Interaction of the Antidiabetic Vanadium Compounds with Hemoglobin and Red Blood Cells
Condensation of the Research
Purpose of the Study
This study sought to explore the interaction of the VIVO2+ ion with hemoglobin. The major reason for undertaking this study was to investigate the binding of hemoglobin to the VIVO2+ ion at normal pH.
Interaction of the Antidiabetic Vanadium Compounds in the body
Background
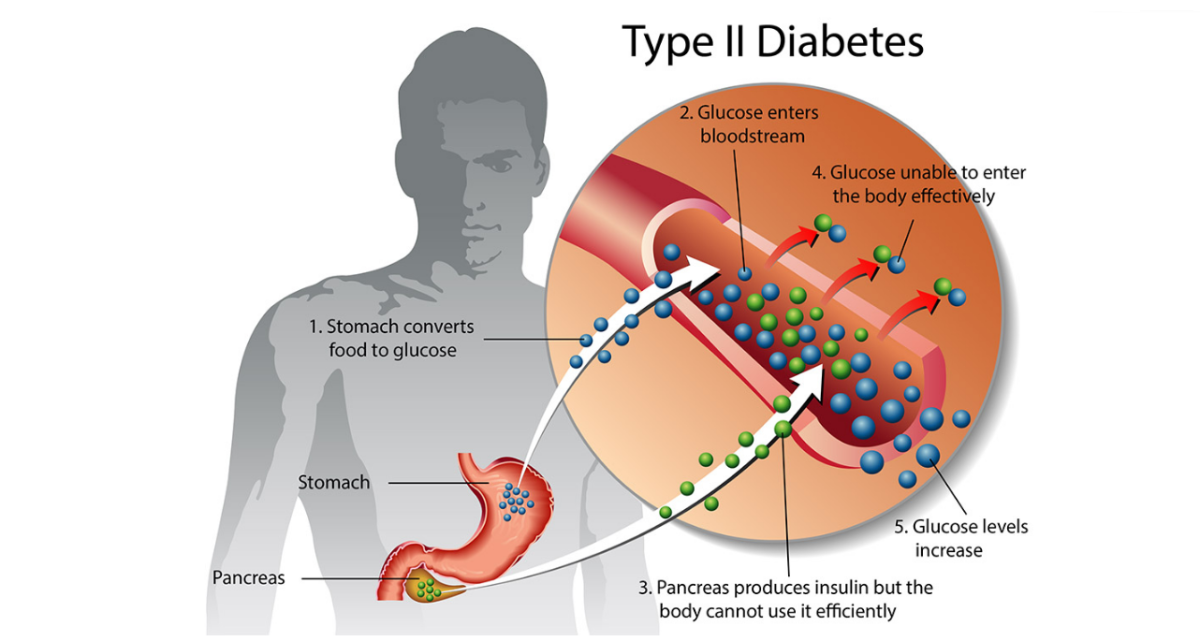
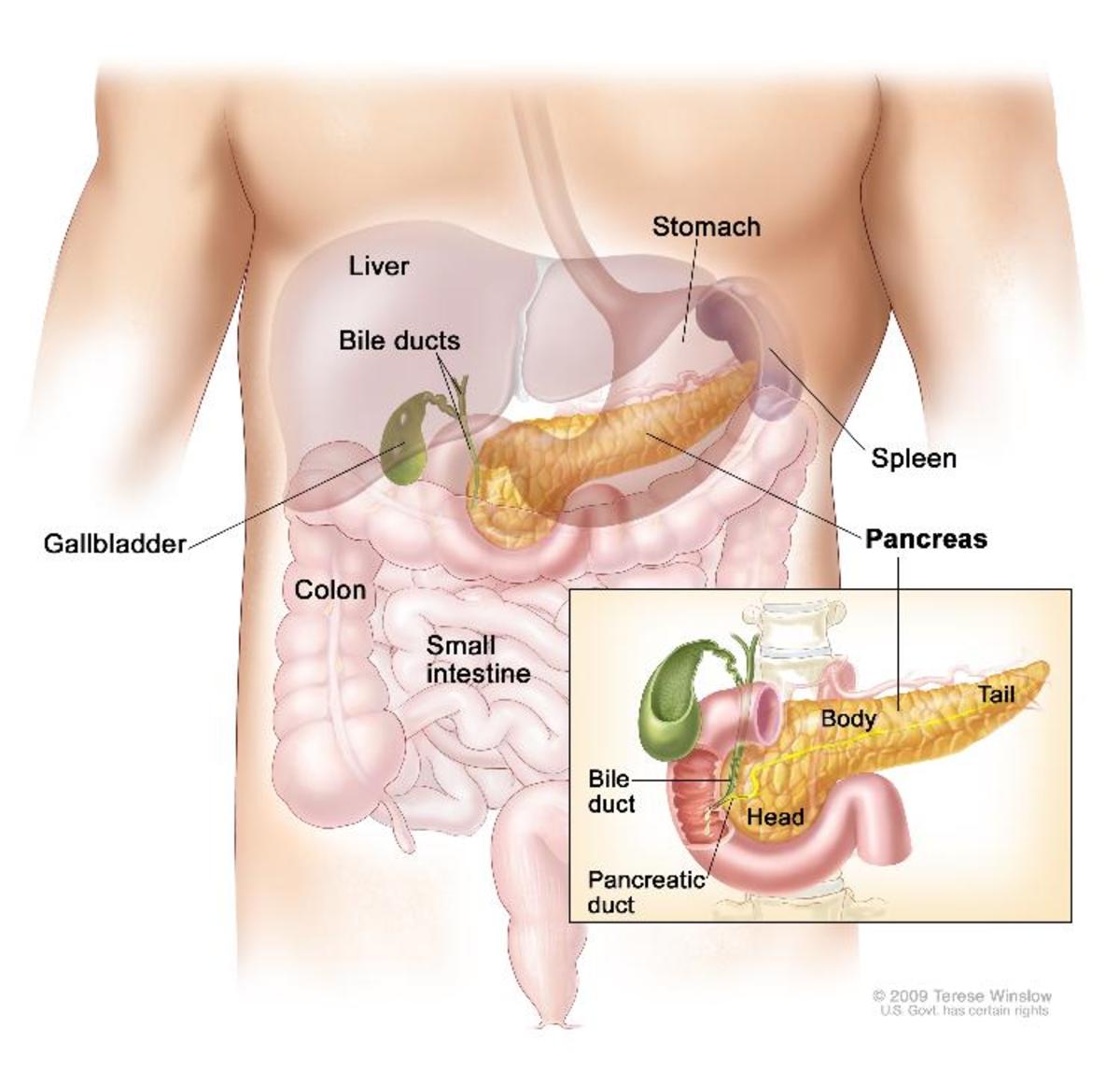
Diabetes mellitus habitually referred to as diabetes is caused by a decline in insulin secretion by the cells of the pancreatic islet resulting into a surge in blood glucose concentration, a condition known as hyperglycemia. Diabetes insipidus is a disorder defined by the secretion of huge quantities of highly diluted urine; this is regardless if a reduction in fluid intake. This is as a result of a deficit of anti-diuretic hormone (ADH) also known as vasopressin produced by the posterior pituitary gland. The disorder diabetes mellitus is associated with extreme reduction in weight, a higher urge for urination, also known as polyuria, higher levels of thirst (polydipsia) and an extreme craving to eat known as polyphagia. This disorder has been categorized as Type 1 or insulin dependent diabetes and Type 2 or non-insulin dependent diabetes another type is Gestational diabetes. The Type 1 diabetes mellitus is portrayed by a deficiency of the insulin-secreting beta cells of the islets of Langerhans found in the pancreas; this leads to a scarcity of insulin. The principal cause for this deficiency of beta cells is a T-cell mediated autoimmune onslaught. In children, Type1 diabetes is known as juvenile diabetes. The Type 2 diabetes mellitus is as a result of insulin resistance or diminished insulin sensitivity coupled to a reduction in insulin production.
The faulty receptiveness of body tissues to insulin normally entails the insulin receptor found in cell membranes. The other type, gestational diabetes happens in women who have not been diagnosed with diabetes before and show elevated blood glucose amounts during gestation. There is no explicit reason that has been acknowledged, but it is assumed that the hormones secreted during gestation decrease a woman's receptivity to insulin, thus, elevated blood sugar amounts. Management of the levels of blood sugar via oral medications, tailored dietary sugar ingestion, insulin therapy and physical exercise have been recommended for the management of Type 1 diabetes mellitus. Research involving nano-medicine in the recent years has been intended for the use of nano-particles for the treatment of Type 1 diabetes mellitus via efficient insulin delivery.
