What is Oxygen?

The Mysterious Oxygen
When taking in a lung full of air, many gases enter the lungs. These gases include; Nitrogen, Oxygen, Carbon Dioxide, and others. But, what the lungs really need, is the Oxygen in the air. Why is this? Because we, as human being, need oxygen to prosper. Those tiny O2 atoms give life to our cells, which enables our cells to work and us to live. What is Oxygen though? Oxygen is the third most abundant element on our planet. This element has many properties, an interesting history, and a broad number of uses.
Oxygen's Properties
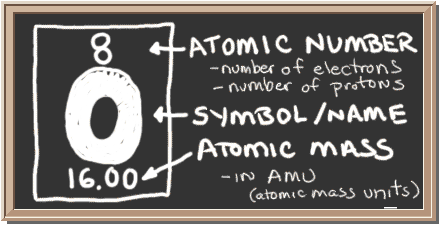
Oxygen has many characteristics, it is the element with the atomic number 8, a mass number of 16, and oxygen is represented as an O on the periodic table. Oxygen has 8 protons, 8 neutrons, and 8 electrons. Oxygen belongs to the chalcogen group on the periodic table and is a highly reactive nonmetallic gas.
In most situations, oxygen is seen as O2, two oxygen atoms bounds together to form dioxygen. Oxygen has a density of .001429g/cm3, and makes up nearly 21% of Earth’s atmosphere. It has a melting point of -218.79c and a boiling point of -182.95c.
Oxygen has three stable isotopes; 16 O, being a primary isotope, is the most abundant, while 17O and 18O are both secondary isotopes.
Oxygen also has fourteen radioisotopes, with the most stable being 15O. Oxygen is a highly reactive element and is capable of combining with most other elements. It is required by most living organisms and for most forms of combustion.
Oxygen is also a component of thousands of organic compounds. While oxygen is most commonly a gas, it can be in liquid or crystal form.
Oxygen
Oxygen's History
Oxygen was discovered by two different men independently.
Carl Wilhelm Scheele discovered oxygenin 1773, in Uppsala, and called it “fire air” because it was the only known supporter of combustion at the time.
Joseph Priestly discovered it in 1774, in Wiltshire, and called it “dephlogisticated air.” While oxygen had been produced by other scientist before 1773, these two men were the first to recognize it as a distinct element, but credit is usually given to Priestly because he published his findings first.
In 1777 the name oxygen was created by Antoine Lavoisier who incorrectly believed that oxygen was necessary to form all acids.
The nameoriginates from the Greek words gennan meaning 'generate' and oxus meaning 'acid' - so named because it was believed that all acids contained oxygen.

What Uses Oxygen Has
There are many uses for oxygen. The earliest being life. Humans and most other organisms need oxygen to survive. O2is essential for cellular respiration in all aerobic organisms. Oxygen is used in mitochondria to help generate ATP during oxidative phosphorylation, because of this oxygen is often used in medicine.
Oxygen therapy is used to treat emphysema, pneumonia, some heart disorders, and any disease that impairs the body's ability to take up and use gaseous oxygen.Hyperbaric (high-pressure) medicine uses special oxygen chambers to increase the partial pressure of O2around the patient and, when needed, the medical staff.
Carbon monoxide poisoning, gas gangrene, and decompression sickness (the bends) are sometimes treated using these devices. Oxygen is also used in industries. By injecting O2 in to molten iron, it will remove sulfur and carbon and create an environment hot enough to make steel.
Oxygen is also used to oxidize rocket fuel and to treat water. Scientifically oxygen is used to measure the ratio of 18O and16O in the shells and skeletons of marine organisms to determine what the climate was like millions of years ago. In order to have a fire there has to be combustible material and a ready supply of oxygen. One of the greatest uses of oxygen is that it is used to form water. H2O is very important to life. We need it, plants need it, and other organisms need it.
Fin
Oxygen is a very important element in our lives. This invisible tiny element has an abundance of characteristics, an interesting history, and many uses. Our atmosphere is made up of 21% of oxygen that we need to live. Oxygen was discovered by two scientists, and it’s needed so we can live. All of this is caused by oxygen. We need oxygen to have life, much like how fire needs oxygen to burn.
“Among the notable things about fire is that it also requires oxygen to burn - exactly like its enemy, life. Thereby are life and flames so often compared”(Otto Weininger).