Properties of Matter
Properties of Matter
A. Physical Properties
Properties of matter which can be observed without changing the substance into some new kind of matter are called physical properties. Physical properties like odor, color, taste, transparency and physical states are determined by our senses and therefore they are not always reliable. While density, boiling point and freezing point are physical properties that are reliable because they can be measured and numerical values are assigned to them.
B. Chemical Properties
Chemical properties are those properties that can be observed only when the substance undergoes a change in composition. These are also the properties that determine the ability of a substance to react with other substances. The capacity of carbon to combine with oxygen at elevated temperature to form carbon dioxide and iron oxide to react with coke to produce metallic iron and carbon dioxide are chemical properties.
Physical States of Matter
There are four physical states of matter-gas, liquid, solid and plasma.
1. Gases are substances that neither have definite shape nor definite volume.
2. Liquids are substances that have definite volume but no definite shape, they just follow the shape of the container.
3. Solids are materials that have definite volume and definite shape.
4. Plasma is a mixture of sub-atomic particles nuclei and electrons.
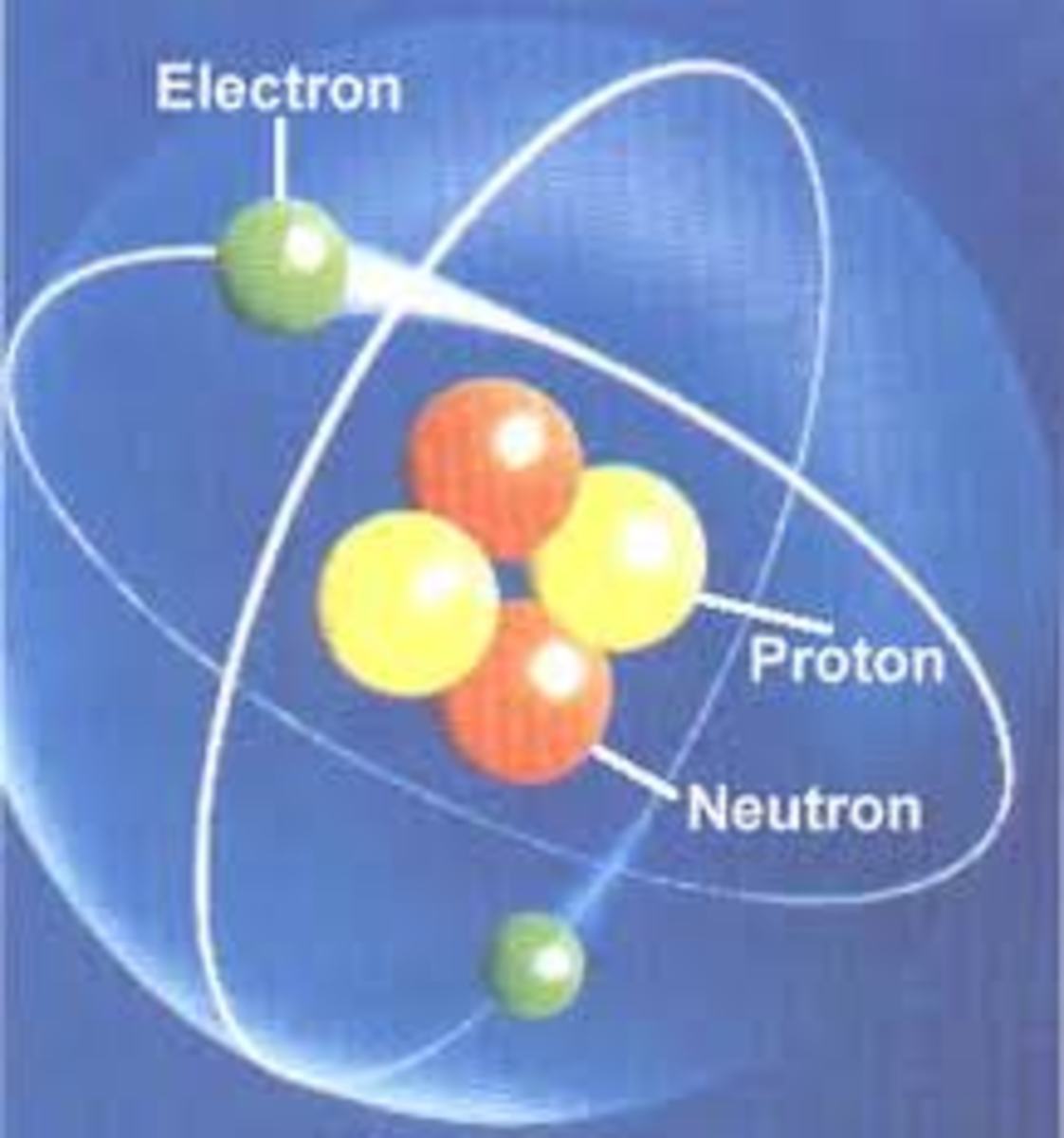
Structure of an Atom
The basic building block of matter is called atom. An atom is extremely very small, it measures only from less than one to two Angstrom. An Angstrom is equivalent to one ten-thousandth of a micron.
Parts of an Atom
A. Nucleus is the tiny central part of an atom that contains the protons and the neutrons.
B. Sub-atomic Particles
1. Protons are the positively charged particles of an atom. Each proton carries an electronic charge of+1.
2. Neutrons are the neutral particles of an atom since they have the charge.
Protons and neutrons have roughly the same mass. The number of protons in the nucleus is unique for each element and this number is called the atomic number. While the total number of protons and neutrons in a neucleus is called the atomic mass.
