Extensive and detailed clinical highlights on Diabetes Mellitus
all about Diabetes






INTRODUCTION.....
Hello guys! Welcome to the first treatment experience of the endocrinology department of my internet Hospital. Before we kick off, I would like to encourage you that this is a very interesting, detailed, extensive and 'complete' hub about the clinical and medical evaluation of Diabetes Mellitus. Before you read on, I will urge you to take a look at the link below for a better preamble of what you are to expect here in this hub. You could open it in another tag, so that you can easily come back to this well-informative page about Diabetes. Here is the link
Diabetes Mellitus in simple plain language
When you are done, you can come back here to continue. Here are the highlights of what you are to expect:
1. Introduction to Diabetes Mellitus (Physiology of Insulin)
2. Action of Insulin
3. Type 1 Diabetes
4. Type 2 Diabetes
5. Pathogenesis and clinical difference between Type 1 and Type 2
6. Etiologic classification of Diabetes Mellitus
7. Stages of Diabetes Mellitus development
8. Degrees of severity of Diabetes Mellitus
9. Physical examination of a patient with DIabetes Mellitus
10. Atherosclerosis in Diabetes Mellitus
11. Allergic reactions
12. Diabetic Blisters
13. Diagnosis of Diabetes Mellitus
14. Long term complications of Diabetic Mellitus which includes sexual problems in men (Erectile dysfunction and retrogade ejaculation) and sexual problems in women (decreased vaginal Lubrication and decreased or Absent sexual responses)
For those of you doing some research of compilation of resources, you can go to the bottom or end of this hub and click on print to download this hub is a very fine PDF format. This can be with you for as long as you want. Read and share! That is why I am here, its a community service which is done through the internet. I hope you will enjoy and appreciate it.
INSULIN PHYSIOLOGY
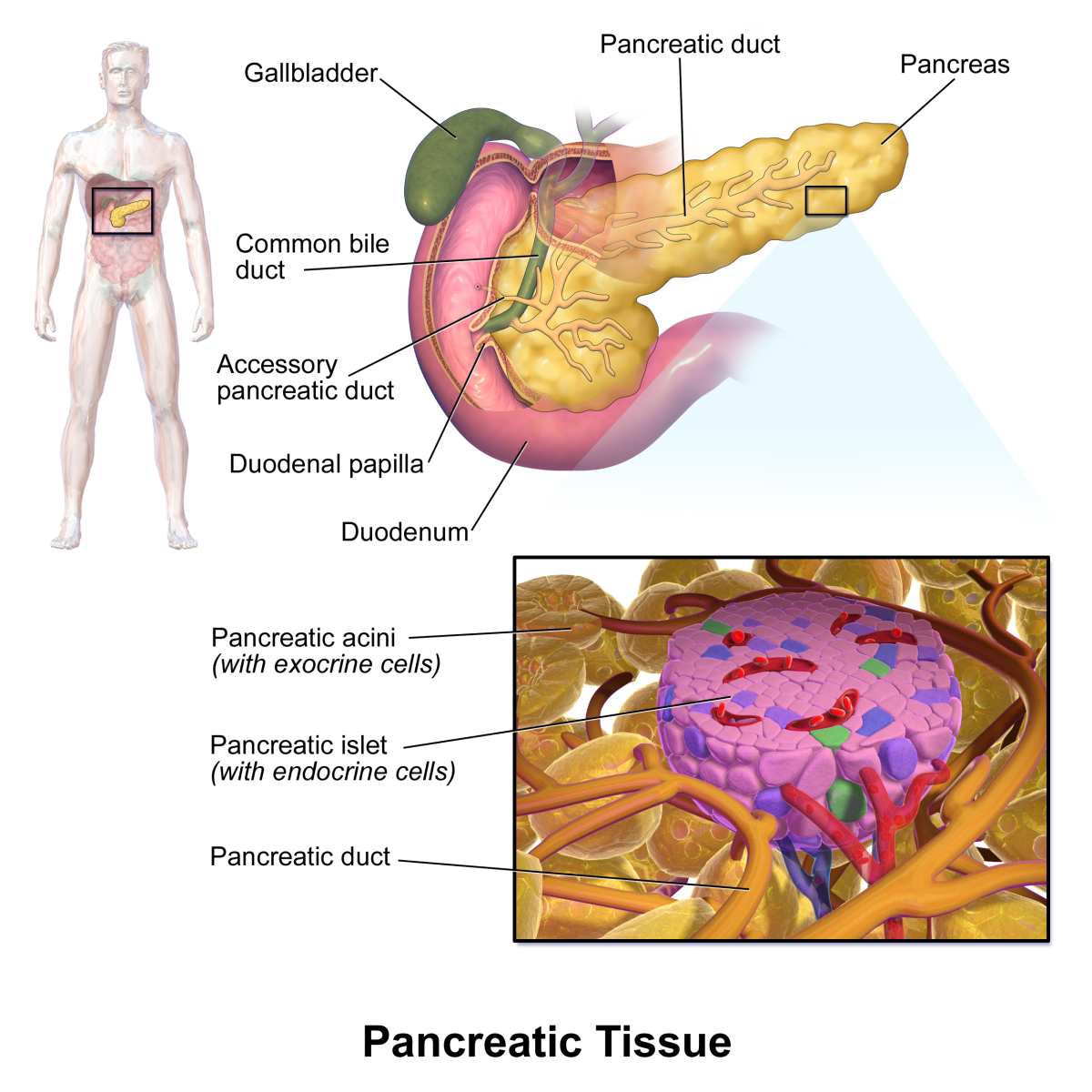
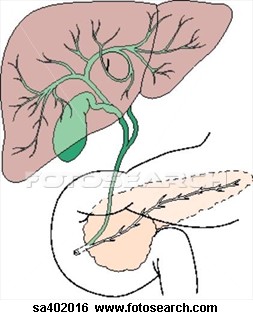
The pancreas is an elongated organ nestled next to the first part of the small intestine. The endocrine pancreas refers to those cells within the pancreas that synthesize and secrete hormones.
The endocrine portion of the pancreas takes the form of many small clusters of cells called islets of Langerhans or, more simply, islets. Humans have roughly one million islets. In standard histological sections of the pancreas, islets are seen as relatively pale-staining groups of cells embedded in a sea of darker-staining exocrine tissue.
Pancreatic islets house three major cell types, each of which produces a different endocrine product:
· Alpha cells (A cells) secrete the hormone glucagon.
· Beta cells (B cells) produce insulin and are the most abundant of the islet cells.
Delta cells (D cells) secrete the hormone somatostatin, which is also produced by a number of other endocrine cells in the body.
Interestingly, the different cell types within an islet are not randomly distributed - beta cells occupy the central portion of the islet and are surrounded by a "rind" of alpha and delta cells. Aside from the insulin, glucagon and somatostatin, a number of other "minor" hormones have been identified as products of pancreatic islets cells.
Islets are richly vascularized, allowing their secreted hormones ready access to the circulation. Although islets comprise only 1-2% of the mass of the pancreas, they receive about 10 to 15% of the pancreatic blood flow. Additionally, they are innervated by parasympathetic and sympathetic neurons, and nervous signals clearly modulate secretion of insulin and glucagon.
The term Diabetes Mellitus refers to the excretion of large quantities of sweet urine. Diabetes is an old word for siphon and means “dieresis”, mellitus means “sweet”. The clinical syndrome known as DM comprises a wide variety of symptoms, physical findings and laboratory abnormalities, in which multiple etiologic factors are involved, the pathophysiology is partly understood and treatment is unsatisfactory. The hallmark of DM is hyperglycemia.
Diabetes Mellitus (DM) – is endocrine – metabolic disease, which develops due to absolute or relative insulin insufficiency and characterized by chronic hyperglycemia, changes of different systems and organs of patient.
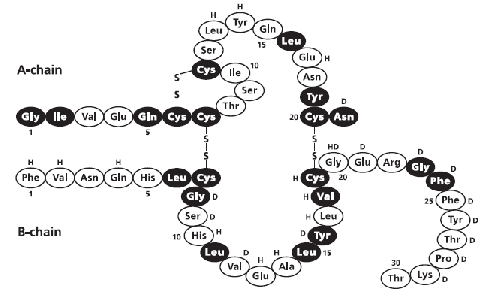
Insulin is a rather small protein, with a molecular weight of about 6000 Daltons. It is composed of two chains held together by disulfide bonds.
Insulin is secreted in primarily in response to elevated blood concentrations of glucose. This makes sense because insulin is "in charge" of facilitating glucose entry into cells. Some neural stimuli (e.g. sight and taste of food) and increased blood concentrations of other fuel molecules, including amino acids and fatty acids, also promote insulin secretion.
Our understanding of the mechanisms behind insulin secretion remain somewhat fragmental. Nonetheless, certain features of this process have been clearly and repeatedly demonstrated, yielding the following model:
· Glucose is transported into the B cell by facilitated diffusion through a glucose transporter; elevated concentrations of glucose in extracellular fluid lead to elevated concentrations of glucose within the B cell.
· Elevated concentrations of glucose within the B cell ultimately leads to membrane depolarization and an influx of extracellular calcium. The resulting increase in intracellular calcium is thought to be one of the primary triggers for exocytosis of insulin-containing secretory granules. The mechanisms by which elevated glucose levels within the B cell cause depolarization is not clearly established, but seems to result from metabolism of glucose and other fuel molecules within the cell, perhaps sensed as an alteration of ATP:ADP ratio and transduced into alterations in membrane conductance.
· Increased levels of glucose within B cells also appears to activate calcium-independent pathways that participate in insulin secretion.
Stimulation of insulin release is readily observed in whole animals or people. The normal fasting blood glucose concentration in humans and most mammals is 80 to 90 mg per 100 ml, associated with very low levels of insulin secretion.
