Lucid Knowledge Of, “Gold Number In The Chemistry Of Colloids”:
“Concept of Gold Number” at a Glance
The concept of gold number can be explained in short as below.
(1) Suppose we have an unstable colloidal solution of gold containing a fix amount of gold. It is known as standard gold sol.
(2) Due to its unstable nature, it readily coagulates (means gets precipitated) when small quantity of some electrolyte is added into it.
(3) But, we want to protect this gold sol from coagulation. Means we want to make it stable.
(4) We know that this can be done by adding a small quantity of peculiar substance into it before adding electrolyte. Such substance which can stabilize the given sol is called protective sol.
(5) The minimum quantity of protective sol required thus is called, “Gold Number” of that protective sol.
Thus "gold number" is a measure of strength of protective sol.
This will become clearer from following example:
(a) Suppose we want to determine the gold number of starch sol (means colloidal solution of starch) which is a well known protective sol.
(b) Gold sol (means colloidal solution of gold) is an unstable sol. This means when some electrolyte is added into gold sol it coagulates or gets precipitated. In this way colloidal properties of gold sol are destroyed by addition of electrolyte.
(c) We know that coagulation of gold sol can be prevented if we add some starch sol before adding electrolyte into it.
(d) We do not know how much quantity of protective sol is needed for this purpose.
(e) By conducting a series of experiments, we conclude that the minimum amount of starch required for this purpose is say "x" milligrams.
Here, "x" is called gold number of starch.
[Note: This example is given only to provide the basic concept of gold number. In actual practice, "standard gold sol" and "standard sodium chloride solution" are used].
The Definition of Gold Number
The concept of gold number was introduced by scientist, “Zsigmondy”.
Gold number is defined as,
“The minimum quantity of protective colloid expressed in milligrams, which is required to prevent the coagulation of 10 milliliters of standard gold sol, against the addition of 1 milliliters of 10 % aqueous solution of sodium chloride”.
Meaning of the terms: Sol, Stable Sol and Unstable Sol
There are three types of solutions:
(a) True solution,
(b) Colloidal solution and
(c) Suspensions.
All these three types of solutions differ in that the size of solute particle they contain is different.
True solution contains such solute particles, diameter of which is less than 1 nano meter (1 n. m.).
On the other hand suspension contains such solute particles, diameter of which is greater than 1000 n. m.
Sols: The colloidal solution contains such solute particles, diameter of which is in between of 1 n. m. and 1000 n. m. Thus diameter of solute particles of a colloidal solution is intermediate of above two types of solutions. Such colloidal solutions are also known as, “sols”.
Due to characteristic size of solute particles, colloidal solutions possess some special properties.
Stable sols: These are the sols which do not get coagulated (means precipitated or settle down) on addition of small quantity of electrolyte. This means their colloidal characteristic is not destroyed easily, hence they are stable.
Examples of such sols are: gum, gelatin, starch etc.
Because the colloidal particles (which are also called dispersed phase) of such sols have strong attraction with solvent molecules (which is also called dispersion medium), such type of sols are known as, “lyophilic sols”.
Unstable sols: These are the sols which are readily coagulated on addition of small quantity of electrolyte. This means their colloidal characteristic is destroyed easily, hence they are unstable.
Examples of such sols are: colloidal solutions of metals like gold, silver, platinum; metal hydroxide like ferric hydroxide [Fe(OH)3]; some metal sulphide like arsenic sulphide (As2S3) etc.
As the colloidal particles of such sols have no attraction but have hatred with solvent molecules, they are also known as, “lyophobic sols”.
What is meant by, “Coagulation of Sol?”
The loss of colloidal property of sol, by precipitation of colloidal particles, is known as coagulation of sol.
Coagulation can be done by adding a small quantity of electrolyte into the sol.
The mechanism of coagulation can be explained by following three points.
(A) It is found that in a colloidal solution, all colloidal particles carry either positive or negative charge. Due to this they repel each other which give kinetic energy to them. Hence all the colloidal particles are in random motion called, "Brownian motion". This prevents them to settle under the effect of gravity.
(B) Further, gravitational force of earth pulls the particles towards bottom of container so as to settle them.
(C) Thus, all colloidal particles are under the influence of these two opposite forces acting simultaneously.
(D) If the magnitude of force produced due to kinetic energy of particles exceeds that of gravitational, sol does not settle and becomes stable.
However, in case the magnitude of gravitational force exceeds that of kinetic energy of particles, colloidal particles would settle down under effect of gravity (means coagulation) and sol becomes unstable.
(E) On addition of electrolyte, electrical charge on colloidal particle is neutralized due to adsorption of oppositely charged ions of electrolyte.
In this situation, due to absence of repulsive forces they undergo collision leading to aggregation. This results in increase in size and mass of the resulting particle.
Now, gravitational force becomes appreciable and particles start settling, sol becomes unstable. This is known as coagulation of sol.
What is meant by, “Protective Sol?”
The unstable lyophobic sol can be protected from coagulation caused by addition of electrolyte. This can be achieved by previous addition of a suitable lyophilic sol in proper quantity. This phenomenon is known as protection of sol, and the lyophilic sol added is called, “protective sol”.
