The Domesication of Corn: Teosinte Glume Architecture (tga1)

Historically important factors and modern day ramifications of the domestication of teosinte were covered in Corn Domestication and Importance. In this hub, one of the genes that was selected by humans will be looked at for a further, more in-depth view of corn domestication. Teosinte glume architecture (tga1) will be the focal point of this hub.
This gene controls the attributes of the kernels (seeds) on both the teosinte and corn plants. It controls the way the kernel is structured and attached to the plant.
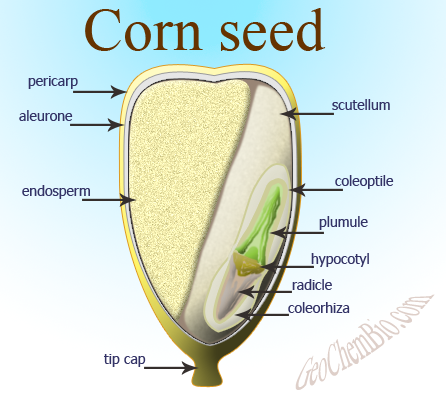
Kernel Anatomy
Before we get into talking about how the kernel changed, lets first talk about its anatomy, which is comprised of four parts:
- Pericarp: This is the outer layer or "shell". It is not a part of the embryo, but, "'packaging' from the parent" (Barnhart and Hopper 2012). The pericarp is the ovary wall and often refereed to as the fruitcase. The kernel is actually a fruit as the pericarp and seed coat are fused.
- Aleurone: A layer of cells, which may or may not be colored, found inside the pericarp (Barnhart and Hopper 2012). This protein and lipid casing separates the pericarp from the endosperm.
- Endosperm: The starchy part of the seed and takes up the majority of the seed (Barnhart and Hopper 2012). This is the edible portion of the plant and is the nutritional part humans were looking to consume.
- Embryo: This is the part of the kernel, that if planted, germinates into a new corn plant.
Kernel Changes
The kernels are attached to the plant by the ear. This portion of the cornstalk is female. Our ancestors selected against the attributes of teosinte kernels that made it capable of survival on its own. This portion of the hub will be very simplistic and straight to the point.
The most obvious difference between teosinte and corn are the number of kernels. Teosinte typically has 5 - 12 kernels, while maize can have 500 or more. This change allowed agriculturalists to plant fewer seeds with a higher crop turn out. This in itself could have been a big enough reason to genetically modify a plant. With less work, more food can be produced.
Another change to the kernel from teosinte to maize is the physical structure of the kernel. Teosinte seeds are constructed with a hard outer casing that allows the kernel to pass more easily through the digestive tract. From a survival of the fittest standpoint, this is great. For a cash crop, this is not good. People had to laboriously grind up the kernels for human consumption. However, agriculturalists started to select against the genes of the teosinte plant. This later transformed into the corn cob we now know today. The kernels lost their stony casing and became much easier to grind up while making the starchy part more easily accessible.
With that said, what gene was selected to enable these changes and what exactly did it do?




Teosinte Glume Architecture (tga1)
Teosinte glume architecture (tga1) is the sole gene, which controls the exposure of the kernel on the surface of the ear. It is a large effect quantitative trait locus (QTL), which are stretches of DNA containing or linked to the genes that underlie a quantitative trait, that segregates as a single mendelian locus with a genetically identical background, which suggests that it may represent a single transcription unit (Bomblies et al. 2005; Doebley 2005). It is a part of the SBP-domain family, which act as transcriptional regulators. Transcriptional regulators control the gene "information" for forming their final product in the cell or virus.