Immediately after the increasing the level of glycemia begins, plasma insulin levels increase dramatically. This initial increase is due to secretion of preformed insulin, which is soon significantly depleted. The secondary rise in insulin reflects the considerable amount of newly synthesized insulin that is released immediately. Clearly, elevated glucose not only simulates insulin secretion, but also transcription of the insulin gene and translation of its mRNA.
Physiologic effects of insulin
Stand on a streetcorner and ask people if they know what insulin is, and many will reply, "Doesn't it have something to do with blood sugar?" Indeed, that is correct, but such a response is a bit like saying "Michael Jackson? Wasn't he some kind of a musician?"
Insulin is a key player in the control of intermediary metabolism. It has profound effects on both carbohydrate and lipid metabolism, and significant influences on protein and mineral metabolism. Consequently, derangements in insulin signalling have widespread and devastating effects on many organs and tissues.
THE ACTION OF INSUIN
Insulin is an anabolic hormone (promotes the synthesis of carbohydrates, proteins, lipids and nucleic acids).
The most important target organs for insulin action are:
- liver
- muscles
- adipocytes.
The brain and blood cells are unresponsive to insulin.
Insulin and Carbohydrate Metabolism
Glucose is liberated from dietary carbohydrate such as starch or sucrose by hydrolysis within the small intestine, and is then absorbed into the blood. Elevated concentrations of glucose in blood stimulate release of insulin, and insulin acts on cells thoughout the body to stimulate uptake, utilization and storage of glucose. The effects of insulin on glucose metabolism vary depending on the target tissue.
The effects of insulin on carbohydrate metabolism include:
1. Insulin facilitates entry of glucose into muscle, adipose and several other tissues.
2. Insulin stimulates the liver to store glucose in the form of glycogen .
3. Insulin inhibits glucose formation – from glycogen (glycogenolysis) and – from amino-acid precursors (glyconeogenesis).
As a result - well-known effect of insulin is to decrease the concentration of glucose in blood, which should make sense considering the mechanisms described above. Another important consideration is that, as blood glucose concentrations fall, insulin secretion ceases. In the absence of insulin, a bulk of the cells in the body become unable to take up glucose, and begin a switch to using alternative fuels like fatty acids for energy. Neurons, however, require a constant supply of glucose, which in the short term, is provided from glycogen reserves.
In the absence of insulin, glycogen synthesis in the liver ceases and enzymes responsible for breakdown of glycogen become active. Glycogen breakdown is stimulated not only by the absence of insulin but by the presence of glucagon, which is secreted when blood glucose levels fall below the normal range.
Insulin and Protein Metabolism:
1. Insulin transfers of amino acids across plasma membranes.
2. Insulin stimulates of protein synthesis.
3. Insulin inhibites of proteolysis.
Insulin and Lipid Metabolism
The metabolic pathways for utilization of fats and carbohydrates are deeply and intricately intertwined. Considering insulin's profound effects on carbohydrate metabolism, it stands to reason that insulin also has important effects on lipid metabolism. Important effects of insulin on lipid metabolism include the following:
1. Insulin promotes synthesis of fatty acids in the liver.
2. Insulin inhibits breakdown of fat in adipose tissue (lipolisis) by inhibiting the intracellular lipase that hydrolyzes triglycerides to release fatty acids.
From a whole body perspective, insulin has a fat-sparing effect. Not only does it drive most cells to preferentially oxidize carbohydrates instead of fatty acids for energy, insulin indirectly stimulates accumulation of fat is adipose tissue.
Insulin and Nucleic acids Metabolism:
1. Insulin stimulates nucleic acid synthesis by stimulating the formation of adenosine triphosphate (ATP), DNA and RNF.
Other effects:
1. Insulin stimulates the intracellular flow of potassium, phosphate and magnesium in the heart.
2. Insulin inhibits inotropic and chronoropic action (unrelated to hypoglycemia).
The action of insulin can be decreased by:
- glucagons: stimulates glycogenolysis and glyconeogenesis;
- somatostatin: inhibits secretion of insulin and regulates glucose absorption from alimentary tract into blood;
- glucocorticoids: decrease of glucose utilization by tissues, stimulate glycogenolysis and glyconeogenesis, increase lipogenesis (in patients with insulinoresistancy);
- catecholamines (adrenaline): inhibits β-cells secretion, stimulates glycogenolysis and ACTH secretion;
- somatotropin: stimulates α-cells (which secret glucagon), increases activity of enzymes which destroy the insulin, stimulates glyconeogenesis, increases of glucose exit from the liver veins into blood, decreases of glucose utilization by tissues;
- ACTH: stimulates glucocorticoides secretion and β-cells secretion;
- thyroid hormones: increase glucose absorption into blood, stimulate glycogenolysis, inhibit fat formation from the carbohydrates.
Absolute insulin insufficiency means that pancreas produce insulin in very low quantities or doesn’t produce it at all (due to destruction of beta-cells by inflammative, autoimmune process or surgery).
Relative insulin insufficiency means that pancreas produces or can produce insulin but it doesn’t “work”. (The pathologic process can be on the next levels:
- beta cells: they can be not sensitive for the high level of glycemia;
- insulin: abnormal insulin, insulin antibodies, contrainsulin hormones, absence of enzyme, which activates proinsulin (into insulin));
- receptors (decreased receptor number or diminished binding of insulin).
The Pancreas and Insulin
Chronic Complications of Diabetes Mellitus
Classification of chronic (long-term) complications of DM.
I. Diabetic angiopathy:
1. Microangiopathy:
1) nephropathy;
2) retinopathy;
3) angiopathy of lower extremitas.
2. Macroangiopathy:
1) ischemic heart disease;
2) angiopathy of lower extremities.
II. Diabetic neuropathy:
1) central (encephalopathy);
2) peripheral;
3) visceral (dysfunction of inner organs).
The long-term degenerative changes in the blood, vessels, the heart, the kidneys, the nervous system, and the eyes as responsible for the most of the morbidity and mortality of DM. There is a causal relationship and the level of the metabolic control.
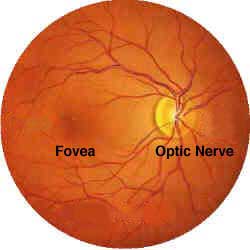
Diabetic retinopathy.
Background retinopathy (the initial retinal changes seen on the ophthalmoloscopic examination) does not significantly alter vision, but it can lead to processes that cause blindness (e.g., macular edema or proliferative retinopathy with retinal detachment or hemorrhage).
Evidence of retinopathy, rarely present at diagnosis in 1DM, is present in up to 20 % of type 2 DM patients at diagnosis. About 85 % of all diabetics eventually develop some degree of retinopathy.
Diabetic retinopathy is classified according to the changes seen at background during ophthalmoscopic examination with pupils dilated.
I. Nonproliferative or background retinopathy (it is usually the earliest sigh and consists of retinal microaneurysms, hard and soft exudates).
II. Maculopathy or preproliferative retinopathy (it is characterized by macular edema and/or hemorrhages).
III. Proliferative retinopathy (the hallmark of this complication is neovascularization, i.e., growth of new vessels in areas of hypoperfusion. Adhesion of the vessels to the vitreous leads to retinal detachment, vitreous hemorrhage and others. The prognosis is extremely poor. 5 years after recognition of this complication 50 % of the patients are blind).
The mechanisms involved in the development of retinopathy are not clearly known. Genetic predisposition, growth hormone, hypoxia, and metabolitic abnormalities particularly of lipids, have been implicated.
Diabetic nephropathy.
It is usually asymptomatic until end stage renal disease develops, but it can course the nephrotic syndrome prior to the development of uremia. Nephropathy develops in 30 to 50 % of type 1 DM patients and in small percentage of type 2 DM patients. Arteriolar hyalinosis, a deposition of hyaline material in the lumen of the afferent and efferent glomerular arterioles, is an almost pathognomic histologic lesion of DM.
Classificationof diabetic nephropathy by Mogensen.
I. Hyperfunction of kidneys. It is characterized by:
- increased renal blood circulation;
- increased glomerular filtration rate (GFR) (> 140 ml/min);
- hypertrophy of kidneys;
- normoalbuminuria (<30 mg/day).)
II. Stage of initial changes of kidney structure. It is characterized by:
- mesangial changes due to accumulation of immunoglobulins (IgG, IgM), complement and other nonimmunologic proteins (lipoproteins, fibrin);
- high GFR;
- normoalbuminuria.)
III. Initial nephropathy. (It is characterized by:
- microalbuminuria (30 to 300 mg/day);
- high or normal GFR;
- periods of blood hypertension.)
IV. Nephropathy or nephrotic stage. (It is characterized by:
- persistent proteinurea (>500 mg/day);
- normal or decreased GFR;
- persistent blood hypertension.)
V. Chronic renal failure or uremia.. It is characterized by:
Physical signs of chronic renal failure
- decreased GFR;
- blood hypertension;
- increased serum creatinine;
- signs of intoxication.
Diabetic angiopathy of lower extremities
Atherosclerosis of large vessels (macroangiopathy) leads to intermittent claudication, cold extremities and other symptoms which can be also find while arteriols and capillaries are affected (microangiopathy).
Classification of lower extremities’ angiopathy.
I. Nonclinic stage. (Changes could be find only during instrumental examination.)
II. Functional stage. (It is characterized by cold extremities, numbness, tingling, pain during physical examination.)
III. Organic stage. (It is characterized by trophyc changes: dry skin, hypo- or atrophy of muscles, ulcers, gangrene.)
Ischemic heart disease.
1. Cardiovascular changes tend to occur earlier in patients with DM when compared with individuals of the same age.
2. Frequency of myocardial infarction (MI) and mortality is higher in diabetics than that in nondiabetis og the same age.