3. Electrons are the negatively charged particles of an atom that are found outside and revolving around the nucleus. Each electron carries a charge of-1, its charge is exactly the opposite of that of a proton. Electrons have very little mass, as a result, almost the total mass of atom is contributed by the protons and neutrons in the nucleus, even though the extranuclear-region constitutes most of the volume of the atom.8
C. Shells contain electrons travelling at the same average distance from the nucleus. They are named from letters K, L, M, N, O, P, & Q starting from the innermost to the outermost. As the atomic mass from an atom increases, the number of shells also increases.9
Kinds of Matter
Matter is either pure substance or mixture. Pure substances are defined as those whose composition in terms of its elemental constituents does not change when subjected to processes such as freezing, boiling, condensation, evaporation, recrystallization, and solution. Copper, sugar cane and distilled water are examples of pure substances. There are two kinds of pure substances: elements and compounds. Element is made up of only one kind of atom while compound is made up of 2 or more different kinds of atoms. There are three kinds of element namely: metal, non-metal and metalloid. Metals are good conductors of heat and electricity while non-metals are good insulators against heat and electricity. Zinc, iron, aluminum, tungsten, gold and lead are some examples of metals while sodium, hydrogen, oxygen, and helium are some examples of nonmetals. Metalloids are called borderline forms of elements or intermediate elements since they possess both the characteristics of metals and non-metals. There are only four metalloids, namely: arsenic, boron, germanium and silicon.
Compounds may be organic or inorganic. Organic compounds contain carbon atoms while inorganic compounds usually do not have carbon atoms. Methane, carbohydrates, proteins, gasoline and kerosene are examples of organic compounds while carbon dioxide, sodium chloride, water, sulfur dioxide and ammonia are examples of inorganic compounds.
Mixture is made up of two or more pure substances jumbled together either homogeneously or heterogeneously. Homogeneous mixture is made up of components that cannot be easily distinguished even through the aid of a powerful microscope. Salt and water, sugar and water are two examples of homogeneous mixtures. A heterogeneous mixture, meanwhile is made up of components that can be easily identified. Oil and water or rock and water are two examples of heterogeneous mixtures.