What are Elements?
The Basics
Everything is composed of basic substances called elements. An element is a substance that cannot be broken down to simpler substances with different properties by ordinary chemical means. There are 92 naturally occurring elements that serve as the building blocks of matter. Six elements-- carbon, hydrogen, nitrogen, oxygen, phosphorus, and sulfur-- make up 95% of the body weight of all organisms. The acronym CHNOPS will help you remember these six elements.

What are atoms?
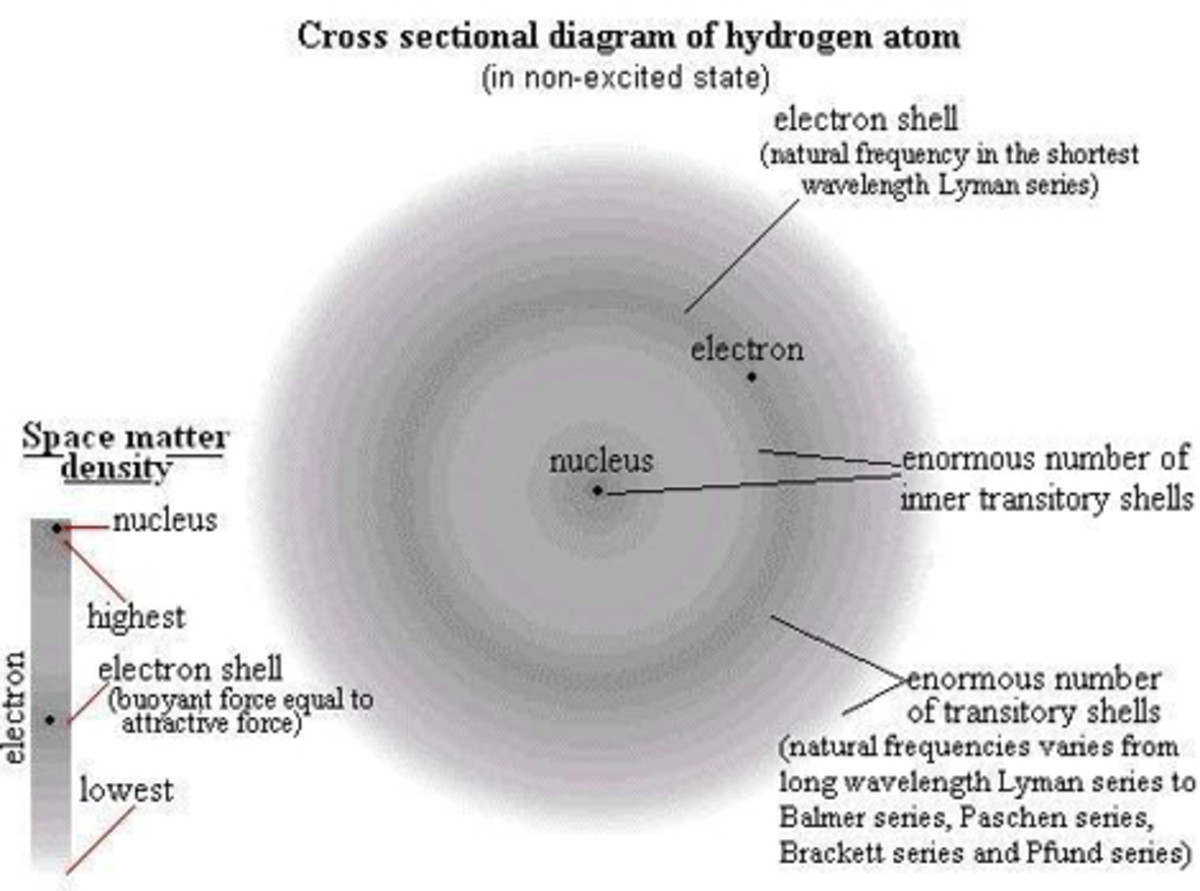
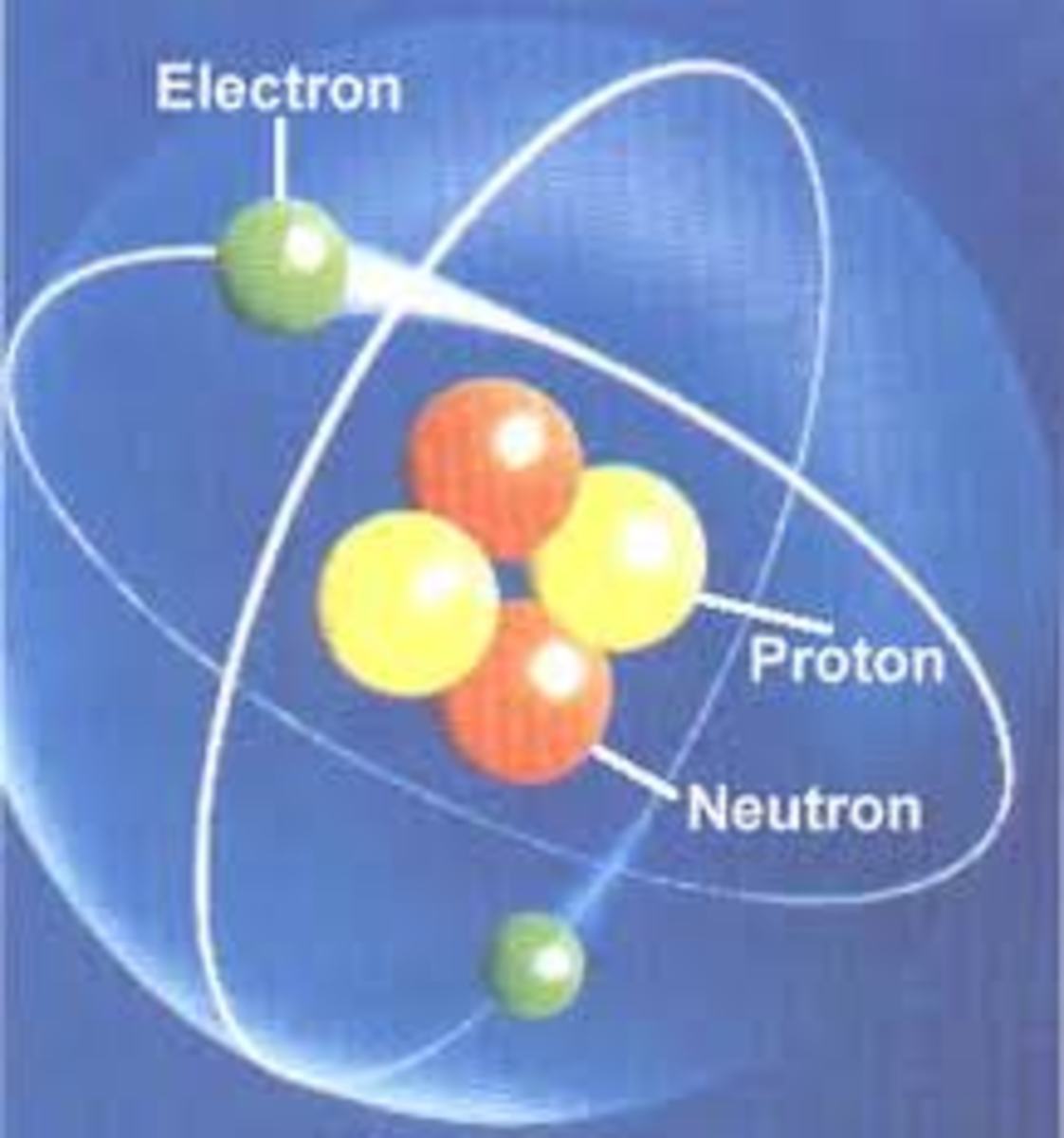
Elements consist of tiny particles called atoms. An atom is the smallest part of an element that displays the properties of the element. A number of subatomic particles make up atoms. The three you are probably familiar with: protons (which are positive) , neutrons (which are neutral), and electrons (which are negative). Protons and neutrons are found within the nucleus of an atom. Electrons move about the nucleus within the electron shell.
Atoms have an atomic number, symbol, and mass number. Protons and neutrons in the nucleus determine the mass number of an atom. The atomic number indicates the number of protons in the nucleus of an atom.
What is the periodic table?
The periodic table was devised once a number of elements were discovered. Chemists realized there were recurring chemical and physical characteristics in the elements. The periodic table arranges elements by their atomic numbers and according to common characteristics.
In Summary
Everything is composed of elements. Living things are made mostly of six elements known as CHNOPS. Elements are made of subatomic particles called atoms. Elements are arranged on the periodic table by their atomic numbers and according to common characteristics.