3. The prognosis is even worse if ketoacidosis, or other complications of DM are present.
4. Diabetic patients have more complications of MI (arrhythmias, cardiogenic shock and others) than nondiabetic ones.
5. Often can observe atypical forms (without pain).
6. Male : female = 1 : 1 (nondiabetics = 10 : 1).
Diabetic neuropathy.
Classification of diabetic neuropathy.
I. Encephalopathy (central neyropathy) is characterized by decreased memory, headache, unadequate actions and others.
II. Peripheral polyneuropathy (radiculoneuropathy). There are three types of radiculoneuropathy:
- distal polyradiculoneuropathy (It is characterized by symmetrical sensory loss, pain at night and during the rest, hyporeflexia, decreased response touch, burning of heels and soles. The skin becomes atrophic, dry and cold, hair loss may be prominent. The decreased response to touch and pain predisposes to burns and ulcers of the legs and toes.);-
- truncal polyradiculoneuropathy (It is an asymmetric, and characterized by pain (which is worse at night), paresthesia and hyperesthesia; muscular weakness involves the muscles of the anterior thigh; reflexes are decreased; weight loss is common.);
- truncal monoradiculoneuropathy (It is usually involves thorasic nerves and the findings are limited to the sensory abnormalities in a radicular distribution.).
III. Visceral dysfunction:
When you want to lift your arm or take a step, your brain sends nerve signals to the appropriate muscles. Internal organs like the heart and bladder are also controlled by nerve signals, but you do not have the same kind of conscious control over them as you do over your arms and legs. The nerves that control your internal organs are called autonomic nerves, and they signal your body to digest food and circulate blood without your having to think about it. Your body's response to sexual stimuli is also involuntary, governed by autonomic nerve signals that increase blood flow to the genitals and cause smooth muscle tissue to relax. Damage to these autonomic nerves is what can hinder normal function.
1) gastrointestinal tract:
- esophageal neuropathy (It is characterized by segmental distribution with low or absent resting pressure in the low or absent resting pressure in the lower esophageal sphincter and by absence of peristalsis in the body of the esophagus.);
- diabetic gastroparesis (It leads to the irregular food absorption and is characterized by nausea, vomiting, early satiety, bloating and abdomen pain.);
- involvement of the bowel (It is characterized by diarrhea (mostly at nighttime, postural diarrhea), constipation, malabsorption and fecal incontinence;
2) cardiovascular system:
- orthostatic hypotension (It is characterized by dizziness, vertigo, faintness, and syncope upon assumption of the upright posture and is caused by failure of peripheral arteriolar constriction.);
- tachicardia (but it does not occur in response to hypotension because of sympathetic involvement).
3) urinary tract:
- Bladder dysfunction can have a profound effect on quality of life. Diabetes can damage the nerves that control bladder function. Men and women with diabetes commonly have bladder symptoms that may include a feeling of urinary urgency, frequency, getting up at night to urinate often, or leakage of urine (incontinence). These symptoms have been called overactive bladder. Less common but more severe bladder symptoms include difficulty urinating and complete failure to empty (retention). These symptoms are called a neurogenic bladder. Some evidence indicates that this problem occurs in both men and women with diabetes at earlier ages than in those without diabetes.
Neurogenic Bladder
In neurogenic bladder, damage to the nerves that go to your bladder can cause it to release urine when you do not intend to urinate, resulting in leakage. Or damage to nerves may prevent your bladder from releasing urine properly and it may be forced back into the kidneys, causing kidney damage or urinary tract infections.
Neurogenic bladder can be caused by diabetes or other diseases, accidents that damage the nerves, or infections.
Symptoms of neurogenic bladder include
- urinary tract infections
- loss of the urge to urinate when the bladder is full
- leakage of urine
- inability to empty the bladder
4) sexual disorders:
In Men
Erectile Dysfunction
Estimates of the prevalence of erectile dysfunction in men with diabetes range from 20 to 85 percent. Erectile dysfunction is a consistent inability to have an erection firm enough for sexual intercourse. The condition includes the total inability to have an erection, the inability to sustain an erection, or the occasional inability to have or sustain an erection. A recent study of a clinic population revealed that 5 percent of the men with erectile dysfunction also had undiagnosed diabetes.
Men who have diabetes are three times more likely to have erectile dysfunction as men who do not have diabetes. Among men with erectile dysfunction, those with diabetes are likely to have experienced the problem as much as 10 to 15 years earlier than men without diabetes.
In addition to diabetes, other major causes of erectile dysfunction include high blood pressure, kidney disease, alcoholism, and blood vessel disease. Erectile dysfunction may also occur because of the side effects of medications, psychological factors, smoking, and hormonal deficiencies.
If you experience erectile dysfunction, talking to your doctor about it is the first step in getting help. Your doctor may ask you about your medical history, the type and frequency of your sexual problems, your medications, your smoking and drinking habits, and other health conditions. A physical exam and laboratory tests may help pinpoint causes. Your blood glucose control and hormone levels will be checked. The doctor may also ask you whether you are depressed or have recently experienced upsetting changes in your life. In addition, you may be asked to do a test at home that checks for erections that occur while you sleep.
Retrograde Ejaculation
Retrograde ejaculation is a condition in which part or all of a man's semen goes into the bladder instead of out the penis during ejaculation. Retrograde ejaculation occurs when internal muscles, called sphincters, do not function normally. A sphincter automatically opens or closes a passage in the body. The semen mixes with urine in the bladder and leaves the body during urination, without harming the bladder. A man experiencing retrograde ejaculation may notice that little semen is discharged during ejaculation or may become aware of the condition if fertility problems arise. His urine may appear cloudy; analysis of a urine sample after ejaculation will reveal the presence of semen.
Poor blood glucose control and the resulting nerve damage are associated with retrograde ejaculation. Other causes include prostate surgery or some blood pressure medicines.
Sexual Problems in Women With Diabetes
Decreased Vaginal Lubrication
Nerve damage to cells that line the vagina can result in dryness, which in turn may lead to discomfort during sexual intercourse. Discomfort is likely to decrease sexual response or desire.
Decreased or Absent Sexual Response
Diabetes or other diseases, blood pressure medications, certain prescription and over-the-counter drugs, alcohol abuse, smoking, and psychological factors such as anxiety or depression can all cause sexual problems in women. Gynecologic infections or conditions relating to pregnancy or menopause can also contribute to decreased or absent sexual response.
As many as 35 percent of women with diabetes may experience decreased or absent sexual response. Decreased desire for sex, inability to become or remain aroused, lack of sensation, or inability to reach orgasm can result.
Symptoms include
· decreased or total lack of interest in sexual relations
· decreased or no sensation in the genital area
· constant or occasional inability to reach orgasm
· dryness in the vaginal area, leading to pain or discomfort during sexual relations
Neuropathic arthropathy (Charcot’s joints)
is characterized by painless swelling of the feet without edema or signs of infection. The foot becomes shorter and wider, eversion, external rotation, and flattening of the longitudinal arch. This arthropathy is associated with sensory involvelvement, particularly impairment of afferent pain proprioceptive impulses.
Diabetic foot.
Appearance of diabetic foot is caused by a combination of vascular insufficiency, neuropathy, and infection.
Diabetic foot is divided on:
- ischemic;
- neuropathy;
- mixed.
Treatment of long-term complications
The main principle: adequate metabolic control.
Diabetic retinopathy.
1) careful ophthalmologic examination (at least yearly) by ophthalmologist experienced with diabetes;
2) nonproliferative retinopathy:
- anabolic agents (nerabol 5 mg, nerabolil 1mg/week 1,5 – 2 month, retabolil 1ml/3 weeks 3 – 6 times);
- hypocholesterol agents (lipamid, lovostatin);
- antioxydative therapy (emoxipin, trental);
- vitamins B,A,E,PP;
- anticoagulants;
3) preproliferative or proliferative retinopathy: treatment by photocoagulation.
Diabetic nephropathy.
1) low-protein diet (less than 40 g of protein daily);
2) inhibitors of ACF (renitec);
3) hypotensive therapy;
4) hemodyalisis, kidney’s transplantation.
Diabetic angiopathy of lower extremities.
1) patient education in foot care; early detection of risk factors, ulcers, infections, calluses, exposed nails, diminished pulses, deformities;
2) anticoagulants;
3) preparations for improvement blood circulation.
Diabetic neuropathy.
1) Treatments for neuropathy include preparations of α-lipoid acid (Berlition, Espa – lipon, Tiogama),inhibitors of aldose reductase (sorbinil), multivitamins, phenytoin, carbamazepin (Tegretol),amitriptyline, nootropil, piracetam;
2) physiotherapy (inductotermia, magnitolazerotherapy and others).
3) Specific treatment for few types of visceral neuropathy:
- Treatments for erectile dysfunction caused by nerve damage vary widely and range from oral pills, a vacuum pump, pellets placed in the urethra, and shots directly into the penis, to surgery. All these methods have strengths and drawbacks. Psychotherapy to reduce anxiety or address other issues may be necessary. Surgery to implant a device to aid in erection or to repair arteries is another option.
- Retrograde ejaculation caused by diabetes or surgery may be improved with a medication that improves the muscle tone of the bladder neck. A urologist experienced in infertility treatments may assist with techniques to promote fertility, such as collecting sperm from the urine and then using the sperm for artificial insemination.
