Rota Virus Vaccination Effects
What the Rota Virus Is
Rota virus is the most common cause of diarrhea in infants and toddlers. It is not related to the flu but is often referred to as the stomach flu and may include fever and vomiting. The rota virus infects the intestinal lining, usually lasts from three to eight days and is the second most common illness in the U.S. Adequate rest and increased fluid intake is usually all that is needed to fully recover.
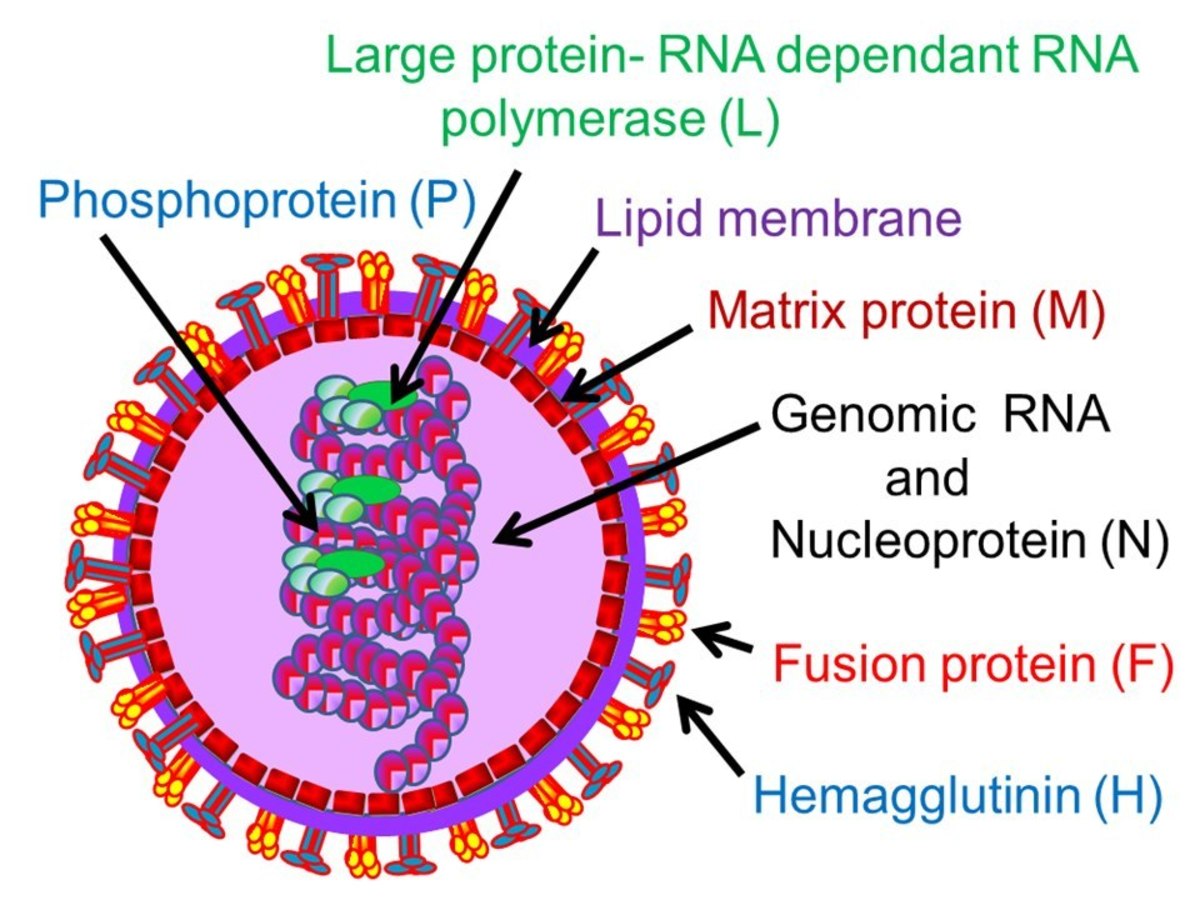
Rota is Latin for wheel to indicate the microscopic appearance of the virus. It was discovered in 1973 and is said to spread through contaminated food or water and by contact with an infected person. Infection from the virus mainly becomes a concern for those in developing countries who live without sanitary water and living conditions.
The majority of symptoms occur in children under two and immunity against rota virus strengthens with every episode of having caught it throughout life. It is a childhood illness that is easily managed with rest, hydration and adding small amounts of salt and sugar to the drinking water to boost electrolytes if needed. Most adults are no longer susceptible to rota virus and are rarely affected by it.
Potential Complications from the Rota Virus Vaccination
Human rota virus was first grown in monkey kidney cell cultures in 1981. A vaccine went to market in 1998 by Wyeth called Rota Shield. Vaccination was so adverse it caused severe bowel obstruction in the small intestine in one of every 12,000 infants. Since many people, whether doctor, patient or parent, do not submit adverse reports, it is likely the rate of side effects was much higher. Wyeth withdrew the vaccine from the market after only one year.
