Three Major Classes of Plant Hormones and their Practical Uses
There have been discrepancies over the definition of hormones. They were classically defined as, Feher (2012, p761) said, an “organic substance that is released into the blood at low concentrations by a ductless gland, and that travels through the circulation to a remote target tissue where it evokes systemic adjustments by engaging specific responses”, nonetheless it is clear to this definition is not relevant for plants and has numerous issues e.g. they are not just released into the blood. The general definition for plant hormones is that they are organic compounds that are synthesised in one part of the plant and they are translocated under low concentrations to another part of the plant, where it causes a physiological response, however there are some exceptions, for example ethylene is not translocated (TNAU, 2008).
The categories of plant hormones are: auxins, gibberellins, cytokinins, ethylene, dormins (such as Abscissic Acid and Phaseic Acid), flowering hormones (such as florigin, anthesin, vernalin), phenolic substances (e.g. coumarin), miscellaneous natural substances (e.g. vitamins, phytochrome tranmatic substances), synthetic growth retardants (e.g. phosphin-D, morphacting, malformis) and miscellaneous synthetic substances (such as synthetic auxins, synthetic cytokinins) (TNAU, 2008). The chosen major classes for discussion are: Ethylene, Cytokinin and Auxin [Table 1].
Table 1 Three Hormone Classes and their Practical Uses
Hormone
| Practical Uses
|
|---|---|
Ethylene
| Stimulates ripening, latex production, formation of aerenchyma in submerged roots and stems, senescence, initiation of root hairs, abscission, thigmomorphogenesis, epinasty stimulation, seed germination, inhibits shoot and stem growth
|
Cytokinin
| Mimic the effects of Phytochrome and red light, stimulates the transition of cells from the G2 phase to M phase of the cell cycle, delay senescence, promoting cellular division and expansion in cotyledons, organogenesis, bud activation and fruit and embryo development
|
Auxin
| Delay senescence, cause abscission, stimulates apical dominance, stimulate fruit development and maturation, cell elongation, formation of roots in cuttings, tropisms, vascular tissue differentiation, stimulate the development of adventitious roots
|
Figure 1 Differences in lysigenous aerenchyma formation and patterns of radial O2 loss (ROL) in rice roots under drained soil conditions and waterlogged soil
Click thumbnail to view full-size
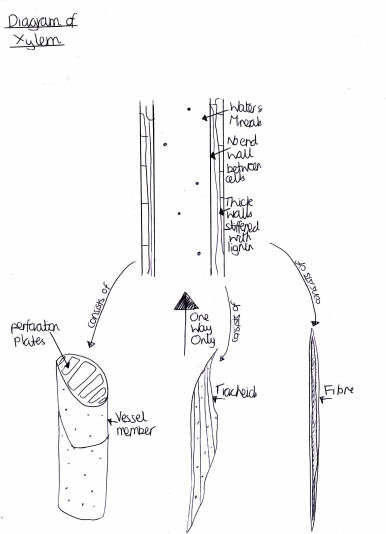
Figure 2 Diagram of Xylem
Click thumbnail to view full-size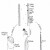
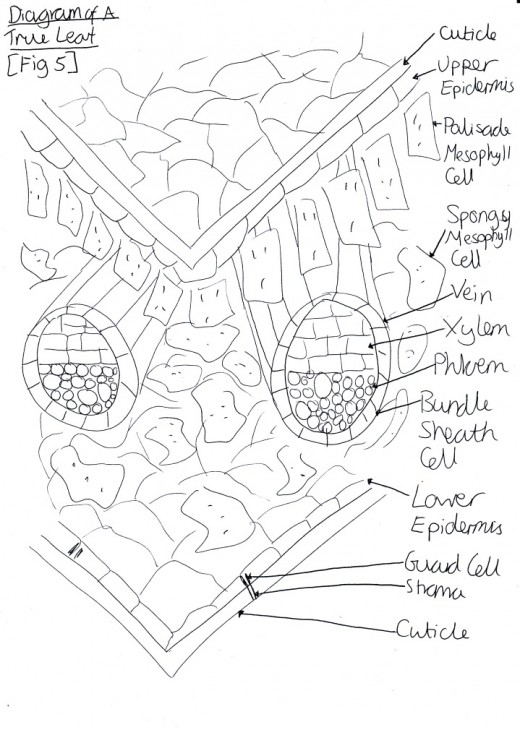
Figure 3 Diagram of a True Leaf
Click thumbnail to view full-size
Ethylene and its Practical Uses
Ethylene is a ripening hormone which is biosynthesised by methionine to S-adenosyl-L-methionine, using Met Adenosyltransferase as a catalysis [Table 1]. It is the only gaseous plant hormones and has the most simple structure (Mauseth, 2008). The hormone was found to be the produced in illuminating gas which caused trees to lose leaves and develop thick, short stems (Shimamura et al., 2010).
