started school when he was 12. He nailed a message to his father's door that in addition to his learning, he would supply the students with free paper, pens and ink. These items were rarest of commodities at the time. At 15, Dalton was tempted to go into agriculture in partnership with his uncle. At a later stage, he joined his brother, Jonathan, who was running a technical school. Dalton had also started work on weather forecast. The unlettered people of the Cumberland County looked to him as an uncanny weather prophet, for he had begun to take a daily, almost hourly reading of the weather - a practice which he continued for 57 years.
His Works
The academic contributions of the great scientist John Dalton (1776-1844) included investigations in meteorology. He disproved the false science of alchemy and established the atomic theory in chemicals. His books and essays include On Colour Blindness, The Atomic Theory, The Molecular Theory and New System of Chemical Philosophy.

His Life And Achievements
Dalton undertook a series of lectures on natural philosophy based on his personal observations. He planned this series to include talks on the laws of motion, colour, wind, sound, harvest moons, lunar eclipses, planets and tides. But these lectures were not successful because people could not understand them.
Dalton heard that, in Manchester, the Presbyterians had founded a college dedicated to truth, liberty and religion - an institution designed to serve as a protest against dominant British universities which excluded Unitarians and Quakers. He applied for a position to teach natural philosophy and mathematics and secured the job. However, he found the academic restrictions of his new life unpalatable and decided to return to his private tutoring. In order to meet his expenses, modest as they were, he was obliged to teach both day and night.
Dalton tried to increase his earnings so that his expenses could be comfortably met. Once he started collecting different types of flowers and pressing them in his books with the object of selling them because they looked pretty. To his botanical collection, he also added a collection of common insects. All these efforts did not succeed and hardly any of his specimens were sold. On another occasion, he prepared a book on grammar to provide further aid to his early retirement but this was also did not generate additional money. He published a series of essays on his meteorological investigations with the prefatory remark that he had relied not on books but on his own observations. His knowledge came almost always out of his personal experience.
John Dalton was colour blind. This was disclosed when he purchased a pair of stockings for his mother. To John, they were a dark conservative blue but to his mother they were as red as a cherry. He was determined to investigate the inconsistency between his vision and that of others. Dalton was able to find some more cases of colour blindness and was, thus, able to explain the phenomenon.
For almost 30 years after his resignation from Manchester College, Dalton lived and experimented at the house of a generous clergyman, Reverend Johns. With his blunt and forthright honesty, Dalton offered himself as a willing guest to his willing host. His daily life never varied over a long stretch. Dalton's laboratory was his shrine in which he spent most of his time experimenting. When warm weather set in, he conducted his meteorological experiments in Lake Country and, thus, combined business with pleasure. He climbed mountains not only to test his barometers but also to bring into exercise a set of muscles which otherwise would have grown stiff.
While Dalton was experimenting with chemicals, a momentous idea dawned on him. In the realm of physics, Newton demonstrated that particles of matter were attracted by the weight of their atoms, so Dalton wondered whether or not it was possible that chemical bodies, too, might be found to consist of ultimate particles of atoms? To apply the atomic theory to chemistry would be a starting yet an obvious innovation. He had found in his experiments that in certain compounds of gaseous bodies, the same elements are always combined in the same proportions. The ringing declaration of a fellow scientist never ceased sounding in his ear: "God ordered all things by measure, number, weight." Why would this not be true of physics and chemistry? Why should an atom be the exclusive property of physics?
The possibilities of the theory were immense. Here, at last, was a simple principle fixing the proportions in which all chemical bodies combine. If the relative weight of one atom to that of any other atom were known, the proportions or weights in all its combinations might be ascertained. Once a table of relative weight of these ultimate particles could be established, the chemist would find himself in possession of tools for science. Dalton struck the final, fatal blow against the pretensions of alchemists who had promised to transmute iron into gold and death into life. Chemical analysis and synthesis, he declared, could go no further than to separate particles from one another, and to their reunion. No new creation or destruction of matter is within the reach of chemical agency.
Thus, having introduced the atomic theory from physics into chemistry, Dalton proceeded to set up a table of relative weights of atoms that constitute different elements. This table was rather crude because he neither possessed the skill and nor was his laboratory was properly equipped. However, he had established himself once and for all as a law giver.
During the course of his life, Dalton became quite famous. He was elected president of the Literary and Philosophical Society of Manchester. And he had been invited to lecture at the Royal Institution of London where he met the great Humphry Davy. His lecture tours had become a frequent matter. Once, when he visited Edinburgh, he wrote: "this is the most romantic place and situation I have ever seen." He was especially fond of observing ladies in his audiences and most of the ladies, irrespective of their dresses, looked charming to him. He was also very fond of beer. On one occasion, he suffered serious lead poisoning from a bottle of porter in a London pub. In due time, he recovered. Thus, he was glad to return to Manchester and to his comparatively obscure way of life.
Dalton was also elected by the Royal Society of England and the Academy of France. Sir Humphry presented him with the royal medal, and he responded with a speech. It seemed that the world would not let him stay by himself. He had become a goldfish swimming in the bowel of fame, which was unfortunate because he preferred peace and quiet.
For a while, nothing could lure Dalton away from Manchester. Humphry Davy invited him to a polar expedition sponsored by the Royal Society with the backing of the Admiralty. This opportunity meant a hefty sum of money and fame, but Dalton did not avail it.
Finally, however, he allowed himself once more to be lured into the world and the temptress was the city of Paris. Here, he met two of the most renowned contemporary fellow scientists, Humboldt and Laplace. These three scientists discussed the secrets of the heavens and the substances of earth. Wherever he went to Paris, Dalton was lionised. When he entered the precincts of the institute, the president and the members rose to and bowed - an honour which had not been accorded even to Napoleon. He was warmly welcomed wherever he went.
When he returned home from the Parisian triumph, he put away his sentimental memories and renewed the perpetual struggle of the mind against the stubborn fortress of ignorance. In recognition to Dalton's work, the world inscribed his name in shining words upon the rolls of the scientific academies of Berlin, Munich and Moscow. They had interceded with the British king to grant him a pension. A fund had been raised to erect a marble statue to immortalise him.

Death
Towards the end of his life, Dalton suffered a paralytic stroke and partially recovered to return to his laboratory, but the fires of his life were slowly being put out. One night, shortly after the completion of his statue, he tottered into his laboratory and groped for the books in which he had been recording the weather reports. Night after night for 50 years, he had paid the same, rigorous attention to the same, humble task -- nearly 200,000 readings in all. He noted the hour - - quarter to nine. It was precisely at this hour that he made his nightly recording. His hand trembled and he could hardly write in the final column: "Little rain". Dalton's head nodded and he began to put down his pen. Evening departed and morning came but Dalton's eyes remained closed - forever.