For example, if a proper quantity of gelatin is previously added to gold sol, it will not coagulate on addition of small quantity of sodium chloride. Here, gelatin is called protective sol.
Mechanism of Protection of Sol
By now we know that the phenomenon of aggregation of smaller particles of colloids produces bigger particles. These bigger particles, due to their greater mass, settle down under the effect of gravity which is called coagulation. However, due to repulsion among similar charged colloidal, aggregation does not take place. But adsorption of oppositely charged ions of electrolyte nullifies the charge on colloids leading to aggregation.
It is thus clear that, in order to stop the phenomenon of aggregation the adsorption of ions of electrolytes on the surface of colloidal particles must be stopped. This can be done as below.
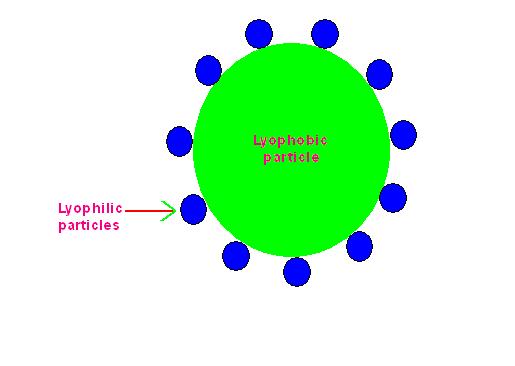
It is believed that when protective sol (means lyophilic sol) is added to unstable lyophobic sol, the molecules of lyophilic sol are adsorbed on the surface of molecule of lyophobic sol.
Due to this, the entire surface of molecule of lyophobic sol is covered with molecules of lyophilic sol.
This prevents the ions of electrolyte to get adsorbed on lyophobic sol molecules.
In this way lyophobic sol is protected.
See the following figure.
Picture Showing Action of Protective Colloid
Factors affecting the Color of Sol
The color of given sol depends upon following two factors:
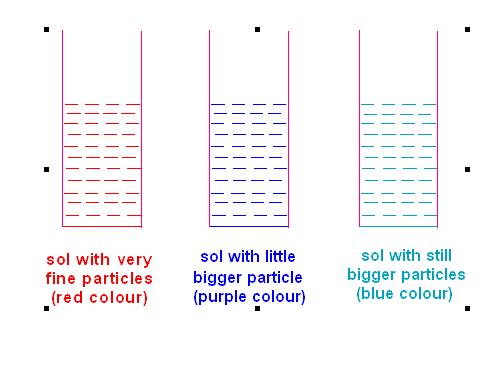
(1) Wavelength of light scattered by its colloidal particles.
But wavelength of light scattered depends upon the size of colloidal particles.
This means the same sol may show different colors with different size of colloidal particles.
This will become clearer from following example of gold sol.
Color of gold sol is red when particles are fine, but as the size of particles goes on increasing, its color changes first to purple then to blue and finally to golden-yellow!
This will become clearer by following figure.
(2) The way the observer receives the light.
For example, consider the color of a mixture of milk and water.
If the observer views the reflected light coming from this mixture, its color appears to be blue; but if the observer views the transmitted light coming from this mixture, its color appears to be red.
Picture Showing Variation in Color of Gold Sol with increasing size of Gold Particle
How to Prepare Gold Sol?
"Gold Sol" can be prepared by following two methods:
(1) Physical or mechanical method called, "Bredig’s arc method" and
(2) Chemical or reduction method.
Both of these methods have their own importance and application. They are discussed in detail as follows.
(1) Physical or Mechanical Method called, "Bredig’s Arc Method"
This method is also known as, “electrical dispersion method”.
In this method, two electrodes of pure gold are taken and immersed in ice-cold water. On passing electricity of high voltage through the electrodes, an electric arc is produced between the two electrodes. This results in vaporization of gold due to intense heat of arc. The vaporized gold particles are immediately condensed in cold water giving colloidal solution of gold. Addition of little quantity of stabilizing agent like potassium hydroxide is helpful.
(2) Chemical or Reduction Method
The colloidal solution of gold can also be prepared by reduction of its compound like gold chloride.
In this method a solution of gold (III) chloride (which is known as auric chloride, having chemical formula: AuCl3) is taken and treated with reducing agent like tin (II) chloride (which is also known as, stannous chloride, having chemical formula: SnCl2).
This results in formation of fine gold particles of colloidal size which remain suspended in the resulting solution to give gold sol.
The equation of chemical reaction taking place during this phenomenon is as under:
2 AuCl3 (aq) + 3 SnCl2 (aq) → 3 SnCl4 (aq) + 2 Au (s)
This reaction can also be carried out in aqueous formaldehyde medium as follows:
2 AuCl3 (aq) + 3 HCHO + 3 H2O (l) → 3 HCOOH (l) + 6 HCl (aq) + 2 Au (s)
(In above equations: aq = aqueous state, s = solid state and l = liquid state).
This method gives purple gold sol-called, "purple cassius".
