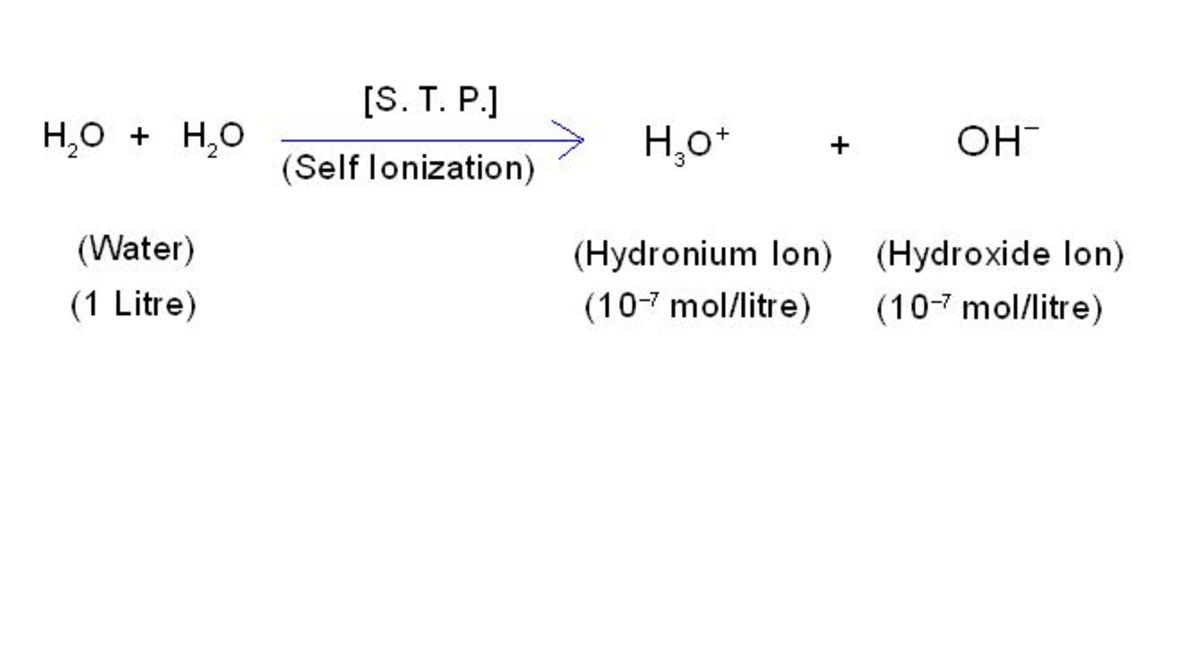
Types Of Reactions
Reactions can be of many types, ranging from allergic reactions to highly devastating nuclear reactions. The reactions that appear in Physics or Chemistry are entirely different from the Biological reactions. Biological reaction are associated with the body and are related to the formation of bacteria which trigger negative symptoms on it. One of the most common biological reaction, as we all know, is the allergic reaction which is quite a nuisance at times.
Here the topic in hand is not related to biology, rather the topic will be concerned only about the Chemical reactions. Generally Chemistry is all about reactions, equations and symbols, but there are also many sub-branches of Chemistry that deal with these reactions in a more mathematical manner.

Every reaction has its in own Kinetics which mean 'energy changes'. Some reaction have extremely high energy change which can be either endothermic or exothermic, these things are significant in the study of chemical reactions. As Chemistry is required in almost every industry and the reaction in the making of a product are a day to day procedure, a detail analysis of every reaction must be known to assist us in proper manufacturing of stuff.
Some reaction can be highly inflammmable or in other words devastating. For example the combustion of petrol or methane is a highly exothermic reaction, releasing a lot of heat.