- Treatment for neurogenic bladder depends on the specific problem and its cause. If the main problem is retention of urine in the bladder, treatment may involve medication to promote better bladder emptying and behavior changes to promote more efficient urination, called timed urination. Occasionally, people may need to periodically insert a thin tube called a catheter through the urethra into the bladder to drain the urine. Learning how to tell when the bladder is full and how to massage the lower abdomen to fully empty the bladder can help as well. If urinary leakage is the main problem, medications or surgery can help.-
Ischemic
Temperature of the skin: decreased
Color of the skin: pallor or cyanotic
Pulsation on peripheral vessels: decreased or absent
Odema: absent
Sensibility: partly decreased or normal
Ulcers: peripheral (distant)
Gangrene: Dry
Neuropatic
Temperature of the skin: normal
Color of the skin: normal or pink
Pulsation on peripheral vessels: normal
Odema: can be
Sensibility: decreased or absent
Ulcers: under the pressure
Gangrene: moist
Clinical summary about DIabetes Mellitus
TYPE 1, or insulin-dependent DIABETES MELLITUS is characterized by pancreatic islet beta cell destruction and absolute insulinopenia.
These individuals are ketosis prone under basal conditions. The onset of the disease is generally in youth, but it can occur at any age. Patients have dependence on daily insulin administration for survival.
Current formulation of the pathogenesis of type 1 DM includes the following:
1. A genetic predisposition, conferred by diabetogenic genes on the short arm of chromosome C, either as part of it or in close proximity to the major histocompatibility complex (MMHC) region (more than 95 % of type 1 diabetes individuals are HLA DR3, DR4 or DR3/DR4; on the other hand, HLA DR2 confers protection against the development of type 1 DM);
2. Putative environmental triggers (possibly viral infections (Coxsackie B, rubella, mumps) or chemical toxins (nitrosourea compounds)) that in genetically susceptible individuals might play a role in initiating the disease process.
3. An immune mechanism gone awry, either initiation of immune destruction or loss of tolerance, leading to slow, progressive loss of pancreatic islet beta cells and eventual clinical onset of type 1 diabetes.
Stages of type 1 DM development (by Flier, 1986)
I. A genetic predisposition or changes of immunity.
Normal β-cells
II. Putative environmental triggers.
III. Active autoimmune insulinitis with β-cells destruction.
Insulinitis
IV. Progression of autoimmune insulities with destruction of >50 % of β-cells.
V. Development of manifest DM.
VI. Total β-cells destruction.
β-cells destruction
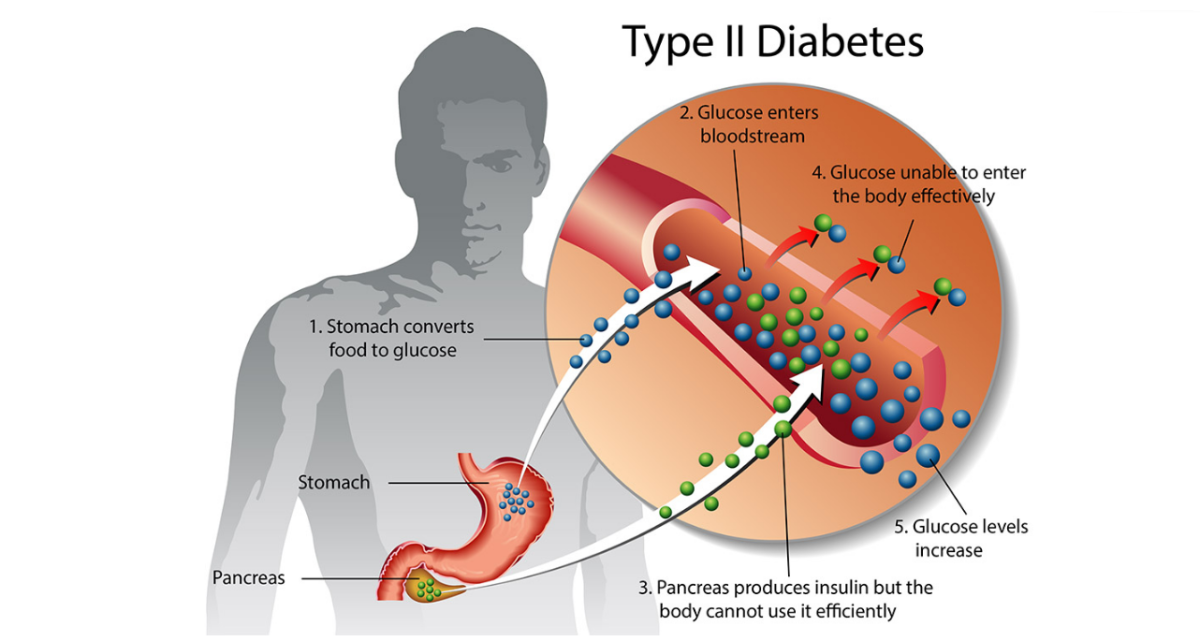
TYPE 2or non-insulin-dependent DIABETES MELLITUS is the most common form of diabetes, accounting for 95 – 90 % of the diabetic population. Most investigators agree that genetic factors underlie NIDDM, but it is probably not caused by defects at a single gene locus. Obesity, diet, physical activity, intrauterine environment, and stress are among the most commonly implicated environmental factors which play a role in the development of the disease. In patients with type 2 DM mostly we can find relative insulin insufficiency (when pancreatic gland secrets insulin but it can have changed structure or weight, or circulating enzymes and antibodies destroy normal insulin, or there are changes of insulin receptors).
Pathogenetic and clinical difference of type 1 and type 2 DM.
Signs
Type 1 DM
1. Age: Young (under 40)
2. Beginning of disease; Acute
3. Duration: Labile
4. Ketosis, ketoacidosis: Often develops
5. Body weight: Decreased or normal
6. Treatment: Insulin, diet
7. Degrees of severity: Middle, hard
8. Season of disease beginning: Frequently autumn-winter period
9. Connection with HBA-system: Present
10. Level of insulin and C-peptide: Decreased or absent
11. Antibodies to β-cells: Present in 80-90% of patients on first week, month
12. Late complications: Microangiopathies
13. Mortality: Less than 10%
14. Spreading: 10-20%
Type 2:
1. Age: middle (above 40)
2. Beginning of disease: Gradual
3. Duration: Stable
4. KEtosis, ketoacidosis: Rarely develops
5. Body weight: Obesity in 80-90% of patients
6. Treatment: Diet, Oral hypoglycemic agents or Insulin
7. Degrees of severity: Mild, middle, hard
8. Season of disease beginning: Absent
9. Connection with HBA-System: Absent
10. Level of Insulin and C-peptide: Frequently normal level.
11. Antibiotics of B-cells: Absent
12. Late complications: Macroangiopathies
13. Mortality: More than 20%
14. Spreading: 80-90%
Etiologic classification of DM (WHO, 1999)
I. Type 1 of DM (destruction of β-cells which mostly leads to absolute insulin insufficiency):
- autoimmune;
- idiopathic.
II. Type 2 of DM (resistance to insulin and relative insulin insufficiency or defect of insulin secretion with or without resistance to insulin).
III. Other specific types:
- genetic defects of β-cells function;
- genetic defects of insulin action;
- pancreatic diseases (chronic pancreatitis; trauma, pancreatectomy; tumor of pancreatic gland; fibrocalculosis; hemochromatosis);
- endocrine disease;
- drug exposures;
- infections and others.
IV. Gestation diabetes.
(Gestation diabetes is defined as hyperglycemia diagnosed for the first time in pregnancy. It occurs in individuals who have an inherited predisposition to develop diabetes and may take the form of either type 1 or type 2 diabetes. Gestation diabetes is associated not only with increased rate of perinatal morbidity and neonatal mortality but also with high incidence of subsequent diabetes in mother. Treatment is with diet modification and insulin. Insulin does not cross placenta while oral hypoglycemic agents cross placenta and therefore contrindicated.)
Stages of DM development
1. Prediabetes (risk factors or predispose factors):
- positive family history of DM;
- persons which were born with weight more than 4,0 kg;
- women in which: = were born children with weight more than 4,0 kg; =had abortions and dead child in anamnesis;
- persons with:
= atherosclerosis, hypertension;
= autoimmune diseases;
= furunculosis;
= rubella, mumps, coxsackie virus, infectious hepatitis, cytomegalovirus, infection mononucleosis.
2. Impaired glucose tolerance (latent DM).
3. Clinical manifestation of DM.
Degrees of severity of DM
1.Mild degree:
1) compensation can be achieved by diet;
2) fast serum glucose is less than 8.4 mmol/l;
3) glucosuria less than 20 gr./l (2 %);
4) proneness to ketosis does not occur; long-term (chronic) complications are rare or only functional stages can be observed.
2.Moderate degree:
1) compensation can be achieved by oral hypoglycemic agents (in patients with type 2 DM) or insulin (in patients with type 1 DM);
2) fast serum glucose is 8.4 to 14.0 mmol/l;
3) glucosuria is 20 to 40 gr./l (2 – 4 %);
4) ketosis can occur; long-term (chronic) complications can be observed (but not last stages).
3.Severe degree:
1) compensation can be achieved by insulin or oral hypoglycemic agents;
2) fast serum glucose is over 14,0 mmol/l;
3) glucosuria is over 40 gr./l (4 %);
4) ketosis is common and last stages of long-term (chronic) complications are present.
Stages of compensations:
1. Compensation.
2. Subcompensation.
3. Decompensation.
Criteria of compensative stage.
1. Patient hasn’t new complains.
2. Fast serum glucose level is normal (but can be under 8.0 mmol/l in patients which haven’t complications and under 11.0 mmol/l in patients with long-term complications).
3. Glucose in urine is absent.
4. Glucose level fluctuation is under 4.4-5.5 mmol/l during the day .
5. Comatose and precomatose status are absent.
6. HbA1c <7,0 % (DM type 1), <6,5% (DM type 2)
Criteria of subcompensative stage.