RotaTeq made by Merck & Co., was approved in 2006 as another vaccine for rota virus. They claimed their trials showed no relationship to the risk of intestinal blockage, however in the first year it had four more events reported than Rota Shield. Although Wyeth recalled their vaccine, Merck continued to sell the product to parents who are probably not made aware of the potential harm it may inflict upon their baby. The FDA issued a warning that bowel obstruction is a serious and potentially life-threatening condition associated with the RotaTeq vaccine.
Dose Schedule
Vaccinations for rota virus are administered in three doses in the first year of life only. It consists of an oral serum with the first dose at or about 14 weeks of age and the last dose at six months with the maximum age of eight months. Determining the sole cause of symptoms is further complicated by other vaccines on the schedule administered on or about the same time.
Cow, Monkey and Pig DNA in Rotavirus Vaccine
In a report published by the National Vaccine Information Center (NVIC), it states Merck’s RotaTeq is genetically engineered containing five human-cow re-assortment strains of rotavirus created at the Children’s Hospital of Pennsylvania. There the strains of rotavirus that give cows diarrhea were combined with rotavirus strains that cause diarrhea in humans.
The mix was then sent to Merck where African Green Monkey kidney cell cultures were used to produce master seed stock from which all RotaTeq is made. Fetus cow serum and porcine (pig) trypsin, which breaks proteins into smaller peptides, was used to make the seed stock. Small amounts of cow serum and monkey viral DNA remain in the finished product, as well as pig viruses.
Merck's RotaTeq package insert includes the following as potential adverse reactions:
Vomiting, *diarrhea, irritability, otitis media (ear infection), naso-pharyngitis (pus and bacteria in the nose and at the back of the throat), broncho spasm, bronchiolitis, gastric upset, *pneumonia, fever, urinary tract infection, seizures, intussusception (bowel obstruction), *death or SIDS.
*The purpose of the vaccine is to reduce the potential for infant diarrhea, which can lead to dehydration which can lead to death, yet the vaccine itself may cause diarrhea and death. The vaccine may also cause pneumonia in children which has shown in studies to cause death in some.
Rotavirus Vaccination Symptoms
Side effects to report immediately to the child's doctor, even several weeks past the last dose of rota virus vaccine, include: Stomach pain, vomiting, diarrhea, blood in stool, a change in bowel movements, or difficulty breathing.
Submitting Adverse Events to the Reporting System VAERS
A few formal studies are all that is required to gain approval for a vaccine and release it to the public. The study trials for vaccine safety and effectiveness are conducted by the drug manufacturers themselves. The true testing of most vaccines occur after they have been put on the market. People more than likely presume it is the physicians duty to contact the vaccine adverse event reporting system (VAERS). Although there are many that do, there are also many who choose not to.
Some physicians are concerned about the potential legal liability from side effects after vaccinations. Even though the National Vaccine Injury Compensation Program (established by federal statute in 1986) has resulted in very few suits filed against physicians for vaccination-related injuries.
Rotavirus Vaccines Contaminated with Two Pig Viruses:
In March, 2010 the FDA recommended suspending the use of Rotarix, made by GS&K, after it was found to have a pig virus, pcv1 DNA. Since the pig virus wasn't found in Merck's vaccine during the preliminary tests, the FDA advised physicians to change from Rotarix to RotaTeq.
It was announced on May 5, two days before a meeting, that a more dangerous pcv2 pig virus DNA had been found in Merck's RotaTeq vaccine. Studies were being conducted by an independent group of researchers who discovered the virus. It is called porcine circovirus and causes a severe wasting disease and death in piglets. No one from Merck & Co. attended the meeting, however GS&K attended and pledged to remove the milder pcv1 pig virus.
PCV2 infects pigs worldwide causing a potentially fatal wasting syndrome which causes a progressive loss of body condition; lung damage, enlarged lymph nodes, and sometimes diarrhea, pale skin, liver dysfunction and death. The FDA allowed both rota virus vaccines to remain on the market.
The Clinical and Vaccine Immunology journal stated that vaccinating pigs with pcv2 actually caused them to catch the deadly infection rather than establish immunity. A study published in the journal, Nutrition, found that nasal inoculation of pregnant sows with pcv2 caused abortion and reproductive failure.
A study published in 2011 in the journal, Vaccine, proved that a human cell line can be infected with pcv1. One in 2005 found that human white blood cells are susceptible to pcv1 infection, and another study from 2004 in the journal, Xenotransplantation, showed human cells expressing and replicating the genes from both virus types.
Human Effects from Animal DNA
It is well known that DNA from animal viruses can infect human cells and change human DNA to cause disease in humans. RotaTeq still contained the more lethal pig virus, however since they didn't attend the meeting, it wasn't addressed. The FDA's justification to allow millions of infants to be exposed to pcv1 and pcv2 DNA, and possibly live viruses transmitted through vaccination, is under the mistaken notion that the viruses can not infect humans.
Cross-species transmission of animal viruses are well known to be more harmful than viruses that have evolved within a host. A virus which may cause no harm to the animal within which it is naturally found, monkeys or pigs for example, may be highly lethal to another species such as humans.
Choosing whether or not to vaccinate a baby against rota virus is an important decision best left up to the parents. Obtaining information from reputable sources from whom do not have a vested financial interest helps us make decisions easier by being better informed.
Lizolivia
Resources:
vran.org
activistpost.com
nvic.org