The Chemistry Behind Global Warming

What Is Global Warming?
Definition: The gradual increase of the earth's atmosphere believed to be due to the 'greenhouse effect'.
Facts about Global Warming:
- In the last 20 years the amount of CO2 in our atmosphere has nearly doubled.
- Average temperatures in Alaska, western Canada, and eastern Russia have risen at twice the global average.
- The last two decades were the hottest decades in 400 years.
- Arctic ice is rapidly disappearing, and it is predicted that the Arctic may have its first completely ice-free summer by 2040.
- Coral reefs, which are highly sensitive to small changes in water temperature, suffered the worst bleaching in response to stress ever recorded.
- Things such as industrialization, deforestation, and pollution have greatly increased the concentrations of of water vapor, carbon dioxide, methane, and nitrous oxide in our atmosphere.
The Greenhouse Effect - Not Always Bad!
The words 'Greenhouse Effect' when talked about in the media or in general conversation always have negative connotations. You hear the words and you immediately imagine dying polar bears and rising sea levels however, the greenhouse effect isn't always a bad thing.
Without greenhouse gases our earth would be 35o lower than it is now and would be completely covered in ice.
Greenhouse gases determine the temperature of our planet more than the proximity to the sun does!

The Science
The main greenhouse gases are:
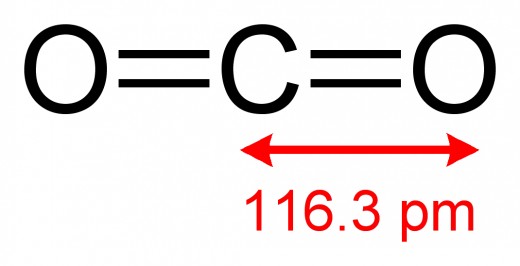
- CO2 - Carbon Dioxide
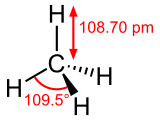
- CH4- Methane
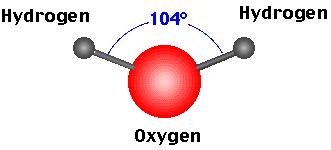
- H2O - Water (vapour)
- N2O - Nitrous Oxide
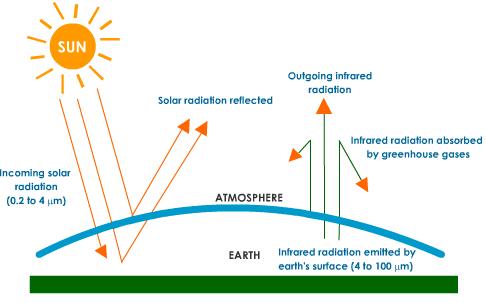
- The earth receives most of its energy in the form of visible electromagnetic radiation from the sun.
- This short-wave solar radiation is not impeded by the greenhouse gases and thus enters our atmosphere and passes straight through to the earths surface.
- The solar energy is absorbed at the surface and some is released back into the atmosphere but as longer waved, infrared radiation.
- The various greenhouse gases in the atmosphere will absorb this infrared radiation and emit this energy back towards earth.
How do the gases absorb the radiation?
- The bonds of theCO2, CH4orH2O can bend and absorb the infrared radiation which causes the molecule to vibrate.
- This absorption gives the molecule kinetic energy.
- The vibrating molecule then emits some of this energy in the form of infrared radiation.
- This extra kinetic energy and radiation may then be transmitted to other molecules and this causes a general heating of the atmosphere.
- The greenhouse effect of a gas depends not only on its concentration in the atmosphere, but also its ability to absorb infrared radiation.
- This absorption-emission process keeps heat close to the Earth's surface.
Global Warming Potential (GWP)
- The ability of a gas to cause global warming is described by its GWP.
- The GWP of a gas takes into account the lifetime of the gas in the atmosphere as well as its ability to absorb infrared radiation.
- Some chloroflourocarbons (CFCs) are 25,000 times more efficient at absorbing infrared radiation that carbon dioxide but luckily the use of CFC's have been banned and since then the effect that they had is fading.

Greenhouse Gas
| Sources
| Estimated Proportion Of Total Effect (excluding water vapour)
|
|---|---|---|
Methane
| Agriculture, Fuel Leakage
| 20%
|
Carbon Dioxide
| Fossil Fuel Burning, Deforestation, Volcanic Eruptions
| 60%
|
Nitrous Oxide
| Combustion, Ariculture
| 6%
|
Halocarbons
| Refrigerants
| 14%
|
To learn more about the environment and the effects that humans have on it here are 2 hubs about deforestation and coral reefs that may interest you.