Management of Type 2 diabetes normally involves thorough medical rehabilitation and a methodological approach; these include initial lifestyle adjustments, the incorporation of oral anti-diabetic drugs (OADs) as well as the addition of insulin. On a positive note, early instructive intercession can stop or even overturn disease development. For instance, Tuomilehto et al. and the Diabetes Prevention Program Research Group explored the consequence of rigorous nutrition and exercise therapy on the development of impaired glucose tolerance to Type 2 diabetes (1). There was a 58% virtual decrease in the development to diabetes in both studies correlated with regular diet and exercise regimen. Regimens that encourage an improved diet and an increasingly dynamic lifestyle can be very triumphant in enabling patients to accomplish weight reduction in the short term. The patients initiated in such regimens drop roughly 10% of their total body weight in approximately 20 to 26 weeks; this can offer considerable decline in hemoglobin A1c(HbA1c) amounts (approximately 1% to 2%) plus enhancement in cardiovascular disease peril factors (1).
On the flip side, this method single-handedly fails to attain sufficient glycemic therapy in a year for the bulk of patients. Modifying lifelong habits is exceptionally hard for patients, a good number will eventually need pharmacotherapy to re-establish normoglycemia (1). In spite of the capacity of lifestyle alterations as well as timely intervention to cut short disease development, the bulk of patients presently have deprived glycemic management, with fewer than 50% attaining the suggested American Diabetes Association (ADA) objective of HbA1c amounts lesser than 7%. The accomplishment of OADs is restricted by their mode of action; these habitually tackle the symptoms of diabetes instead of its fundamental pathophysiology. OADs can also include adverse side effects. For example, up to 2.5% and 17.5% of patients treated with the sulfonylurea (SU) encounter main and trivial hypoglycemia, respectively, whereas GI troubles concern up to 63% of metformin-, 36% of thiazolidinedione(TZD)-, and 30% of acarbose-managed patients. Marginal edema is seen in up to 26% of TZD-treated patients moreover body weight augmentation of 1 to 5 kg is widespread with both SU and TZD treatment (1). These side effects may encompass negative repercussions on patient observance to therapy, thus higher HbA1c amounts and augmented threats for hospitalization and mortality for all causes.
Presently, insulin treatment is the lone medication with the confirmed capacity to convey any patient to the glycemic objective at any position in the development of the ailment. It is normally given after OADs have become futile, and unfortunately regularly soon after than is perfect. Glucose is the most important catalyst of insulin production; glucose amounts in healthy persons are kept within moderately constricted confines. The physiological plasma insulin outline in healthy persons exhibit little but steady insulin amounts in fasting circumstances, with jagged prandial peaks curtly (in 30 minutes) following meals followed by a gradual come back to basal levels when amplified insulin production is no longer essential. To steer clear of glycemic digressions, exogenously incorporated insulin would preferably strongly imitate the vigorous physiological pharmacokinetic insulin outline.
Direct limitations.
Regrettably, usual human insulin is linked to numerous differences that inhibit its capacity. Unchanged human insulin introduced intravenously possesses a half-life of 17-minute plus a short period of action. On the other hand, when subcutaneously introduced for mealtime management, human insulin has a gradual commencement of an action and a lingering effect in comparison with endogenous insulin. However, it ought to be injected 30 to 60 minutes before the meal so as to evade postprandial hyperglycemia as well as between-meal hypoglycemia. Moderately-acting insulin, such as neutral protamine Hagedorn [NPH] utilized for basal glucose management has distinct non-physiological spikes in serum levels 4 to 8 hours following injection. This inability to imitate the physiological insulin outline habitually results in a disparity between blood glucose and insulin spikes, prejudicing the patient to hypoglycemia. Night-time hypoglycemia is of increasing concern, given that patients are not likely to be familiar with the warning symptoms or rouse during an episode and are consequently implausible to take any pre-emptive action.
Two randomized studies comparing NPH insulin to the insulin equivalents; insulin glargine and insulin detemir, correspondingly, have shown the higher frequency of overall and night-time hypoglycemia related to the non-physiological outline of human insulin. During these trials, the amount of hypoglycemic and night-time hypoglycemic occurrences, respectively, for each patient-year were 16.0 and 3.3 using NPH insulin against 8.6 and 1.5 using insulin detemir (together P < .001), plus 17.7 plus 6.9 using NPH insulin against 13.9 and 4.0 using insulin glargine (P < .02 plus P < .001, in that order). The elevated frequency of hypoglycemia related to NPH insulin is a lucid drawback of this therapy because hypoglycemia is an acknowledged obstacle to attaining glycemic objectives. Insulin treatment is habitually shadowed by weight increases. Hermansen et al. of late evaluated 7 tests of a duration of 20 to 52 weeks in which fundamental insulin equivalents; insulin glargine, insulin detemiror, and NPH insulin were supplemented to the current OAD treatments of insulin-ignorant patients suffering from type 2 diabetes (2). In all the instances following insulin commencement, weight increment was seen, with documented weight increments at the conclusion of the tests varying from 2.0 to 3.9 kg in the instance of insulin glargine, 0.7 to 1.2 kg in the case of insulin detemir along with 1.6 to 2.9 kg in the case of NPH insulin (2). Patients and health care providers identified weight gain as a general alarm preceding insulin commencement, plus are one of the main aspects to blame for the resistance of patients to insulin amplification (2). Lastly, the conventional vial and syringe technique of insulin introduction is linked to instances of stigma, needle avoidance, and nonadherence to regimens.