Ethylene stimulates the ripening processes by causing starches to convert into sugars, flavours and aroma to develop and colour to change (Mauseth, 2008). When ethylene is produced, the positive feedback system occurs in climactic fruits (e.g. apples, mangoes, tomatoes), thus more ethylene is released. This rapid increase (e.g. 320 nL/g/hr) in the hormone causes climacteric, a stage of fruit ripening which involves an increased respiration rate (Pujade-Renaud et al., 1994). Ethylene’s involvement with ripening is important commercially, for example, humans causing the uniform abscission of cherries and cotton or synchronising the ripening and flowering of pineapples to pick them easier. It has also been manipulated to prevent the ripening processes, for example by removing the ethylene (or adding 2-chloro-ethylphosphonic acid to break up the ethylene) from fruit to preserve them and harden them for transportation (Mauseth, 2008).
The hormone ethylene is also used to stimulate latex production by treating the bark of rubber trees with ethephon (an ethylene releaser). Kush et al. (1990) expanded this idea by finding the lacticifer-specific genes in H. brasiliensis which were induced by ethylene. The hormone, it has now been found, upregulates the activity of glutamine synthetase (an important enzyme of nitrogen metabolism and transcript levels in H. brasiliensis) (Pujade-Renaud et al., 1994).
Ethylene is also responsible for the formation of aerenchyma in submerged roots and stems [Figure 1]. Aerenchyma are air channels in the roots of some plants, which allow exchange of gases (Shimamura et al., 2010). They provide low-resistance O2 transport pathway that enhances plant survival during soil flooding. Drew et al. (1985), found a higher adenylate energy charge in adventitious roots of Zea mays under hypoxic conditions that developed aerenchyma, compared with roots in which formation of aerenchyma was blocked by the application of silver ions to inhibit ethylene action.
Ethylene also stimulates many other processes and cells, including: senescence of mature xylem cells [Figure 2], leaves [Figure 3] and flowers; initiation of roots hairs; thigmomorphogenesis and epinasty (leave curvature as the petiole grows) (Jackson, Campbell, 2006). It also plays an important role in inducing leaf abscission and seed germination. Although, the hormone is most well-known for the stimulatory plant responses, it is also responsible for inhibiting other aspects of the plant, e.g. shoot growth, and stem growth (Shimamura et al., 2010).
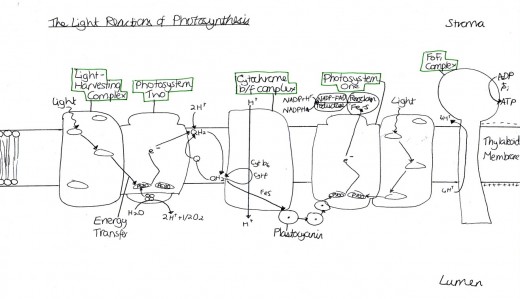
Figure 4 Light Reactions of Photosynthesis
Click thumbnail to view full-size
Cytokinin and its Practical Uses
Cytokinins have their particular name because when they are added to a tissue culture medium containing auxin and sugar, cell division (cytokinesis) is initiated [Table 1]. There are more than 200 natural and synthetic cytokinins, with the most common being Zeatin (isolated from corn) (Maueth, 2008). The highest concentrations of cytokinins are in meristematic regions and areas of growth within roots, young leaves, developing fruits, and seeds. Their discovery was related to work on tissue culture and when Van Overbeek found that plant embryos have more rapid growth when they are exposed to coconut milk. Cytokinin biosynthesis is done using the mevalonate pathway.