1. Patient may have new complains.
2. Fast serum glucose is high.
3. Glucosuria is present.
4. Glucose level fluctuation is over 4.4-5.5 mmol/l during the day.
5. Comatose or precomatose status are absent.
6. HbA1c 7,0 – 7,5 % (DM type 1), 6,5 – 7,0 % (DM type 2)
Criteria of decompensative stage:
1. Comatose or precomatose status are present.
2. HbA1c >7,5 % (DM type 1), >7<0% (DM type 2).
Duration of DM
1. Stabile (glucose level fluctuation is under 4.4-5.5 mmol/l during the day and comatose or precomatose status are absent).
2. Labile(glucose level fluctuation is over 4.4-5.5 mmol/l during the day or comatose and precomatose status are present).
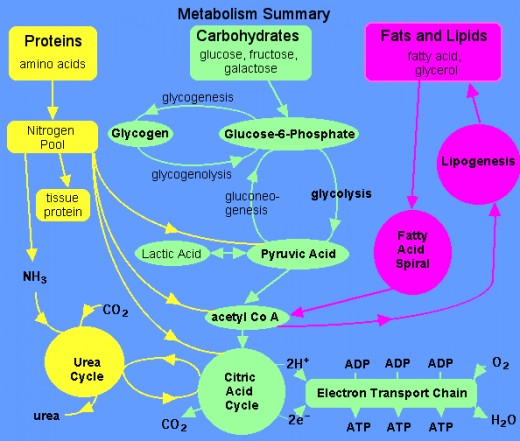
Pathophysiology of DM
Insulin lack
Defective polymorphonuclear function → infection
↑
Hyperglycemia → glucosurea → polyurea → dehydration
↓
Hyperosmolality
Proteolysis → weight loss → muscle wasting → polyphagia
Lipolysis → free fatty acid release → ketosis → acidosis
- polyurea
(once plasma glucose concentration exceeds the renal threshold (about 180 ml/dl or 8 – 9 mmol/l) glucosurea ensues. Osmotic diuresis induced by glucose results in polyurea and subsequent polydipsia);
- polidipsia
(as more water is excreted, the body requires more water intake);
- polyphagia
(this occurs to lack of energy);
- loss of weight
(energy (calories) is lost as glucose in the urine. Loss of water itself also contributes to weight loss. Increased proteolysis with mobilization of aminoacids leads to enhancement of protein catabolism and loss of weight, notably in muscle mass);
- fatigue and weakness
(probably occur as a result of decreased glucose utilization and electrolyte abnormalities);
- acidosis
(develops due to increased lipolysis which cause the release of free fatty acids, which are metabolized to ketones by the liver).
Presenting signs and symptoms of type2 DM include: polyurea, polydipsia, polyphagia; the majority of individuals (80 – 85 %) are obese, but it can also occur in lean persons.
Patients with DM are at risk if developing of chronic degenerative complications.
Physical examination.
Skin
Diabetes can affect every part of the body, including the skin. The skin is a common target of DM As many as one third of people with diabetes will have a skin disorder caused or affected by diabetes at some time in their lives. In fact, such problems are sometimes the first sign that a person has diabetes. Luckily, most skin conditions can be prevented or easily treated if caught early.
Some of these problems are skin conditions anyone can have, but people with diabetes get more easily. These include bacterial infections, fungal infections, and itching. Other skin problems happen mostly or only to people with diabetes. These include diabetic dermopathy, necrobiosis lipoidica diabeticorum, diabetic blisters, and eruptive xanthomatosis.
Bacterial Infections
Several kinds of bacterial infections occur in people with diabetes. One common one are styes. These are infections of the glands of the eyelid. Another kind of infection are boils, or infections of the hair follicles. Carbuncles are deep infections of the skin and the tissue underneath. Infections can also occur around the nails.
Inflamed tissues are usually hot, swollen, red, and painful. Several different organisms can cause infections. The most common ones are the Staphylococcus bacteria, also called staph.
Once, bacterial infections were life threatening, especially for people with diabetes. Today, death is rare, thanks to antibiotics and better methods of blood sugar control.
But even today, people with diabetes have more bacterial infections than other people do.
Fungal Infections
The culprit in fungal infections of people with diabetes is often Candida albicans. This yeast-like fungus can create itchy rashes of moist, red areas surrounded by tiny blisters and scales. These infections often occur in warm, moist folds of the skin. Problem areas are under the breasts, around the nails, between fingers and toes, in the corners of the mouth, under the foreskin (in uncircumcised men), and in the armpits and groin.
Common fungal infections include jock itch, athlete's foot, ringworm (a ring-shaped itchy patch), and vaginal infection that causes itching.
Itching
Localized itching is often caused by diabetes. It can be caused by a yeast infection, dry skin, or poor circulation. When poor circulation is the cause of itching, the itchiest areas may be the lower parts of the legs.
Diabetic Dermopathy
Diabetes can cause changes in the small blood vessels. These changes can cause skin problems called diabetic dermopathy.
Dermopathy often looks like light brown, scaly patches. These patches may be oval or circular. Some people mistake them for age spots. This disorder most often occurs on the front of both legs. But the legs may not be affected to the same degree. The patches do not hurt, open up, or itch.
Necrobiosis Lipoidica Diabeticorum
Another disease that may be caused by changes in the blood vessels is necrobiosis lipoidica diabeticorum (NLD). NLD is similar to diabetic dermopathy. The difference is that the spots are fewer, but larger and deeper.Iit consists of skin necrosis with lipid infiltration and is also characteristically found in the pretibial area. The lesions resemble red plaques with distinct border.s
NLD often starts as a dull red raised area. After a while, it looks like a shiny scar with a violet border. The blood vessels under the skin may become easier to see. Sometimes NLD is itchy and painful. Sometimes the spots crack open.
NLD is a rare condition. Adult women are the most likely to get it. As long as the sores do not break open, you do not need to have it treated. But if you get open sores, see your doctor for treatment.
Atherosclerosis
Thickening of the arteries - atherosclerosis - can affect the skin on the legs. People with diabetes tend to get atherosclerosis at younger ages than other people do.
As atherosclerosis narrows the blood vessels, the skin changes. It becomes hairless, thin, cool, and shiny. The toes become cold. Toenails thicken and discolor. And exercise causes pain in the calf muscles because the muscles are not getting enough oxygen.
Because blood carries the infection-fighting white cells, affected legs heal slowly when the skin in injured. Even minor scrapes can result in open sores that heal slowly.
People with neuropathy are more likely to suffer foot injuries. These occur because the person does not feel pain, heat, cold, or pressure as well. The person can have an injured foot and not know about it. The wound goes uncared for, and so infections develop easily. Atherosclerosis can make things worse. The reduced blood flow can cause the infection to become severe.
Allergic Reactions
Allergic skin reactions can occur in response to medicines, such as insulin or diabetes pills. You should see your doctor if you think you are having a reaction to a medicine. Be on the lookout for rashes, depressions, or bumps at the sites where you inject insulin.
Diabetic Blisters (Bullosis Diabeticorum)
Rarely, people with diabetes erupt in blisters. Diabetic blisters can occur on the backs of fingers, hands, toes, feet, and sometimes, on legs or forearms.
These sores look like burn blisters. They sometimes are large. But they are painless and have no redness around them. They heal by themselves, usually without scars, in about three weeks. They often occur in people who have diabetic neuropathy. The only treatment is to bring blood sugar levels under control.
Eruptive Xanthomatosis
Eruptive xanthomatosis is another condition caused by diabetes that's out of control. It consists of firm, yellow, pea-like enlargements in the skin. Each bump has a red halo and may itch. This condition occurs most often on the backs of hands, feet, arms, legs, elbows, knees and buttocks.
The disorder usually occurs in young men with type 1 diabetes. The person often has high levels of cholesterol and fat (particularly hyperchylomicronemia) in the blood. Like diabetic blisters, these bumps disappear when diabetes control is restored.
Digital Sclerosis
Sometimes, people with diabetes develop tight, thick, waxy skin on the backs of their hands. Sometimes skin on the toes and forehead also becomes thick. The finger joints become stiff and can no longer move the way they should. Rarely, knees, ankles, or elbows also get stiff.
This condition happens to about one third of people who have type 1 diabetes. The only treatment is to bring blood sugar levels under control.
Disseminated Granuloma Annulare
In disseminated granuloma annulare, the person has sharply defined ring-shaped or arc-shaped raised areas on the skin. These rashes occur most often on parts of the body far from the trunk (for example, the fingers or ears). But sometimes the raised areas occur on the trunk. They can be red, red-brown, or skin-colored.
Acanthosis Nigricans
Acanthosis nigricans is a condition in which tan or brown raised areas appear on the sides of the neck, armpits, and groin. Sometimes they also occur on the hands, elbows, and knees.
Acanthosis nigricans usually strikes people who are very overweight. The best treatment is to lose weight. Some creams can help the spots look better.
Subcutaneous adipose tissue
The abdomen type of obesity is common in patients with type 2 DM. Sometimes generalized subcutaneous adipose tissue atrophy can be observed in diabetics.
Bones and Joints
• Osteoporosis, osteoarthropaphy, diabetic chairopathy (decreasing of the movements of joints) can be find in patients with DM also.
Gastrointestinal tract
Paradontosis, gastritis with decreased secretion ability, gastroduodenitis, hepatosis and diarrhea are common in patients with DM.
Cardiovascular system (CVS)
Involvement of CVS, particularly the coronary circulation, is common in patients with DM.
