Medical information and significance about Hypothyroidism, thyroiditis and Cancer of the Thyroid gland
HYPOTHYROIDISM (MYXEDEMA)
It is the characteristic reaction to thyroid hormone deficiency. The spectrum of hormone ranges from a few non – specific symptoms to overt hormone, to myxedema coma. Hypothyroidism occurs in 3 to 6 for the adult population, but is symptomatic only in a minor of them. It occurs 8 to 10 times more often in woman than in men and usually develops after the age of 30.
Classification
1.Congetial.
2. Acquired: 1. Primary (thyroid gland disturbances).
2. Secondary (due to pituitary disease).
3.Tertiary (due to hypothalamic disease).
4.Peripheral.
Etiology
A cause is usually evident from the history and physical examination.
1.Primary (thyroidal) hypothyroidism.
1) surgical removal, total thyroidectomy of thyroid carcinoma, subtotal thyroidectomy (hypothyroidism occurs from 25 to 75 of patients in different series);
2) irradiation (hypothyroidism results from external neck irradiation therapy in doses 2000 rads or more such as are used in the treatment of malignant lymphoma and laryngeal carcinoma); I131 therapy for hyperthyroidism (it results in hyperthyroidism in 20 % to 60 % of patients within the first year after therapy and in 1 % to2 % each year there after);
3) during or after therapy with propylthyouracil, methimazole, iodides or beta-blockers;
4) autoimmune processes (hypothyroidism usually occurring as a sequel to Hashimoto’s thyroiditis and results in shrunken fibroid thyroid gland with a little or no function and infiltrative diseases (tuberculosis, actynomycosis);
5) trauma;