Cytokinins mimic the effects of Phytochrome and red light in many cases (Mauseth, 2008). Plant growth and development is modulated by internal cues e.g. hormonal balance and external factors (such as the light in the environment). Fankhauser (2002, p1) stated that “a cytokinin receptor was recently identified together with several elements acting in this signalling pathway. ARR4, a response regulator working downstream of a cytokinin receptor, has been shown to regulate phytochrome B-mediated light signalling”. This therefore means that cytokinins have a role in the regulation of light and photosynthesis [Figure 4].
The hormones also have a role in cell division of somatic plant cells, since cytokinin stimulates the transition of cells from the G2 phase to M phase of the cell cycle (in the presence of auxin). The hormone stimulates the removal of phosphotyrosine from p34cdc2 kinase, causing the activation capacity of kinase by Cdc25 phosphatase to decline and the cell to enter mitosis (John, 1996).
The presence of cytokinins delays senescence in leaves. The SAG 12 promoter is activated at the onset of senescence, leading to the synthesis of cytokinin (Gan and Amasino, 1995). Cytokinin causes the inhibition of senescence so the promoter is actively attenuated. This results in an autoregulatory loop, which is when part of a protein allows it to regulate its own genetic expression, preventing the overproduction of cytokinin. Therefore expression is confined to the tissues that had already initiated senescence (Gan and Amasino, 1995).
Cytokinins are also responsible for promoting cellular division and expansion in cotyledons (although not involving acidification), organogenesis (high cytokinin: auxin ratios favour the formation of shoots while low ratios favour formation of roots), bud activation and fruit and embryo development (Mauseth, 2008).
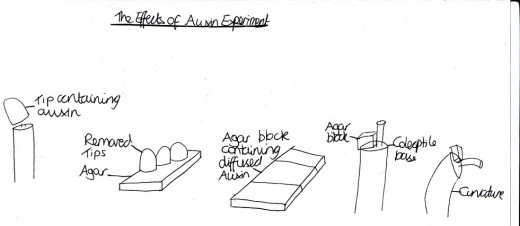
Figure 5 The Effects of Auxin Experiment
Click thumbnail to view full-size
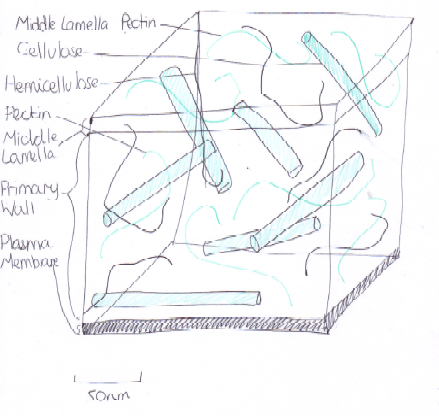
Figure 6 Components of the Cell Wall
Click thumbnail to view full-size
Auxin and its Practical Uses
Auxin was the first plant hormone to be discovered in 1926, because of its positive involvement with phototropism in oat seedlings (Mauseth, 2008) [Table 1]. Although it is now known that auxins are compounds with an aromatic ring and a carboxylic acid group that coordinate development such as cell elongation in stems and the other physiological activates of indoleacectic acid (IAA) at all levels (whether cellular, organs, or the entire organism) in plants, it was not possible to identify the chemical makeup at the time of development, since the concentrations were too small for human research (Mauseth, 2008). Instead, experiments with auxin were completed, for example Went (1926) did an Avena coleoptile test. This particular experiment consisted of: removing the tip and placing it on agar for 1-4 for the auxin to diffuse out, the agar block containing the diffused chemical was then placed on one side of the coleoptile base and after 90-120 minutes, the angle of curvature is measured [Figure 5]. Science has long since developed these methods and now it is known that there are four naturally occurring auxins (two of which are synthesised by plants): IAA, 4-chloroindole-3-acetic acid, phenylacetic acid and indole-3-butyric acid; and many Synthetic auxin analogs (Went, 1926).
