Quantum Physics - Niels Bohr atomic theory
Niels Bohr

Niels Bohr had a very successful family, he was born in 1885 in Copenhagen, his farther was the professor of physiology at the University of Copenhagen and has the bohr shift attributed to him. His mother came from a very affluent Jewish family who were prominent in the Danish banking and parliamentary circles and his brother was a mathematician and Olympic footballer.
He attended the GammelholmGrammar school, were he was very successful excelling in maths and sciences. In 1903 he joined the undergraduate program at the CopenhagenUniversity where he originally studied philosophy and mathematics.
However in 1905 he decided to change from philosophy and take up physics, and what a choice this was. He received his master’s degree in physics in 1909 and his doctorate in 1911, where he was mentored by Professor C. Christiansen who developed and nurtured Bohr’s natural flair for physics.
While still a student, an announcement by the Academy of Sciences in Copenhagen of a competition to find the solution of a certain scientific problem caused him to take up an experimental and theoretical investigation of the surface tension by means of oscillating fluid jets. This work, which he carried out in his father's laboratory and for which he received the prize offered was published in the Transactions of the Royal Society, 1908.
Bohr’s focus became more and more on the theoretical side of physics, with his dissertation being still a classic today in theoretical physics. It was his work on electron theory relating to metals which first introduced Bohr to Plank’s quantum theory of radiation.
Bohr's model of a Hydrogen Atom
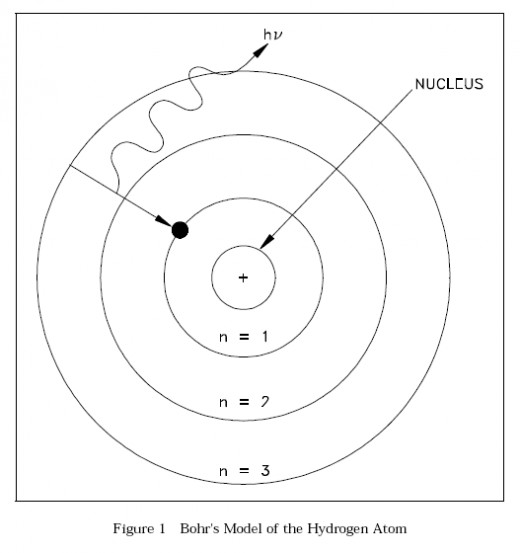
After completing his doctorate he went on to conduct his first experiments at Trinity College Cambridge, In 1912 he moved on to study under the famous Ernest Rutherford at the University of Manchester, it was here that he published his model of the atomic structure introducing the idea that electrons orbited the nucleus of an atom in pre-defined energy levels and if they dropped an energy level they would emit a photon, and this is what became the basis for quantum theory; So a very important paper.
In 1916 he became professor at the University of Copenhagen, where other than a few short terms in other institutions he would remain until his death. With the support of the Danish government he succeeded in founding the Institute of Theoretical Physics in 1921 of which he became director. In 1922 he received the Nobel Prize in recognition of his previous work on the structure of atoms and the emission of radiation from them.
Information on Bohr's contribution to Quantum Physics
Borh and Einsein disscussing Quantum Theory

Great Books on Niels Bohr and his Theories

This institute was the leading department of Theoretical Physics in the 20’s and 30’s and most of the best minds in theoretical physics spent some time here. Bohr also hypothesised the principle of complementarity; which means systems can be separately analysed as having many contradictory properties, for example the wave-particle duality we have been discussing, light behaves as both a wave and a particle dependent upon what we are measuring.
He was known to have heated but friendly arguments with Einstein about his next principle which was the introduction of the new probabilistic quantum physics. Einstein much preferred the classical physics and was famously remembered for saying:
“god does not play dice”
to which Bohr responded:
"Einstein, stop telling God what to do!”
He also had a habit of saying:
“We are all agreed that your theory is crazy. The question which divides us is whether it is crazy enough to have a chance of being correct. My own feeling is that it is not crazy enough.”
Which just shows he was a brilliant mind and capable of accepting and producing theories which are in complete contradiction to reality.
Heisenberg another physicist I will be discussing in this series worked as an assistant to Bohr and as a lecturer from 1926 -27, where under guidance from Bohr he developed the great Uncertainty Principle.
Bohr's activities were since 1930 more and more directed to research on the make up of the atomic nuclei, and of their transmutations and disintegrations. In 1936 he pointed out that in nuclear processes the smallness of the region in which interactions take place, as well as the strength of these interactions.
In 1942 Bohr was arrested by the German police and from which he escaped to Sweden and then traveled to London.
From here Bohr worked on the top secret Manhattan project where he was known as Nicholas Baker. His job was to be a consultant for the project to oversee and advise the project. However Bohr was very concerned about the nuclear arms race taking place and believed passionately that the atomic secrets should be shared with the international science community. Bohr visited Winston Churchill in England to try to convince him after this was suggested by Roosevelt. However Churchill disagreed with the idea of openness with the Russians in fact so strongly he suggested Bohr is locked up because he was very close to committing a very serious crime.
After the War Bohr returned to Copenhagen promoting the peaceful use of nuclear weapons. Until the end he remained clear headed and as alert as ever and developed a keen interest in the new field of molecular biology where the very last of his thoughts were put into an unfinished paper “Light and Life revisited”. He died in 1962 of natural causes.