6) iodine deficiency.
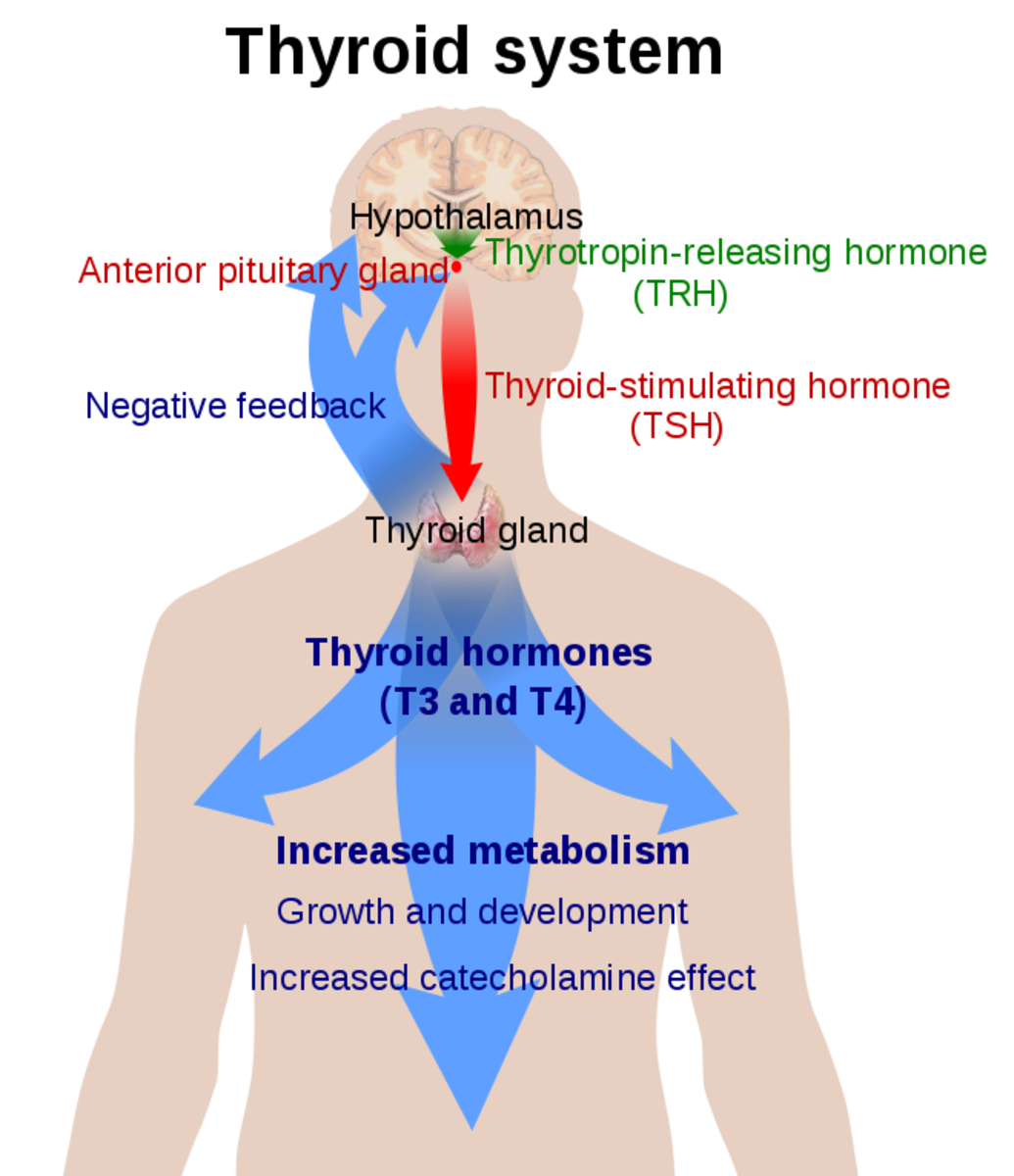
2.Secondary and tertiary hypothyroidism
It occurs due to either deficient secretion of TSH from the pituitary or lack of secretion of TRH from the hypothalamus.
- Tumor;
- Infarction;
- Infiltrative process;
- Trauma;
- Drugs (reserpin, parlodel).
3. Peripheral hypothyroidism:
- peripheral tissue resistance to thyroid hormones;
- decreasing of T4 peripheral transformation into T3 (in liver or in kidneys) ;
- production of antibodies to thyroid hormones.
Congenital:
- Maldevelopment –hypoplasia or aplasia;
- Inborn deficiencies of biosynthesis or action of thyroid hormone;
- Atypical localization of thyroid gland;
Classification. (cont.)
B. 1. Laboratory (subclinical) hypothyroidism.
2. Clinical hypothyroidism, which can be divided on stages of severity: mild, moderate, severe.
C. 1. Compensation.
2. Subcompensation.
3. Decomposition.
D. 1. Without complications.
2. With complications (myopathy, polyneuroparhy, encephalopathy, coma).
Clinical featuresThe major symptoms and signs of hypothyroidism reflect showing of physiologic function. Virtually every organ system can be affected. The onset of symptoms may be rapid or gradual, severity varies considerably and correlates poorly with biochemical changes. Because many manifestations of hypothyroidism are non-specific, the diagnosis is particularly likely to be overlooked in patients with other chronic illnesses and elderly.
Nervous system
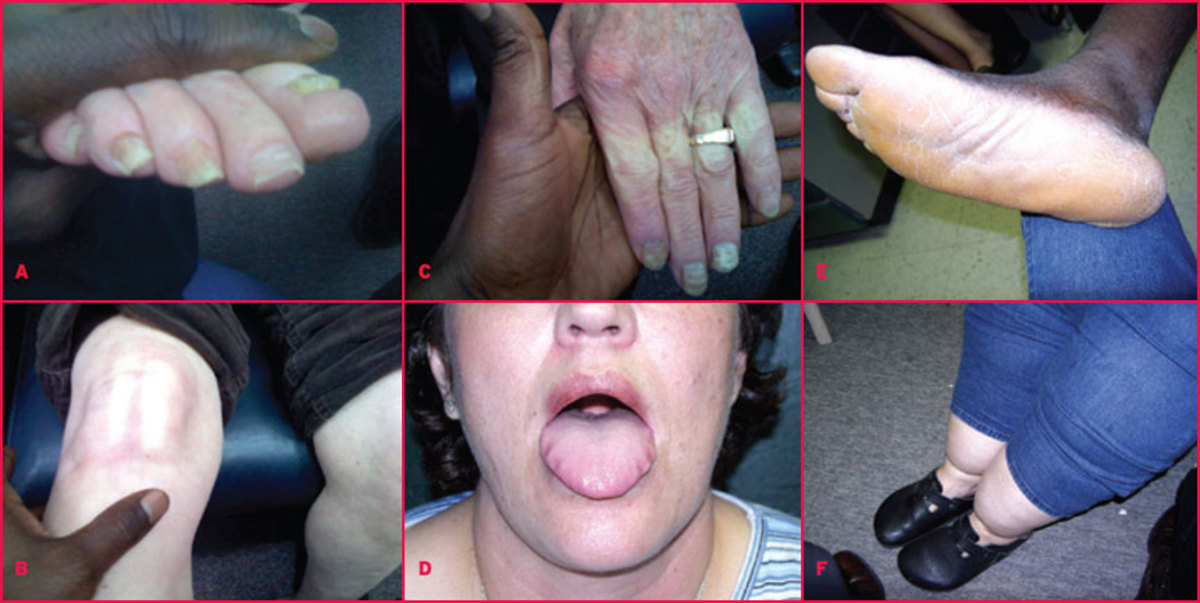