A practical use of auxin is its ability to delay senescence (the ageing process). When the environmental stimuli are adequate, it causes a decrease in IAA production, meaning that some signal stimulates the cells in the abscission zone to produce ethylene (Gan and Amasino, 1995). This causes the cells to expand, suberise, and produce cellulase and pectinase enzymes, which digest the middle lamella [Figure 6]. Thus the cells separate and abscission occurs.
Auxins are also responsible for the process of apical dominance. This is when auxin is produced (for example) in the apical bud which requires growth, and is transported to the lateral bud growth, while interacting with other hormones e.g. cytokinins, where it suppresses development to stop the competition of light and nutrients between buds on the same plant. The effects of these processes can be seen when an apical tip is removed and lateral buds and shoots begin to form. The processes are manipulated by horticulturists, when they prune plants to get the required shape.
Auxins, in particular IAA cause fruit development and maturation from the seeds. Achenes are dry fruit that contain a seed that is attached to the pericarp (wall), for example strawberries (where the strawberry fruit is the receptacle). When the achenesare removed, no fruit growth occurs unless an auxin solution is added. This process is taken advantage of by humans, in the process of increasing the size of seedless grapes (Mauseth, 2008).
Other responses to auxin include: cell elongation, formation of roots in cuttings, tropisms, vascular tissue differentiation (e.g. vascular cambium is stimulated because of the flow of IAA from the leaves) and the development of adventitious roots (Mauseth, 2008).
References
Drew, M. (1985), ‘Larger Adenylate Energy Charge and ATP/ADP Ratios in Aerenchymatous Roots of Zea Mays in Anaerobic Media as a Consequence of Improved Internal Oxygen Transport’, Planta, 165: 51–58.
Fankhauser, C. (2002), ‘Light Perception in Plants: Cytokinins and Red Light Join Forces to Keep Phytochrome B Active’, Trends Plant Sci. 2002 Apr;7(4):143-5, Available at http://www.ncbi.nlm.nih.gov/pubmed/11950603 (Accessed 06/02/13).
Feher, J. (2012), Quantitative Human Physiology: An Introduction, Academic Press, pages 761-2.
Gan, S. and Amasino, R. (1995), ‘Inhibition of Leaf Senescence by Autoregulated Production of Cytokinin’, Science, 22;270, Available at http://www.ncbi.nlm.nih.gov/pubmed/8592746/ (Accessed 06/02/13).
Jackson and Campbell (2006), ‘Waterlogging and Petiole Epinasty in Tomato: The Role of Ethylene and Low Oxygen’, New Phytologist, Volume 76 (issue 1), pages 21-29.
John, P. (1996), ‘The Plant Cell Cycle: Conserved and Unique Features in Mitotic Control’, Progress in Cell Cycle Research, Volume 2, 59-72.
Kush, A. et al. (1990), ‘Lacti-cifer-Specific Gene Expression in Hevea brasiliensis (Rubber Tree)’, Proceeding of the Natural Academy of Sciences USA 87, 1787-1790.
Mauseth, J. (2008), Botany: an introduction to plant biology, 4th edition, Jones and Bartlett.
Pujade-Renaud, V. et al. (1994), ‘Ethylene-Induced Increase in Glutamine Synthetase Activity and mRNA Levels in Hevea brasiliensis Latex Cells’. Plant Physiol.,105:127–132.
Schnell, R. and Priyadarshan, P. (2012), Genomics of Tree Crops, Springer (2012 edition), pages 279-281.
Shimamura, S. et al. (2010), ‘Stem Hypertrophic Lenticels and Secondary Aerenchyma Enable Oxygen Transport to Roots of Soybean in Fooded Soil’, Annals of Botany, 106: 277–284.
TNAU: TamilNadu (2008), Agricultural University, Plant Growth Regulators, Available at http://agritech.tnau.ac.in/agriculture/agri_pgr_introduction.html (Accessed: 03/02/13).