What is called “Standard Gold Sol”?
The gold sol prepared above may contain varying quantity of gold particles. However, to get a comparable and constant value of gold number, it must contain a fixed quantity of gold in it.
For this purpose a gold sol containing 0.0055% gold content in it is taken and is termed as, “Standard Gold Sol”.
The Characteristics of Standard Gold Sol
The gold sol prepared thus has following characteristics:
(1) It is red in color
(2) It is unstable in nature, means it readily coagulates on adding an electrolyte like sodium chloride
(3) It contains colloidal particles of gold which are negatively charged, and
(4) It has the concentration of gold to be about 0.0055 %.
Gold Numbers of Some Common Protective Colloids
Different protective colloids have different capacities to protect standard gold sol from coagulation on addition of an electrolyte.
It is due to this reason that they have different gold numbers.
Gold numbers of some common protective sols are given in the following table.
Values of Gold Numbers of Some Protective Sols
Name of sol
| Value of its gold number
| |
|---|---|---|
Gelatin
| 0.005-0.01
| |
Casein
| 0.01-0.02
| |
Haemoglobin
| 0.03-0.07
| |
Albumen
| 0.1-0.2
| |
Gum Arabic
| 0.15-0.25
| |
Starch
| 20-25
|
Note that starch has the highest value of gold number.
Significance of the Value of Gold Number
The high value of gold number indicates that higher quantity of that protective sol is required to stabilize the standard gold sol.
It is therefore clear that, “higher is the value of gold number of the protective colloid, lesser is its protective capacity, and vice versa”.
It can be understood from the table that gelatin having smallest value of gold number has the highest capacity to protect gold sol from precipitation, while starch having highest value has lowest capacity.
Experimental Determination of Gold Number
(1) Knowledge of material required and their preparation:
In order to determine the gold number of given sol, following material is required which must be prepared carefully and accurately.
(a) "Standard gold sol:
“It is colloidal solution which contains a fixed quantity (0.0055 %) of gold”.
(b) Quantity of standard gold sol required for each determination:
The quantity of standard gold sol required is 10 milliliters. This means, 10 milliliters colloidal solution containing 0.0055 % gold is required for determination of gold number of given sol.
(c) Characteristic of electrolyte which should be added to check the coagulation of standard gold sol:
Electrolyte required here is, “1 milliliter of 10 % aqueous solution of sodium chloride”.
(d) Minimum quantity (in milligrams) of given protective sol which can stop the coagulation of said gold sol:
This can be determined by progressive addition of extremely small quantity of protective sol. If minimum quantity found thus is Z mg then “Z mg” is called, “Gold Number” of that particular protective sol.
For example, gold number of starch is 25.
This means when 25 mg of starch is added to 10 milliliters of standard gold sol and then-after if 1 milliliter of 10% aqueous solution of sodium chloride is added to it, no coagulation of gold sol will occur!
Numerical for Practice
During an experiment, it was found that coagulation of 100 milliliters of gold sol can be prevented, if a minimum quantity of 0.25 grams of starch is added to it before adding 1 milliliter of 10 % aqueous solution of sodium chloride. Determine the gold number of starch.
Here, amount of starch added to 100 milliliters of gold sol = 0.25 grams
= 250 milligrams
Hence the amount of starch required for 10 milliliters of gold sol = 25 mg.
Therefore, gold number of starch = 25.
References:
(1) A Text Book of PHYSICAL CHEMISTRY for COMPETITIONS, by Dr. O. P. Tandon and Dr. A. S. Singh, Published by G. R. Bathla & Sons, 654, Shivlok, W. K. Road, Meerut, (U. P.), India
(2) Pradeep's New Course Chemistry, Class XII, Vol. I, 27th edition, Pradeep Publication, Jalandhar, India
(3) A TEXT BOOK OF PHYSICAL CHEMISTRY, 4th Edition, by Dr. R. K. Gupta & R. K. Amit, Published by: Arihant Prakashan, Kalindi, T.P. Nagar, Meerut-250002, India
(4) Modern's abc Of Chemistry, For Class XII, Part-I, by Dr. S. P. Jauhar, Published by: Modern Publishers, MBD House, Railway Road, Jalandhar City, India
(5) I. I. T. Chemistry, by Dr. O. P. Agarwal, 135th edition, Jai Prakash Nath Publications, Meerut, India
(6) Nootan ISC Chemistry, Class XI, by Dr. H. C. Srivastava, Published by: Nageen Prakashan (Pvt.) Ltd., 310, Western Kutchery Road, Meerut-250001, U.P., India
Previous
- Lucid Understanding Of, “Dipole Moment Of A Molecule”:
Do You Know On Which Basis Shape Of A Molecule Is Determined? Do You Know How Percentage Of Ionic Character Of A Covalent Compound Is Determined?
Next:
- Lucid Guideline For I.U.P.A.C. Nomenclature Of Organic Compounds: Part-1:
Do You Know there are about seven million known compounds in the dictionary of organic chemistry? IUPAC system of naming does not require memory power to remember the name of organic compound.