Vanadium is known to have a variety of roles in the natural systems; it has been found in various compounds in nature (3). In human beings, V compounds show a broad range of pharmacological characteristics, and several complexes have been tried as spermicidal, antiviral, anti-tuberculosis, anti-HIV and anti-parasitic properties. It is also among the most efficient metal ions with a capacity of anti-diabetic properties. As a result of this insulin mimicking effect, vanadium has found a major application in the management of patients suffering from type 2 diabetes mellitus (4). A group of complexes that shows potential consists of impartial VIVO complexes with bidentate anionic ligands L−(L is also named organic carrier) with composition VOL2. Bis(maltolato)oxidovanadium(IV) (or BMOV) became the standard compound for the novel molecules with antidiabetic properties, its derivative bis(ethylmaltolato)oxidovanadium(IV) (or BEOV) got to the second phase of the clinical trials.
Researcher’ Approach
The researchers investigated the interaction of the VIVO2+ ion with hemoglobin. This interaction was probed with the combined use of spectroscopic, spectro-photometric and computational methods. Binding of Hb to VIVO2+ in vitro was proven, three unspecific sites (named α, β, and γ) were determined, with the likely synchronization of His−N, Asp−O−, and Glu−O− donors. The significance of log β for (VO)Hb was found to be 10.4, considerably lesser than that for human serum apo-transferrin (hTf). In the systems with VIVO probable antidiabetic compounds, assorted classes cis-VOL2(Hb) (L =maltolate (ma), 1,2-dimethyl-3-hydroxy-4(1H)-pyridinonate (dhp)) were seen with the central binding of an available His residue, where no ternary complexes are seen with acetylacetonate (acac). The relationship was probed by using human serum bought from Sigma. Blood samples were also obtained from consenting adults and processed. An EPR spectra were recorded in an X-band at 9.4 GHz. This was done using a Bruker EMX spectrophotometer, coupled to an HP 53150A microwave frequency counter. Spectrophotometric measurements were done using a Perkin-Elmer Lambda 35 spectrophotometer. The binding of Hb to VIVO2+ was done using DFT simulations with the Gaussian 09 software.
Observations
The study was undertaken with the previous studies on the interaction between VIVO2+ and blood serum proteins as a template. The experiment proved that the VIVO2+ was mainly attached to hemoglobin. EPR noted on frozen solutions containing VIVO2+ and hemoglobin showed that hemoglobin can coordinate this ion. The signal associated with Fe2+ is only found in the low field area; it does not interfere with the resonances of the premier z component of the anisotrophic spectrum of VIVO2+ ion. The highest intensity of the EPR signals was obtained when the concentration of VIVO2+ was approximately twice that of hemoglobin; this shows that approximately two or three binding sites are available for metal coordination.
From the results obtained in the EPR spectra, it can be seen that there are two types of signals, this depends on the VIVO2+ to hemoglobin ratio. The MI = 7/2 resonance seems to be great and asymmetric it can explain the existence of at least two VIVO2+ classes with dissimilar synchronization mode plus an Az of 166.8 and 163.3 × 10−4 cm−1. However, it cannot be disregarded that, below the big band centered in the region of 408 mT, there are resonances belonging to other sites; consequently, in correspondence to what was lately reported for albumin, the stoichiometry of the binary complex VIVO2+−Hb will be shown with (VO)xHb, x = 2−3. The two sites visible by EPR at a pH of 7.4 were named; β and γ. By an evaluation of the four spectra reported previously, it can also be reasoned that, by escalating the metal to ligand proportion, the MI = 7/2 resonances are placed under a slight shift towards elevated magnetic field values, signifying the occurrence of a set of fragile donor atoms. The spectral limits are reported subsequently. On the grounds of the standards of the 51V hyperfine pairing constant on the z-axis (Az), the synchronization of VIVO2+ in the precise iron sites, that is, the heme cluster, can be removed.