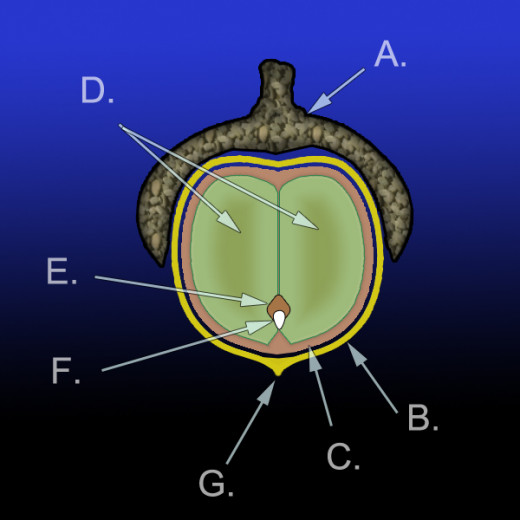
One key aspect present on teosinte, which is imperative for its survival, is the presence of the cupulate fruitcase. This fruitcase is formed from an invaginated internode (cupule), which holds the kernel and is covered by a glume to hide it (Bomblies et al. 2005). The best way to describe this is to think of an acorn. The acorn has a cup shaped bottom shell (invaginated internode), which is covered on top (glume) to protect the nut inside (kernel). When maturity is reached, the epidermal cells are filled with silica, which gives the fruitcase its stony appearance and hardness (Bomblies et al. 2005; Doebley 2004; Doebley and Dorweiler 1997). Maize is very different and uses the glumes and cupules to form the cob. The organs forming the teosinte fruitcase are present on maize, but form the cob for which the naked kernels attach (Bomblies et al. 2005; Doebley 2004; Doebley and Dorweiler 1997).
Developmental analyses of tga1 have shown that it affects several aspects of fruitcase development for teosinte. Doebley and Dorweiler (1997) conducted a study to show tga1 transformed multiple aspects of the cupulate fruitcase. The allele found in teosinte is responsible for creating the cup (invaginated internode), forming longer and thicker glumes, increased hardening of glumes due to thicker abaxial mesoderm of lignified cells, and it directs silica depositions to the epidermal cells of the fruitcase (Bomblies et al. 2005; Doebley 2004; Doebley and Dorweiler 1997).
However, the early stages of development in teosinte and maize produce a bisexual plant. Thus, at some stage in the plants development either the female or male organs become dominant. Adult sex is determined by an internal signal that then aborts either the male or female organs based on the signal that has been triggered (Bomblies et al. 2005; Doebley 2004; Doebley and Dorweiler 1997). In other words, if the male organ (tassel) is chosen then the female (ear) is aborted and vice versa. The study by Doebley and Dorweiler (1997) showed that no later than the I1 stage is when differentiation begins and that tga1 expression is restricted to female plants. The I signifies immature and the 1 has to do with spikelet or unit of flowering length. In total, there were eight stages and I1 was the second stage. This goes to show that tga1 does in fact control the characteristics of the female teosinte plant as previously described and it has to be activated at a certain stage in the plants growth. tga1 activates invagination and elongation of the internode, glume growth, silica deposition, and pattern of lignification in female teosinte plants(Bomblies et al. 2005; Doebley 2004; Doebley and Dorweiler 1997).
Proving this showed that an allele change in tga1 from teosinte to maize was vital in the evolution of the crop. Past agriculturalists selected against this allele variation found in teosinte to reduce the cupulate fruitcase and expose the kernel. When the allele variation found in maize proved to be more productive, from a subsistence standpoint, they began to select that variation to allow for the crop to be more easily consumed. Instead of having to grind up the outer fruitcase to get to the kernel, this maize allele allowed for easy access to the consumable part of the plant.
(The video below, uploaded by THEN3TWORK, is a look into modern changes of crops.)
Genetically Modified Foods
Conclusion
This hub looked at the tga1 gene and the affects it had on teosinte and how humans selected variations of this plant that brought about the domestication of maize. tga1 locked in the kernel and kept it in its cupulate fruitcase. While this proves to be useful in ensuring plant survival, it is not good for human consumption. People had to laboriously grind up the fruitcase to reach the edible kernel. Selecting against this gene and decreasing its teosinte allele frequency exposed the kernel and provided a stable staple crop. The selection of this gene allowed for exposed kernels to be placed on a fully developed core that proved to be very suitable for human consumption.
Sources
2012 Barnhart. M & Hopper T. Laboratory manual for general biology 121, 2nd Edition 60.
2005 Bomblies, K. et al. The origin of the naked grains of maize. Aug. 4, 2005; 436, 7051; ProQuest pg. 714.
1999 Coen, E., Cubas, P., Doebley, J., Lauter, N. The tcp domain: A motif found in proteins regulating plant growth and development. The Plant Journal 18(2), 215-222.
2004 Doebley, J. The genetics of maize evolution. Annual Review of Genetics 38, 37-59.
1997 Doebley, J. Developmental analysis of teosinte glume architecture: A key locus in the evolution of maize (poaceae). American Journal of Botany 84(10), 1313-1322.
© hockey8mn, 2012. All Rights Reserved.