The heart, arteries, arterioles, and capillaries can be affected. Cardiovascular changes tend to occur earlier in patients with DM when compared with individuals of the same age. Several factors play a role in the high incidence of coronary artery disease seen in patients with DM. These include age of the patient, duration and severity of the diabetes, and presence of other risk factors such as hypertension, smoking and hyperlipoproteinemia. It has been suggested that in some patients with DM, involvement of the small vessels of the heart can lead to cardiomyopathy, independent of narrowing of the major coronary arteries. Myocardial infarction is responsible for at least half of deaths in diabetic patients, and mortality rate for the diabetics is higher than that for nondiabetics of the same age who develop this complication.
Hypertension is common in patients with DM, particularly in the presence of renal disease (as a result of atherosclerosis, destruction of juxtaglomerular cells, sympathetic-nervous-system dysfunction and volume expansion).
Atherosclerosis of femoral, popliteal and calf larger arteries may lead to intermittent claudication, cold extremities, numbness, tingling and gangrene.
Respiratory system
Mucomycosis of the nasopharinx, sinusitis, bronchitis, pneumonia, tuberculosis are more common in patients with diabetes than in nondiabetics.
Kidneys and urinary tract
Renal disease include diabetic nephropathy, necrosing renal papillitis, acute tubular necrosis, lupus erythematosus, acute poststreptococcal and membranoproliferative glomerulonephritis, focal glomerulosclerosis, idiopathic membranous nephropathy, nonspecific immune complex glomerulonephritides, infections can occur in any part of the urinary tract. Last are caused when bacteria, usually from the digestive system, reach the urinary tract. If bacteria are growing in the urethra, the infection is called urethritis. The bacteria may travel up the urinary tract and cause a bladder infection, called cystitis. An untreated infection may go farther into the body and causepyelonephritis, a kidney infection. Some people have chronic or recurrent urinary tract infections.
There is thicken of basement membranes and mesangial expansion and Kimmelstiel -Wilson nodule
Symptoms of urinary tract infections may include
- a frequent urge to urinate
- pain or burning in the bladder or urethra during urination
- cloudy or reddish urine
- fatigue or shakiness
- in women, pressure above the pubic bone
- in men, a feeling of fullness in the rectum
Obviously, these abnormalities, with exception of diabetic nephropathy, are not at all peculiar to DM and can be observed in many other conditions.
Eyes
Complications of the eyes include: ceratities, retinatis, chorioretinatis, cataracts. The last one occurs commonly in the patients with long-standing DM and may be related to uncontrolled hyperglycemia (glucose metabolism by the lens does not require the presence of insulin. The epithelial cells of the lens contain the enzyme aldose reductase, which converts glucose into sorbitol. This sugar may be subsequently converted into fructose by sorbitol dehydrogenase. Sorbitol is retained inside the cells because of its difficulty in transversing plasma membranes. The rise in intracellular osmolality leads to increased water uptake and swelling of the lens).
The diagnosis of DM
The diagnosis of DM may be straightforward or very difficult.
(The presence of the marked hyperglycemia, glucosuria, polyuria, polydipsia, polyphagia, lethargy, a tendency to acquire infections, and physical findings consistent with the disease should offer no difficulty in arriving at the correct diagnosis. On the other hand, mild glucose intolerance in the absence of symptoms or physical findings does not necessarily indicate that DM is present.)
THE DIAGNOSIS OF DM INCLUDE:
I. Clinical manifestationsof DM.
II. Laboratory findings.
1) fasting serum glucose (if the value is over 6,7 mmol/l (120 mg/dl) on two or more separate days, the patient probably has DM);
2) the glucose tolerance test (GTT):
If the diagnosis is still in doubt, then perform a GTT.
Conditions for performing an oral GTT have been standardized:
- no special dietary preparation is required for an oral GTT unless the patient has been ingesting <150 gm/day of carbohydrate. Then give 150 – 200 gm carbohydrate daily for 3 days prior to test;
- unrestricted physical activity should proceed the test;
- test is performed in the morning, following overnight fast of 10 to 16 hours;
- subjects should remain seated, without prior coffee or smoking;
- blood for glucose determination is obtained from an antercubital vein before glucose ingestion and every 30 minutes far 2 hours after ingestion ;
- the amount of glucose given is 75 g for adults (100 g pregnant women, and 1,75 g/kg of ideal body weight for children). Patient have to drink glucose dissolved in 250 ml of water;
- the criteria for diagnosing diabetes in pregnant, adults are:
Fasting serum glucose, mmol/l
3,3 – 5,5
2 hours after glucose loading, mmol/l
<7,8
Capillary blood
5,6 – 6,1
Health
7,8 – 11,1
Impaired glucose tolerance
> 6,1
Diabetes mellitus
> 11,1
a) a fasting serum glucose more than 6,1 mmol/l (120 gm/dl);
b) a 2-hour postprandial serum glucose over 11,1 mmol/l (200 gm/dl);
- the criteria for diagnosing of impaired glucose tolerance are:
a) a fasting serum glucose more than 5,5 mmol/l (100 gm/dl);
b) a 2-hour postprandial serum glucose more than 7,8 mmol/l (140 gm/dl) but less than 11,1 mmol/l (200 gm/dl).
The major indication for an oral GTT is to exclude or diagnose DM (mostly 2) in those suspected of having diabetes although fasting or symptomatic hyperglycemia is absent; e.g., in patients with a clinical condition that might be related to undiagnosed DM (e.g., polyneuropathy, retinopathy). Various conditions (other than DM) and drugs can cause abnormalities in the oral GTT. The criteria of DM do not apply to patients treated with drugs that can impair glucose tolerance (e.g., thiazids, glucocorticoids, indometacin, nicotinic acid, oral contraceptives containing synthetic estrogenes) or to patients who develop nausea, sweating, faintness or pallor during the test, or to have infections, hepatic, renal and endocrine disease that impairs glucose tolerance.
3) islet cell antibody levels will be positive prior to any insulin administration in 60 – 80 % of patients with type 1 DM;
4) C-peptide (it is not affected by antibodies to exogenous insulin and is used to distinguish type 1and 2 DM if there is still a need after clinical determination);
5) glucose level in urine;
6) glycohemoglobin (Hb1Ac) (this test is an indicator of blood sugar control during the previous 2-to-3-month period);
7) acetonurea;
8) blood lipids and others.
III. Instrumental investigations usually are used to diagnose chronic complications of DM.
Diabetes treatment and managements




Acknowledgements








contributors






Detailed treatment
Methods of treatment of Diabetes Mellitus
Diet, Oral hypoglycemic agents or insulin (indications for each vary with the type of Diabetes Mellitus and severity of the disease). Exercise program, physiotherapy (plant’s therapy), Non-traditional methods of treatment and Education of patients.
Education of the patients The first education should be about the nature of the disease, the importance of its control, all aspects of self-management and routine practices to minimize the development or severity of the diabetes complications. Physicians have to educate motivate and monitor progress, the patients must understand the importance of Life-style changing, the nature of diabetes mellitus and importance of metabolic control. The Knowledge about the principles and importance of good nutrition and reasonable exercise program and the principles of adequate foot, dental and skin care by the patient is very essential. Also the patient can know the treatment of Diabetes Mellitus during the periods of illness. The physician has to educate on the techniques of Insulin administration and measurement of Urine and blood glucose level (if taking Insulin); recognition of hypoglycemia, its causes and method of prevention and finally the importance of general and specific measures to minimize in the best possible way diabetic complications and maintenance of good heath overall.
The main principles of diet
• Balanced diet should include physiologic meal components: Carbohydrate comprises 50-60% of total calories; fat-24-25% and protein- 16-15%). Normal calorie diet in patients with type I Diabetes Mellitus (35-50 Kcal/Kg of ideal weight [weight= height-100]) and lowcaloric diet in Obese persons (mostly in patients with type II Diabetes Mellitus (20-25 Kcal/Kg of ideal weight) We try to decrease weight in obese patients on 1-2 Kg/month by such diet.
• Regimen has to consist of 4-6 small feedings a day. The most frequent regimen consists of 4 feedings a day in which: 1. breakfast comprises 30% of total calories 2. dinner- 40% 3. Lunch- 10% 4. supper- 20%. Sometimes patients need second breakfast (when they have a tendency to develop hyperglycemia). In such a case, it comprises 15% of the total calories and we decrease the quantity of calories of the first breakfast and dinner).
• Exclusion of high-Calorie Carbohydrates (sugar, biscuits, white bread, alcohol). Also increasing the quantity of high fiber-containing foods (fruits [exclusion: banana, grapes], vegetables, cereal grams whole grains flours, bran, patients need 40g fibers per day. • Limiting of meat fat, butter margarine in diet decrease red and brown meats, increase poultry and fish, encourage skim milked-based cheeses. Should be used skim or low-fat milk, not more than 2-3 eggs weekly. Finally Alcohol should be avoided as much as possible because it constitutes a source of additional calories it may worsen hyperglycemia and it may potentiate the hypoglycemic effects of Insulin and oral hypoglycemic agents.
Oral hypoglycemic agents
• Inadequate control of hyperglycemia by the diet and exercises interventions suggests the need for a good glucose-lowering agent.
• Oral hypoglycemic agents are useful only in the chronic management of patients with type II Diabetes Mellitus.
• The most commonly used are: 1. The sulfanilureas 2. The Biguanides 3. Alpha-glucosidase inhibitors, 4. Non-sulfanylureas Insulin stimulators (glinides) 5. Thiosolidinediones (glitazones) 6.