Difference Between Chemical and Nuclear Reactions
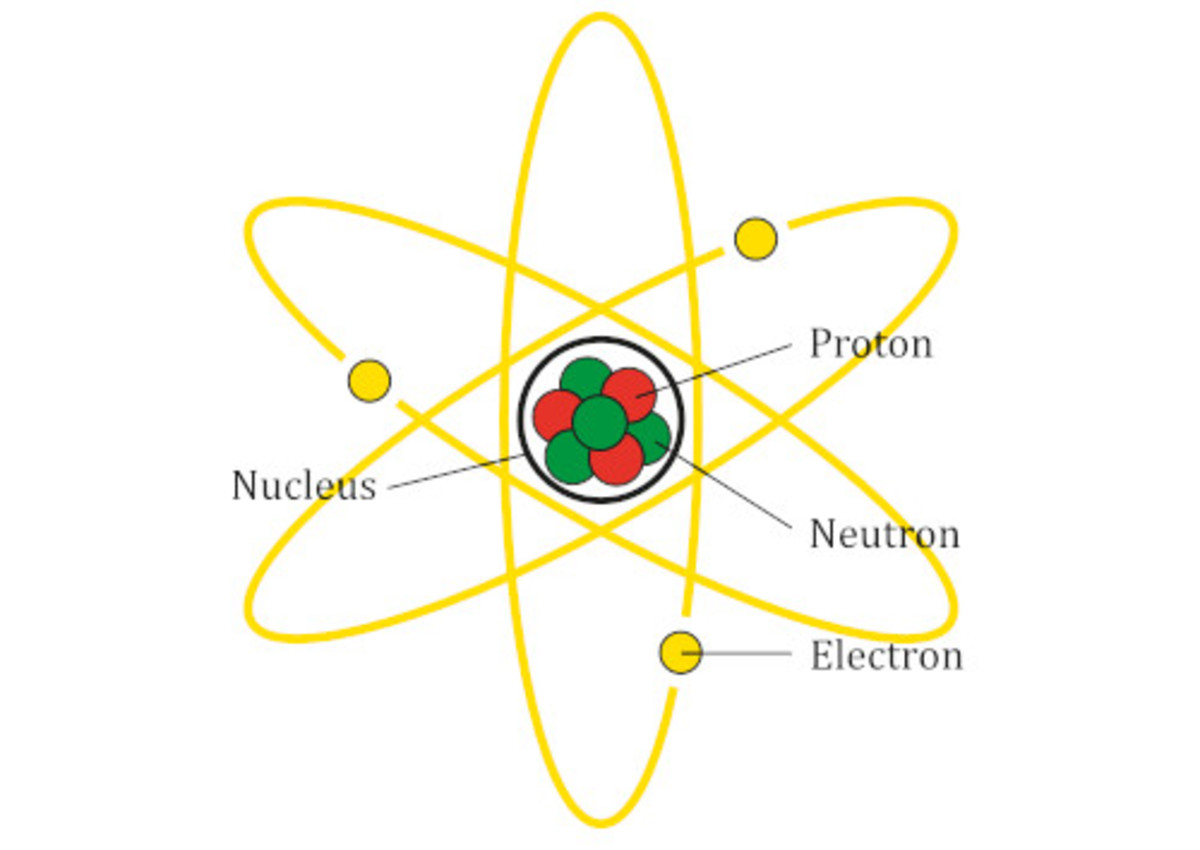
Before I head on to discuss the types of Chemical reactions let me first distinguish between a Nuclear reaction and a Chemical reaction. Often I have seen that some younger fellows are unaware of the fact that both of these are two different things. This unawareness may be due to the lack of conceptual understanding of the structure of atom.
An atom is composed of a nucleus at the center having protons and neutrons packed in it. While the third particle, electron, circles around the nucleus. All the Chemical reactions are associated with the transfer, sharing, loss and gain of electrons. They have nothing to do with the nucleus.
On the other hand, the nuclear reaction are entirely associated with the nucleus of the atom and have nothing to do with electrons. The nuclear reactions are actually associated with the decomposition of the nucleus which changes it to another atom due to the loss of protons and neutrons.
In general a chemical reaction has a very low energy change associated with it, whereas a nuclear reaction has a very high energy change.
Types Of Chemical Reaction
Chemical reactions can be classified in variety a ways, different authors use a different approach of classifying chemical reactions. This is due to the fact that there are a number of changes that occur in the atoms and molecules when they are reacted. Here I will use the most common approach of classifying chemical reactions. These are quite general reaction and all the other classes can have the same examples.

1. Addition,Synthesis or Combination Reaction
As the heading implies the reaction has several names. This is a simple type of reaction and it is the direct opposite to the decomposition reaction, which I will discuss shortly. In this reaction two or more different elements/compounds combine to form a new substance which is more complex than the reacting ones.
Example:
A simple reaction is the formation of NaCl.
2Na + Cl2---------------------> 2NaCl

2.Combustion Reaction
It is the second type of reaction and occurs when anything burns in the presence of oxygen. As we all know that oxygen supports combustion. So it can easily burn a substance by providing a small energy. These reactions are more common with organic material.
Example:
A day to day combustion reaction is the burning of methane.
CH4 + 2O2 ---------------------> CO2 + 2H2O

3.Decomposition Reaction
This is the exact opposite of the the addition reaction. In this reaction a compound breaks down into two or more simpler substance which may be elements or compounds. Energy is required to produce these reactions making them endothermic.
Example:
Decomposition of Calcium Carbonate is type of decomposition reaction.
CaCO3 -------------------> CaO + CO2

4.Single Diplacement Reaction
In this reaction one element displaces another element from a compound. The result is that a compound gets its atom replaced, popping out the atom it was combined with earlier. The elements which replaces another atom should be more reactive otherwise the reaction may not occur.
Example:
Displacement of Hydrogen from HCl.
2Na + 2HCl -------------> 2NaCl + H2

5. Double Displacement Reaction
This is the last type of reaction and the more complex one. In this reaction two different compounds exchange atoms between themselves, producing different products. Usually with this reaction a precipitate or undissolved substance is formed. These reactions are very common with acids and bases.
Example:
A common example is the formation of Silver Chloride.
NaCl + AgNO3----------------------> NaNO3 + AgCl