Most of hypothyroid patients complain of fatigue, loss of energy, lethargy, forgetfulness, reduced memory. Their level of physical activity decreases, and they may speak and move slowly. Mental activity declines and there is inattentiveness, decreased intellectual function, and sometimes may be depression.
Neurological symptoms include also hearing loss, parasthesias, objective neuropathy, particularly the carpal tunnel syndrome, ataxia.
Tendon reflex shows slowed or hung-up relaxation.
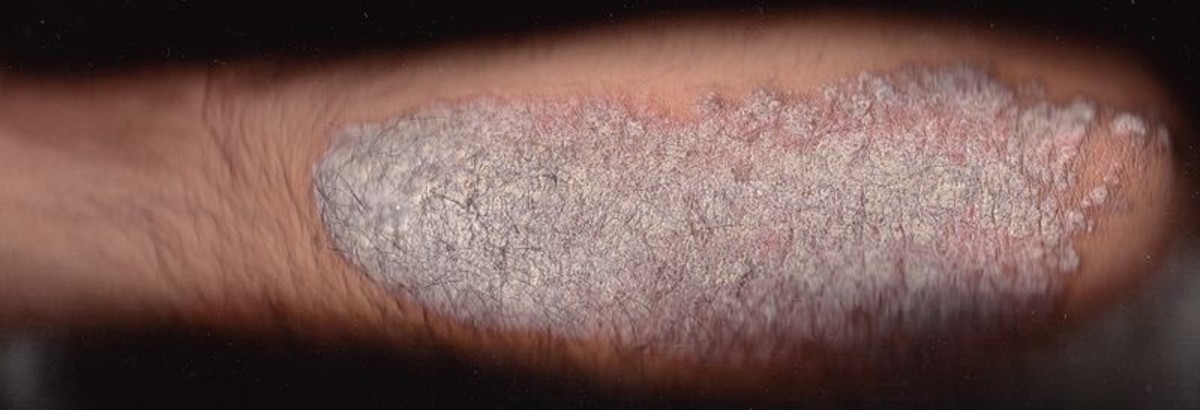
Skin and hair.Hypothyroidism results in dry, thick and silk skin, which is often cool and pale. Glycosoamynoglicanes, mainly hyaluronic acid accumulate in skin and subcutaneous tissues retaining sodium and water. So, there is nonpitting edema of the hands, feet and periorbital regions (myxedema). Pitting edema also may be present. The faces are puffy and features are coarse. Skin may be orange due to accumulation of carotene. Hair may become course and brittle, hair growth slows and hair loss may occur. Lateral eyebrows thin out and body hair is scanty.
Cardiovascular system.
There may be bradycardia, reduced cardiac output, quiet heart sounds, a flabby myocardium, pericardial effusion, cardiac wall is thick, it is increased by interstitial edema. (These findings, along with peripheral edema, may simulate congestive heart failure). Increased peripheral resistance may result in hypertension. The ECG may show low voltage and/or non-specific ST segment and T wave changes. Hypercholesterolaemia is common. Whether or not these is an increased prevalence of ischemic heart disease is controversial. Angina symptoms, when present, characteristically occur less often after the onset of hypothyroidism, probably because of decreased activity.