SulfanilUreas Include:
• 1st generation: Tolbutamide, chloripropamide, Tolazemide, Acetohexamide (now are not used in treatment of diabetes)
• 2nd generation: Gilbenclamide (Maninil [3.5mg, 5mg] Daonil [5mg]). Gliquidon (Glurenorm (0.03); Minidiab (5mg); GLiclazide (Diamicron [0.008]; Glipizide; • 3rd generation: Glimepiride (Amaryl (1mg, 2mg).
commonly used Sulphonylureas
The second generation drugs are commonly used such as: Glibenclamid (Maninil, Euglucan, Daonil, Glonil, Gilamat, Gliben, Glucoven). Gliquidon (Glurenorm, Beglicor) are without hepato and nephrotoxic effects, metabolism through the intestine. Also Gliclazid (Diamicron, DIabeton, predian, GLizid) DIabeton normalizes microcirculation and blood aggregation. Finally, the third generation drugs eg, GLimepirin (Amanyl) are used.
Action of Sulfanilureas
It has influence on the Pancreatic gland by increasing the B-cells sensitivity to glucose and as a result, higher secretion of Insulin and also the stimulation of the exocytosis of Insulin by insulocytes. Its non-pancreatic influences are: Increasing the number of receptors to Insulin, normalization of receptors’ sensitivity to Insulin, Increasing of glucose transportation inside muscle cells, stimulation of glycogen synthesis, decreasing of glycogenolysis and glyconeogenesis. It also decreases glucagons secretion.
Indications and contra-indications to Sulfanilureas Usage Indicated for patients with type II Diabetis Mellitus (Over the age of 35-50 years) who do not suffer severe metabolic abnormalities (hyperglycemia) ketosis or hyperosmolality and its good for those with duration of Diabetes of less than 15 years. It is contraindicated in Type I Diabetes Mellitus, Blood diseases, acute infections, heart, cerebral diseases, trauman, pregnant diabetes or lactation, III to IV stages of angiopathy (but Glurenorm can be used in patients with chronic renal failure, because of gastrointestinal tract excretion); then also contra-indicated in Coma and Pre-coma.
commonly used biguanides
• Metformin (dianormet siofor Metforgamma, Metfodar) Glucophage Forte, Metformin 0.25-0.5 in 1 tablet while Glucophage Forte is 0.85 in 1 Tablet, taken 0.5-1.5; 8-10 hours of action and 0.5-2.0; 12-14 hours of duration of action.
• Buformin (Adebit) 0.05 in 1 tablet. Daily dose is 0.1-0.2, 8-10 hours of action. Buformin, Refard; 0.17; daily dose is 0.17- 0.34, 12- 14 hours of action.
Action of biguanides Inhibition of gastro-intestinal glucose absorption, decreasing of glyconeogenesis, Lipogenesis; enhancing glucose transport into muscle cells; Increasing the quantity of insulin’s receptors; stimulation of anaerobic and partly aerobic glycolis and anorrhexogenic effects.
Indications and contra-indications of biguanides Usage Obese patients with type II Diabetes Mellitus, with middle severity of the disease without ketosis. They can be used with the combination of sulfanilureas when sulfonylureas alone have proven inadequate to treat Diabetes Mellitus. Contra-Indications of biguanides usage are; Type I Diabetes Mellitus; heart and Lung disease with their insufficiency 9Chronic heart and Lung faialure); Status with hypoxemia, acute and chronic liver and Kidney diseases with decreased function; Pregnant diabetes, lactation, Old age, alcoholism; Coma and Pre-coma.
Side Effects 1. Allergy 2. Gastro-Intestinal tract disorders 3. Lacto-acidosis.
Alpha-glucosidase Inhibitors
• Acarbosa (Glucobay, Glucor, prandase, precose)- 0.05-0.7, in 1 tablet; daily dose of 0.15-0.6 and a duration of action of 2.7-9.6.
• Miglitol (0.025; 0.05; 0.7 dose in 1 tablet); 0.05-0.3 daily, 2-4hrs of action. • Guar Gum (Guarem)- 5.0 granules, 15-30 daily dosage.
Action of alpha-glucosidase Inhibitors Inhibition of gastro-intestinal tract absorptions (blocation of alpha-glucosidase). Lowering of post prandial glucose level (Postprandial “spikes’ in blood glucose are increasingly implicated as a major cause of Cardiovascular complications); Partly reducing fasting glucose levels by indirectly stimulating insulin secretion in patients who retain B-cells function (and acarbose has a protective effect on B-cells).
Indications and Contra-Indications to alpha-glucosidase Inhibitors Usage Diabetes Mellitus Type II with or without obesity when diet and exercises are not effective. Diabetes mellitus significant violations of glycaemia during a day; secondary sulfanilureas failure; Insulin resistance, Allergic reactions to other hypoglycemic drugs and Hypercholesterolemia. It is contra-indicated in Type I Diabetes Mellitus; Chronic gastro-Intestinal disorders: Pancreatitis, Colitis, Hepatitis. Side effects are flatulence, abdominal bloating, diarrhea.
Non-sulfanylUreas Insulin-Stimulators
Repaglinid (Novonorm, Reglid; meglitinide analogs); 0.00l-0.004, dose in 1 tablet, 0.004-0.009, daily dosage for 3-4 hours. Nateglinid (starlix) (D-Phenilalanine-derivatives); 0.006-0.18 dosage in 1 tablet of 0.18-0.54 daily dose of 1.5- 3 hours duration.
Action of non-SulfanylUreas Insulin Stimulator Stimulation of Insulin production at meal times; Very rapid absorption from the intestine and metabolizing in the liver; (plasma half-life is less than 1 hour).
Indications and contra-Indications to non-sulfanylUreas Insulin stimulator Can be used in elderly with Type II Diabetes Mellitus (due to short half-life) and in renal impairment (because it is metabolized in liver). SulfanylUreas Insulin stimulators are contraindicated in hypoglycemia, transient elevation of Liver enzymes rash and visual disturbances.
commonly used thiozolidinedones
Rosiglitazour (Avandia, roglit, Rosinorm; 0.002-0.008 dose in 1 tablet, of 0.004-0.008 daily dosage. Also, Proglitazone (Actos, Pionorm) of 0.0015-0.045 dose in 1 tablet of 0.015-0.3 daily dosage. Both are active for 24 hours.
Action of thiozolidindiones Agonist to the receptors of the nucleus PPARY of the fat, muscle tissues and the liver. Also increasing of the glucose passage to these tissues; Increasing of Insulin synthesis in the B-cells, increasing of the insulin amount; increasing of glycogen synthesis in the Liver, decreasing of gluconeogenesis and Decreasing of triglycerides.
Indications and contra-indications to thiozolidindiones Usage Diabetes Mellitus Type II, when diet and exercises are no effective; using with sulfanilUreas, biguanides in case of their insufficient efficacy. However, at present, only pioglitazone is approved for use in combination with Insulin. Contra-indications are Diabetic Coma, Pre-coma, Keto-acidosis, Acute and Chronic diseases of the Liver, Heart failure, Pregnancy, lactation, children, teenagers, Allergic reactions to the drug. Side effects are as follows; • Hypoglycemic conditions (rarely) • Peripheral edema • Anemia • Obesity • Elevations of Liver enzymes
Combined preparations Gilbomet consists of Maninil 2.5mg and siofor 400mg. Avandamet consists of Rosiglitazone maleat 2mg and Metformin 200mg.
Insulin Therapy
In 1921, Banting and Best extracted Insulin from Pancreatic gland of newborn Cow. In 1955, Sanger established molecular structure of Insulin and in 1964, Katsoyanis (USA), and Tzan (1965, Germany) synthesized human Insulin.
Indications The following are indications of Insulin: All patients with Type I Diabetes Mellitus; some patients with Type II Diabetes mellitus, Uncontrolled diabetes by diet or oral hypoglycemic agents, Ketoacidosis, Coma, Acute and Chronic Liver and Kidneys disease with decreased function, Pregnancy and lactation, II to IV stages of angiopathy, Infectious diseases, Acute heart and cerebral diseases and surgery.
Insulin preparations
Insulin preparations of Ultrashort action (human analog, recombinant) 1. Novo Rapid (Novo-Nordisk) 2. Humalog (lilly) 3. Epaidia All beginning for 2-10 minutes, maximum action by 40-50 minutes, then total duration of action is between 3 to 5 hours. Insulin preparations of short action 1. Monodar (Indar) 2. Monodar R 3. Monodar RR 4. Monodar R100 5. MonodarR100R 6. Farmasulin HN 7. Actrapid (MCHM) (Novo-Nordisk) For all, beginning of action is between 1 to 3 hours and duration of action lasts for 5-8 hours.
Insulin preparations of intermediate action 1. Monodar B (Indar) 2. Humodar B (Indar) 3. Farmasulin H NP (Farmak) 4. Protaphan (MC, HM) Novo-Nordisk. 5. Insuman basal Aventis 6. Humulin NPH Lilly 7. Monotard HM Novo-Nordisk Action • Beginning of action is between 1-1.5hours • Maximum action is between 6 to 8 hours • Duration of action is between 12 to 18 hours.
Insulin preparations of Long Action Examples are Farmasulin HL (Farmak); Ultralente Humulin (lilly); Ultratard HM, MC suinsulin Ultralong (Indar); Glargine (Lantus-Aventis); Detemir, Levemir. Action • Beginning of action is between 3 to 4 hours • Maximum action takes place between 10 to 12 hours • Duration of action is between 24 hours to 30 hours
Combined Insulin Preparations Insumani comb 25/75 (Aventis). Its beginning of action is 30 minutes, maximum action is 1.5 to 2 hours and then its duration of action is 12-18 hours. Mixtard 30 HM (Novo-Nordisk). Its beginning of action is 30-40 minutes; and maximum action is 1.5-3 hours. Its duration of action is 12- 48 hours. Monodar K30 (Indar). Its beginning of action is 30-40 minutes, its maximum action is 1-3 hours and its duration of action is 12- 16 hours. Monodar K50 (Indar). Beginning of action- 30 minutes, maximum action- 1-3 hours and Durations of action is 6-10 hours. Humodar K15 (Indar). Beginning of action is 30-45 minutes, maximum action is for 1.5 to 3 hours and then Duration of action is 12-18 hours. Humodar K25 (Indar). Beginning of action is 30-40 minutes; maximum action is 1.5-3hours, then duration of action is 12-18 hours. Humodar K50 (Indar); Beginning of action is 30 minutes, maximum action is 1-3 hours and then duration of action is 6- 10 hours.