What Researchers Accomplished
The results of this research revealed that hemoglobin attaches VIVO2+ ion at physiological pH in at least two positions of analogous strength named β and γ, resulting into a species; (VO)xHb with x = 2−3. It is probable that, in agreement with what was accounted for serum proteins like HSA and IgG, such positions are not detailed for VIVO2+ and entail His-N and Glu/Asp-COO− donors as shown by the DFT analysis. The value of the constancy constant for (VO)Hb (log β = 10.4) was considerably lesser than that for hTf (log β1 = 13.0); this was as a result of the coordination of VIVO2+ to the iron-specific locations in apo-transferrin. Log β for (VO)Hb was analogous with that of IgG(log β = 10.3) and was elevated than that calculated for HSA (log β = 9.1). Consequently, the strength arrangement of the blood proteins for the VIVO2+ ion is hTf ≫ Hb ≈ IgG > HSA. The ternary systems encompassing hemoglobin and three probable antidiabetic compounds produced by maltol, 1,2-dimethyl-3-hydroxy-4(1H)-pyridinone, in addition to acetylacetone, [VO(ma)2], [VO(dhp)2], and [VO(acac)2], illustrate similarities with what was available for the equivalent systems with hTf, HSA, and IgG.14a−d. At physiological pH, cisoctahedral complexes in which Hb attaches VIVO2+ via an imidazole nitrogen of an available His residue, are produced. For cis-VO(ma)2(Hb) and cis-VO(dhp)2(Hb), log β values of 19.6 and 25.8 were calculated, analogous with those of the similar species of apo-transferrin, albumin, and immunoglobulin G, cis-VO(carrier)2(hTf), cis-VO(carrier)2(HSA), and cis-VO- (carrier)2(IgG), where carrier is ma or dhp. No development of ternary groups is seen in the system with Hacac; this can be elaborated, in conformity with what was shown in the literature, with the geometry in the solution of [VO(acac)2], square pyramidal, that inhibits the equatorial harmonization of the protein donors. The testing of uptake of [VO(ma)2], [VO(dhp)2], and [VO(acac)2] by the red blood cells prove that these impartial compounds infiltrate the erythrocyte membrane via passive diffusion (5) . Percentage quantities more than 50% are found in the intracellular medium. Some the VIVO2+ groups produced inside the red blood cells were characterized. [VO(dhp)2] changes quantitatively in cis-VO(dhp)2(Hb); [VO(ma)2] in cis-VO(ma)2(Hb) and in cis-VO(ma)2(Cys-S−) with the mid synchronization of a thiolate S− coming from GSH or, most certainly, from a membrane protein; and lastly [VO(acac)2] yields (VO)xHb and two unrecognized complexes VO(L1,L2) and VO(L3,L4), where L1, L2, L3, and L4 are red blood cell bioligands like cytosol or membrane proteins.
The outcome of this study shows that the relation with the red blood cells cannot be ignored when the conveyance or the pharmacological acts of a V compound is taken into consideration and that an allotment between the erythrocytes and plasma is achieved. The proportion of such a distribution is pegged on the potency of the carrier ligand and the structural characteristics of the complex at physiological settings (square pyramidal or cis-octahedral geometry). These outcomes are in harmony with the methodical determinations of V in the biological fluids, exemplified by bigger values in the blood than in the serum. Current data also proposes that V pools other than the entire serum are probably linked to the antidiabetic accomplishment of the vanadium complexes. As a consequence of the uptake by the red blood cells, a substantial fraction of VIVO complexes is moved in the blood by erythrocytes, in this structure, it arrives at the intended organs. In contrast, the biotransformation in the cytosol can partially or fully neutralize the particular VIVO2+ treatment. This has to be taken into consideration in the future in the research relating to the conveyance and the exploitations of the pharmacologically dynamic V compounds. The destiny of the probable anti-diabetic compounds, after they enter and change in the erythrocytes, is not expected. In reality, it is not acknowledged if these compounds can, in the suitable circumstances, be excreted by the erythrocytes and perform their pharmacological properties or remain restricted in a dormant variety until the death of the red blood cells. In the first instance, the erythrocytes only contributed to the transportation of the anti-diabetic compounds through the blood, while in the second one they acted as a store to amass these compounds in a dormant stance. Since the differing efficacy of the V antidiabetic compounds is pegged on the quantity arriving at the target organs, the significance of establishing their allotment between blood serum and erythrocytes is obvious.
Conclusion
Lastly, it was seen that the procedure discussed in this study, specifically the biodistribution between the plasma and the erythrocytes and the ensuing biotransformation in the red blood cells with the probable interaction with hemoglobin, has to be well thought-out for all of the metal drugs and the pharmacologically active synchronization compounds (6).