Gastrointestinal system.
Hypothyroidism does not cause obesity, but modest weight gain from fluid retention and fat deposition often occurs. Gastrointestinal motility is decreased loading to constipation and abdominal distension. Abdominal distension may be caused by ascities as well. Ascitic fluid, like other serous effusions in myxedema, has high protein content. Achlorhydria occurs, often associated with pernicious anemia.
Renal system.
Reduced excretion of a water load may be associated with hyponatriemia. Renal blood flow and glomerular filtration rate are reduced, but serum creatinine is normal. May be mild proteinuria and infections of urinary tract.
Respiratory system.
Dyspnea of effort is common. This complaint may be caused by enlargement of the tongue and larynx, causing upper airway obstruction, or by respiratory muscle weakness, interstitial edema of the lungs, and for plural effusions which have high protein content. Hoarseness from vocal curt enlargement often occurs.
Musculoskeletal system.
Muscle and joint aches, pains and stiffness are common. Objective myopathy and joint swelling or effusions are less often present. The relaxation phase of the tendon reflexes is prolonged. Serum creatine phosphokinase and alanine aminotransferase activities are often increased, probably as much to slowed enzyme degradation as to increased release from muscle.
Hemopoetic system.
Anemia, usually normocytic, caused by decreased red blood cell production, may occur. It is probably from decreased need of peripheral oxygen delivery rather than hematopoetic defect. Megaloblastic anemia suggests coexistent pernicious anemia. Most patients have no evidence iron, folic acid or cyancobalamin deficiency.
Endocrine system.
There may be menorrhagia (from anovulatory cycles), secondary amenorrhea, infertility and rarely galactorrhea. Hyperprolactinemia occurs because of the absence of the inhibitory effect of thyroid hormone on prolactine secretion (and causes galactorrhea and amenorrhea or Van – Vik – Cheness – Ross’s syndrome).
Pituitary-adrenal function is usually normal. Pituitary enlargement from hyperplasia of the thyrotropes occurs rarely in patients with primary hypothyroidism –such enlargement also may be caused by a primary pituitary tumor, which resulting TSH deficiency.
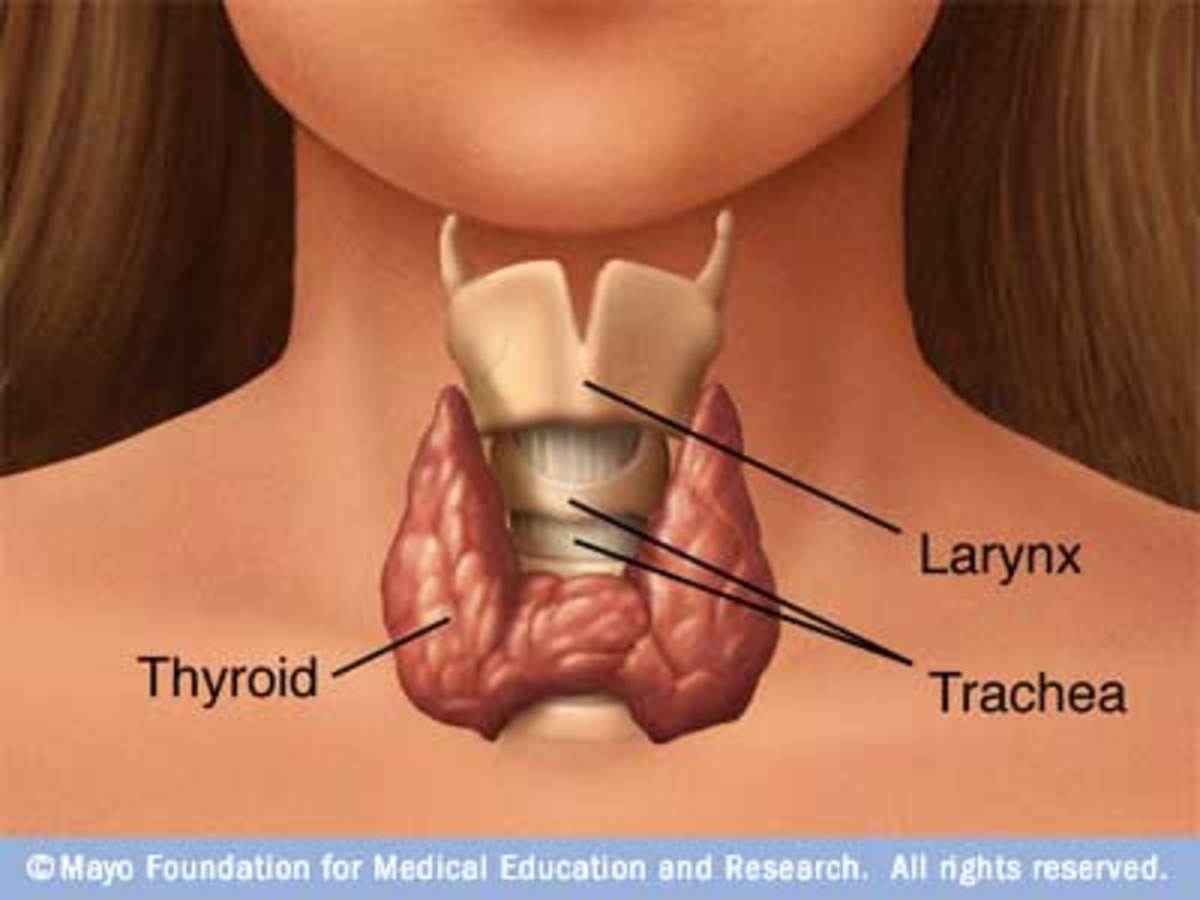
Enlargement of thyroid gland in young children with hypothyroidism suggests a biosynthetic defect. Hypothyroidism in adults is caused by Hashimoto thyroiditis.
Secretion of growth hormone is deficient because thyroid hormone is necessary for synthesis of growth hormone. Growth and development of children are retarded. Epiphyses remain open.
