The Elements
Symbols
Each element has an official symbol, which is derived from its name. The first letter in the symbol is always capitalized, and the second letter (if there is one) must be written in lowercase.
Example: the symbol for magnesium is Mg; the symbol for manganese is Mn. There are numerous websites and printed references where you can find a listing of the elements and their symbols. A good website to refer to is webelements.com.
Some elements have symbols that do not resemble their Englsih names. The symbols for these elements are derived from their ancient names.
Example: the symbol for sodium is Na, which is derived from its Latin name (natrium)
Other examples: Gold (Au, aurum), Silver (Ag, argentum), Mercury (Hg, hydrargyrum), Iron (Fe, ferrum), Copper (Cu, cuprum), Antimony (Sb, stibium), Lead (Pb, plumbum), Tin (Sn, stannum), Potassium (K, kalium), Tungsten (W, wolfram).
Exercise: Which of the these elements is paired with its correct symbol?
A. magnesium, Mn, B. potassium, Po, C. fluorine, F, D. argon, Ar
Answer:
Exercise: Which of the these symbols is paired with its correct name?
A. P, phosphate, B. Si, silver, C. Hg, hydrogen, D. Pb, lead
Answer:
The Periodic Table
The periodic table is a chart that facilitates the understanding of the properties of elements. In a typical periodic table, elements are grouped into 18 vertical columns with two additional (horizontal) rows below the 18 columns. Elements that belong to same vertical column are said to belong to the same family or group. Elements in the same horizontal row are said to belong to the same period.
Representative, transition and inner transition elements.
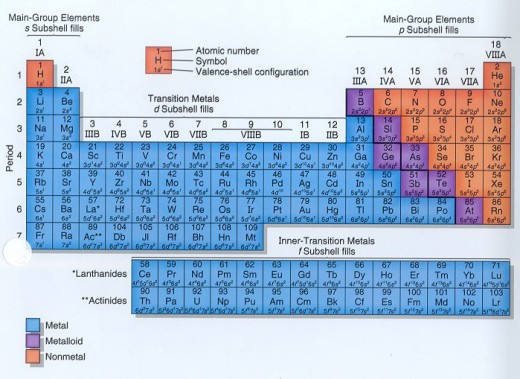
The 18 vertical columns are numbered 1 through 18. In the traditional American periodic table, columns 1, 2, and 13 through 18 are labeled using Roman numerals followed by the letter A. In other words, column 2 is group IIA, and column 17 is group VIIA. Sometimes, column 18 is labeled as group 0 (zero). These "A" groups are known as the main groups or representative groups, and they are indicated in Figure 1 above by the red shading. In the traditional American periodic table, columns 3 through 12 are labeled using IIIB, IVB, VB, VIB, VIIB, VIII (next three columns), IB, and IIB. These "B" groups are called the transition elements or transition metals and they are indicated in the figure above by the blue shading The main group elements Ga, In, Sn, Tl, Pb, and Bi are also known as the post-transition metals. The two rows at the bottom are known as the inner transition elements (lanthanides and actinides); they are indicated above by the yellow shading. In the extended- or long-form of the periodic table, these rows are inserted between group IIA and group IIIIB in rows 6 and 7. In other words, the lanthanides actually belong to period 6 (or row 6) and the actinides belong to period 7.
Some groups have special names. Elements in Group IA, except hydrogen, are called the alkali metals. Elements in group IIB are called the alkaline earth metals. The transition elements are also known as the transition metals. Elements in Group VIIIA (column 18) are called the noble gases. Elements in Group VIIA (column 17) are known as the halogens. Elements in group VIA (column 16) are known as the chalcogens.
Exercise: Which of the following elements is a transition metal?
A. silicon, B. lead, C. sodium, D. chromium
Answer:
Exercise: Which of the following elements is a post-transition metal?
A. silicon, B. lead, C. sodium, D. chromium
Answer:
Exercise: Which of the following elements is a metal that belongs to a main group or representative group?
A. magnesium, B. zinc, C. sulfur, D. uranium
Answer:
Exercise: All of the following are true, except...
A. Sodium is an alkali metal, B. Neon is a noble gas,
C. Calcium is an alkaline earth metal, D. Oxygen is a halogen
Answer:
Metals, Nonmetals, and Metalloids
Elements can be classified as metals, nonmetals, or metalloids. Metals are shiny, malleable, good electrical conductors, and good heat conductors; they are found on the left side of the periodic table as indicated by the red shading in Figure 2 below. Nonmetals are found on the right side. Elements in between (B, Si, Ge, As, Sb, The and Po) are known as the metalloids.
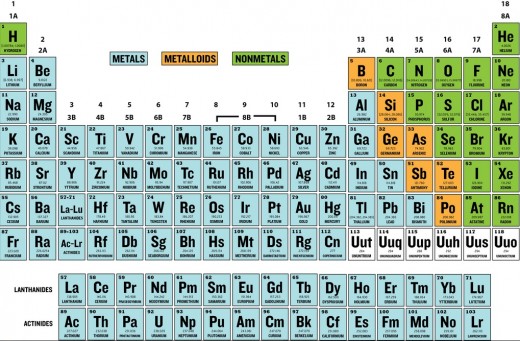
Exercise: Which of the following is a metalloid?
A. Potassium, B. Silicon, C. Iron, D. Helium
Answer:
Exercise: Which of the following elements is the best electrical conductor?
A. carbon, B. germanium, C. manganese, D. iodine
Answer:
Naturally-occurring Forms of Elements
Most naturally-occurring elements are solids under ordinary conditions. The exceptions:
- Liquids: mercury and bromine.
- Gases: hydrogen, oxygen, fluorine, chlorine, and the noble gases.
On a warm summer day, outside an air-conditioned laboratory, the following elements may also be found as liquids: gallium (melts at 29.76oC or 85.57oF) and caesium (melts at 28.44oC or 83.19oF)
Some elements can exist in different forms (called allotropes), with different properties. The differences are due to the differences in the way that the atoms are linked together. For example, oxygen can exist as a collection of O2 molecules or O3 molecules (ozone). O2 is the form of oxygen that makes up about 20% of the air that we breathe. The most common, most stable forms of some elements under ordinary conditions are listed below.
- Diatomic gases: H2, N2, F2, Cl2,
- Diatomic liquid: Br2,
- Diatomic solid: I2
- Polyatomic solids: P4, S8
- Monatomic gases: noble gases
Exercise: Which of these naturally occurs as a monatomic gas under ordinary conditions?
A. helium, B. sodium, C. oxygen, D. nitrogen
Answer:
Exercise: Which of these elements naturally occurs as a diatomic gas under ordinary conditions?
A. bromine, B. iodine, C. mercury, D. chlorine
Answer:
Exercise: Which of these elements is not a gas under ordinary conditions?
A. bromine, B. helium, C. hydrogen, D. oxygen
Answer:
References
© 2015 Discover the World