Initiation and modification of Insulin therapy
It is started as soon as possible in an attempt to “rest” the damaged islet cells and help to “Induce” a remission (“honeymoon” phase). The daily Insulin requirement in patients: On the first year of the disease is 0.3- 0.5 unit of Insulin per Kg of body weight (0.5- if the patient with Ketosis or DKA). On the next years is 0.6- 0.8-1.0 unite/Kg of body weight.
1 Unite
It is activity of 0.04082mg of crystalic insulin (standart). We can use traditional or multiple component insulin program. The last is better. Advantages include the following: • Hypoglycemic reactions may be decreased or prevented because smaller doses of Insulin are needed; • More physiologic match of Insulin to meals is achieved • It uses three or four shots of short-acting insulin (1/3 of total daily dose) Plus intermediate- acting (2/3 of total daily dose) Insulin daily. • 2/3 of the total daily dose we give before breakfast, 1/3 in the evening and then make correction due to the glucose blood level. Insulin doses should be given 30 minutes before meals to allow for adequate absorption of regular Insulin.
Other commonly used Insulin treatment algorithms
• Single pre-breakfast injection of intermediate-acting insulin.
• Intermediate-acting Insulin: pre-breakfast injection of 2/3 total daily dose, 1/3 of daily dose before dinner.
• Combination of intermediate- and short-acting Insulin
• Single pre-breakfast injection of 2/3 intermediate-acting + 1/3 of short-acting
• 2/3- before breakfast, 1/3-before dinner; 2/3-intermediate-acting, ½-short-acting.
• Short-acting Insulin, ½ hour before each meal and a small dose of intermediate-acting Insulin at bedtime.
• Combination of long-acting (in pre-breakfast time) and short-acting Insulin (1/2 hour before each meal).
some peculiarities of Insulin therapy
• Insulin acts faster when is administered intravenously.
• Subcutaneous and intramuscular absorption of Insulin is decreased in the dehydrated or hypotensive patients;
• It is necessary to change the Insulinn injection site (because the absorption is more rapid from the new sites);
• The most rapid absorption from the abdomen
• Exercise accelerates Insulin absorption (before planned exercise program patient has to decrease Insulin dose or take more caloric diet).
Future directions in improving glycemic control
• Nasal Insulin preparations
• Pancreatic transplantation
• Islet replacement therapy
• Genetically engineered pseudo-beta cells.
• Islet cell transplant is the process whereby Islet cells are transplanted from a healthy donor pancreas to a person with Type I diabetes, where they begin producing Insulin.
Side effects and complications of Insulin Therapy
1. Hypoglycemia This complication represents Insulin excess and it can occur at any time (frequently at night). Common symptoms are: early-morning headaches. Precipitating factors are: Irregular ingesting of food’ extreme activity, alcohol interaction, Liver or renal disease; hypopituitarism, adrenal insufficiency etc. It can be treated by preventing coma in such ways:
• Eat Candy or drink sweet orange juice (when the symptoms develop) • Receive intravenous glucose
• 1mg of glucagons administered subcutaneously
• Gradual reduction of Insulin dose in future. Hypoglycemia is a syndrome characterized by sympathetic nervous system and the Stimulation of central nervous system dysfunction that are provoked by an abnormally low-plasma glucose level. Hypoglycemia represents Insulin excess and it can occur at any time. Clinical presentations are as follows: Adrenergic symptoms which are attributed to increased sympathetic activity and epinephrine release. Such symptoms are:
• Sweating
• Nervousness
• Faintness
• Palpitation
• Sometimes hunger
• Impaired vision
• Dizziness
• Fast heartbeat
• Shaking
• Anxiety
• Irritabilityweakness/fatigue The second set of clinical presentations is the Cerebral nervous system manifestations: Confusion inappropriate behavior (which can be mistaken for inebriation); Visual disturbances, stupor, Coma or seizures. (Improvement in the cerebral nervous system manifestations will be with a rise in blood glucose). A common symptom of hypoglycemia is the early morning headache, which is usually present when the patient wakes up. Patients should be familiar with the symptoms of the hypoglycemia but some of them are not heralded by symptoms.
Physical examination
1. The skin is cold, moist
2. Hyper-reflexia can be elicited
3. Hypoglycemic coma is commonly associated with abnormally low body temperatre
4. Patient may be unconscious
5. Laboratory finding shows a very low level of blood glucose Clinical treatment Insulin-treated patients are advised to carry sugar lumps, candy, or glucose tablets at all time. If the symptoms of hypoglycemia develops, the patients have to drink a glass of fruit juice or water with 3 tablespoon of table sugar addd or eat Candy, and to teach their family members to give such treatment if they suddenly exhibit confusion or inappropriate behavior. Another treatment alternative is GLucagon, 0.5- 1Unit (0.5-1ml) Subcutaneous, intramuscular or intravenous. If the patient does not respond to 1 unit of glucagons within 25 minutes,, further injections are unlikely to be effective, and are not recommended. Another alternative also can be an intravenous injection of 20 or 100ml of 40% glucose (10% glucose may be needed) until it clearly can be stopped safely. Finally, Glucocorticoids and adrenaline are helpful as well.
2. Somogyi effect (Somogyi phenomenon, rebound effect)
It is caused by over-insulinazation: hyperglycemia, proceeded by insulin-induced hypoglycemia. Hypoglycemia causes an increase in the secreation of the counter regulatory hormones (glucagons, epinephrine, Cortisol, growth hormone), which inhibit Insulin Secretion and increase glucose output by the liver (as a result of the stimulation of glucogenolysis and glycogenesis). It is treated by the gradual reduction of Insulin dose.
3. Dawn Phenomenon
Many patients with Type I Diabetes demonstrate an early morning (4-8 am) rise in glucose levels, because of activation of counterregulatory hormones. It may be cnfused with the somogyi phenomenon. Sampling of glucose levels throughtout the night might help differentiate the two conditions. Its treatment is by an earlier injection in the morning (5-6 am) and most suggest a late evening )before bedtime) injection of intermediate acting Insulin.
4. Allergic reactions
These include burning and itching at the site of insulin injection’ skin rash, vasculaties, Purpura and anaphylactic reaction. It is treated by the following ways: • Antibistamines • Changing of standard Insulin to pure pork Insulin or to human Insulin • In extreme cases-glucocorticoids.
5. Insulin resistance
Clinical status characterized by Insulin resistance are:
1. Obesity
2. therapy with oral contraceptives
3. glucocorticoid therapy
4. acromegaly
5. cushing’s syndrome
6. acanthosis nigricans
7. chronic liver or renal resistance
8. Non-true insulin resistance maybe caused by long-time injections of Insulin into the one site.
6. Lipodystrophy
It is atrophy or hypertrophy of the adipose-tissue which occur at the site of Insulin Injection. Changing the site of injection and the usage of Human Insulin are the basic treatment strategies of Lipodystrophy
Exercise program
• Exercise is an excellent adjunct to diet therapy, but it is very ineffective when used as the sole weight-reducing modality.
• Exercises must be clearly planned and depend on patient’s ability and the physical conditions exclusion of the competition’s elements.
• Exercises may be valuable adjunct to the management of the Diabetes Mellitus by:
1. Lowering blood glucose concentration
2. decreasing insulin requirements
3. Potentiation of the beneficial effects of diets and other therapy
4. To prevent hypoglycemia, patients should carefully monitor glucose level and taking of Insulin. Mostly they need to reduce the Insulin dosage by 20-25% on the day that strenuous exercises is planned.
Plant therapy (Phytotherapy)
• Hypoglycemic action
• Treatment of chronic diabetes complications
• Influence on the immune reactivity.
Skin
The skin is dry and itchy. There would be infection of the skin by bacteria and fungi, candidiasis of external female genitalia, hyper-keratosis, nail disorders are common in the patients with Diabetes Mellitus but nothing is specific with regard to their development. The most common skin lesion is, diabetic dermopathy (it is characterized by brown, atropic, well-demarcated areas in the pre-tibial region which resemble sears), besides patients sometimes have Xanthoma diabeticorum, which is usually located on the buttocks, elbows and knees, looking like eruptions.
Diabetic retinopathy
Stage I: Background retinopathy is usually the earliest sign and consists of retinal microaneurysms, hard and soft exudates.
Bones and Joints
Osteoporosis and Osteoarthropathy can be found in patients with Diabetes Mellitus also. Diabetes Chairopathy (decreasing of the movements of Joints) could also be another sign. Stage I: Maculopathy or pre-poliferative retinopathy is characterized by macular edemas and or hemorrhages. Stage II: the hallmark of proliferative retinopathy is neurovascularization, i.e growth of new vessels in areas of hypoperfusion. Adhesion of the vessels to the vitreous leads to retinal detachment vitreous hemorrhage and others. The prognosis is extremely poor. 5 years after recognition of this complication 50% of the patients are blind.
ACKNOWLEDGEMENTS
1. Proff Smiyan Svetlana (the Professor and head of Nephronology department for foreign students, Ternopil Medical Univeristy- Ukraine.
2. Dr, Lepyavkon A. Andreivich (MD.)- Department of Internal Medicine (Endocrinology and Pulmonology branch), Ternopil Medical University-Ukraine
3. Dr. Sofia Gusak (MD).- Department of Internal Medicine (Endocrinology branch), Ternopil Medical University-Ukraine.