Metabolic system.
Hypothermia is common. Hyperlipidemia with increase of serum cholesterol and trigliceride occurs because of reduced lipoprotein lipase activity.
Subclinical (laboratory) hypothyroidism.
It is a state in which we cant find clinical features of hypothyroidism and euthyroidism is reached by compensatory increasing of TSH secretion and that’s why synthesis and secretion of such level of thyroid hormone that will be enough for organism. It is an asymptomatic state in which serum T4 and free T4 are normal, but serum TSH is elevated. The therapy may provide the patient with more energy, a feeling of well being, desirable weight reduction, improved bowel function or other signs of better health even though the patient is not aware of these symptoms before therapy.
Peculiaritis of congenital hypothyroidism
• Children are born with increased weight
• Subcutaneous edema
• Hypotermia
• Prolonged jaundice
• Physical (dwarfism) and mental retardation (cretinism
Diagnostic of hypothyroidism is based on:
1) history;
2) clinical features;
3) blood analysis: anemia; hypercholesterolemia;
4) levels of thyroid hormone: both serum T4 and T3 are decreased (but in 25% of patients with primary hypothyroidism may be normal circulating levels of T3);
5) ECG;
6) examination of tendon reflexes;
7) ultrasonic examination;
Differential diagnosis of primary and secondary hypothyroidism:
1) clinical features:
Secondary hypothyroidism is not common, but it often involves other endocrine organs affected by the hypothalamic – pituitary axis. The clue to secondary hypothyroidism is a history of amenorrhea rather than menorrhagia in a woman with known hypothyroidism.
In secondary hypothyroidism, the skin and hair are dry but not as coarse; skin depigmentation is often noted; macroglossia is not prominent; breasts are atrophic; the heart is small without accumulation of the serous effusions in the pericardial sac; blood pressure is low, and hypoglycemia is often found because of concomitant adrenal insufficiency or growth hormone deficiency.
2) laboratory evaluation:
shows a low level of circulating TSH in secondary hypothyroidism, whereas in primary hypothyroidism there is no feedback inhibition of the intact pituitary and serum levels of TSH are very high. The serum TSH is the most simple and sensitive test for the diagnosis of pituitary hypothyroidism.
Serum cholesterol is generally low in secondary hypothyroidism, but high in pituitary hypothyroidism.
Other pituitary hormones and their corresponding target tissue hormones may be low in secondary hypothyroidism.
The TSH test is useful in distinguishing between secondary and tertiary hypothyroidism in the former; TSH is not released in response to TRH; whereas in the later, TSH is released.
Treatment of hypothyroidism.
1. Diet №10.
2. Regimen is not restricted.
3. 1) replacement therapy:
- desiccated animal thyroid (this is an extract of pig and cattle thyroid glands, which standardized based on its iodine content but they are too variable in potency to be reliable and should be avoided);
- synthetic preparations of :
T4 (l-thyroxine)
- T4 is preparation of choice, because it produces stable serum levels of both T4 and T3.
- Absorption is fairly constant 90 to 95% of the dose. T3 is generated from T4 by the liver.
- The initial dosage can be 1.6 mkg/kg of ideal weight or 12.5-25 mkg in older patients and 25-50 mkg in young adult.
- The dosage can be increased in 25-50 mkg increments at 4 to 6 week intervals until clinical and biochemical euthyroidism is achieved. In older patients more gradual increments are indicated. Cautious replacement is particularly warranted in patients with ischemic heart disease, because angina pectoris or cardiac arrhythmia may be precipitated by T4 therapy.
- The average maintenance dosage is 100 to 150 mkg/day orally, only rarely is a larger dosage required. In general, the maintenance dose may decrease in the elderly and may increase in pregnancy.
- The dosage should be minimum that restores TSH levels to normal (though this criterion cannot be used in patients with secondary hypothyroidism).
- Patient takes the whole dose of T4 once a day (in the morning), in the summer the dose may be decreased and in the winter should be increased.
T3 (liothyronine sodium) should not be used alone for long-term replacement because its rapid turnover requires that it be taken. T3 is occasionally used mainly in starting therapy because the rapid excretion is useful in the initial titration of a patient with longstanding hypothyroidism in whom cardiac arrhythmia may occur early in replacement therapy. The risk of jatrogenic hyperthyroidism is therefore greater in patients receiving these preparations.
In addition, administering standard replacement amounts of T3 (25 to 50 mkg/day) results in rapidly increasing serum T3 levels to between 300 and 1000 mkg within 2 to 4 h, these levels return to normal by 24 h. Therefore, when assessing serum T3 levels in patients on this particular regimen, it is important for the physician to be aware of the time of prior administration of the hormone. Additionally, patients receiving T3 are chemically hyperthyroid for at least several hours a day and thus are exposed to greater cardiac risks. Similar patterns of serum T3 concentrations are seen when mixtures of T3 and T4 are taken orally, although the peak levels of T3 are somewhat lower. Replacement regimes with synthetic preparations of T4 reflect a different pattern of serum T3 response increases in serum T3 occur gradually over weeks, finally reaching a normal value about 8 wk. after starting therapy.
Synthetic T3 and T4 combinations (liotrix, thyreocomb). These preparations were developed before it was appreciated that T4 is converted to T3 outside of the thyroid. These preparations should not be used.
2) Symptomatic therapy:
- beta-blockers (should be used in patients with tachycardia and hypertension) in the dose of 20 – 40 - 60 mg/day;
- hypolypidemic agents;
- vitamins (A, B, E);
- diuretics and others.
Subclinical hypothyroidism
Many endocrinologists would treat such patients with T4, especially if hypercholesterolemia were present. Even in the absence of hyperlipidemia, a trial of therapy might be varianted to determine if the patient experiences improvement presumably the normal serum T4 concentrations before therapy did not reflect adequate tissue effects of thyroid hormones in such patients. Unfortunately, it is also reasonable to follow these patients without T4 therapy by surveying thyroid function at 4-to 6 months intervals to determine whether thyroid failure has occurred, as indicated by the serum T4 falling to subnormal levels along with a greater increase in serum TSH and the appearance of clear symptoms.
MYXEDEMA COMA
It is a life-threatening complication of hypothyroidism, which is extremely rare in warm climates but not uncommon in cold areas.
Precipitating factors include:
- exposure to cold;
- infection;
- trauma;
- drugs that suppress the CNS.
Myxedema coma characteristics include a background of long-standing hypothyroidism with extreme hypothermia (temperatures 24 to 32), areflexia, seizures, CO2 retention, and respiratory depression caused by decreased cerebral blood flow. Severe hypothermia may be missed unless special low reading thermometers are used. Rapid diagnosis (based on clinical judgement, history, and physical examination) is imperative because early death is likely.
Treatment of myxedema coma.
It is treated with large doses of T4 (250-500 mkg I/v bolus 3 – 4 times a day) or T3 if available (40 – 100 mkg I/v bolus 3 times a day), because TBG must be saturated before any free hormone is available for response. The maintenance dose for T4 is 50 mkg/day I/v and for T3 10 20 mkg/day I/v until the hormone can be given orally.
The patient should not be rewarmed rapidly because of the threat of cardiac arrhythmia.
Hypoxemia is common, so PaO2 should be measured at the outset of treatment. If alveolar ventilation is compromised, immediate mechanical ventilatory assistance is required.
THYROIDITIS
The various types of thyroiditis encompass a heterogeneous group of inflammatory disorders of diverse etiologies and clinical features. With all forms of thyroiditis, destruction of the normal architecture of the thyroid follicular occurs, yet each disorder has distinctive histologic characteristics. For the purposes of understanding the clinical manifestations, thyroiditis is classified according to either the severity or duration of illness using the following scheme:
1. Acute thyroiditis.
2. Subacute thyroiditis:
- subacute granulamatous thyroiditis;
- subacute lymphocytous thyroiditis.
3. Chronic thyroiditis:
- Hashimoto thyroiditis;
- Ridel struma.
4. Specific thyroiditis.
5. Thyroiditis caused by mechanical or physical factors.
Acute thyroiditis
Etiology
Acute thyroiditis it is an acute bacterial inflammation due to a bacterial pathogen, most commonly staphylococcus aureus, streptococcus hemolytica,streptococcus pneumonie, of anaerobic streptococcal organisms. Infection due to other bacterial pathogens, such as salmonella and escherichia coli have been reported, as well as fungal infections such as coccidiodomycosis. Infection occurs either secondary to hematogenous or lymphatic spread, or as a result of direct introduction of an infective agent by trauma. Persistent thyroglossal duct abnormalities have also been associated with acute thyroiditis.
Clinical features
Fever, chills and other systemic signs or symptoms of abscess formation are present. Anterior neck pain and swelling are usual, with pain occasionally radiating to the ear or mandible.
The physical examination suggests the presence of an abscess, with erythema of the skin, marked tenderness to palpation, and at times fluctuance.
Laboratory tests
Leucocytosis with a left shift, increased ESR are usually present. Thyroid hormone concentrations in blood are normal, although hyperthyroxinemia has been reported.
Treatment
Patient should be treated at surgical department. Parental antibiotics should be administered according to the specific pathogen identified. If fluctuance is present, incision and drainage might be required. Bacterial thyroiditis must be treated early and aggressively, since abscess formation can occasionally dissect downward into the mediastinum. Recurrences of the disorder are very rare.
(Duration of the treatment must be nearly 1,5-2 month).
Subacute thyroiditis
It is an acute inflammatory disease of the thyroid probably caused by a virus.
Subacute granulomatous thyroiditis (giant cell thyroiditis) SAT.
Etiology
SAT is most likely viral in origin .The specific agent responsible for the disorders is not known, although coxsackie virus, adenovirus and the mumps, echovirus, influenza and Epstein-Barr viruses have been implicated in the etiology.
A genetic predisposition is likely because of the association of HLA-BW 35 histocompatibility antigens.
Clinical features
The most common symptom is unilateral anterior neck pain, often associated with unilateral radiation of pain to the ear or mandible. Pain is often proceeded by a few weeks prodrome of myalgias, low-grade fever, malaise and sore throat. Dysphagia is also common. Symptoms of hyperthyroidism (such as tachycardia, weight loss, nervousness, and diaphoresis occur in up to 50% of patients as the disorder processes, pain can migrate to the contralateral side.
Physical examination discloses an exquisitely tender, very hard, nodular enlargement, which is most often unilateral. Tenderness is often so extreme that palpation is limited. Bilateral tenderness and goiter can occur as well. Tachycardia, a widened pulse pressure, warm skin and diaphoresis are also observed when hyperthyroidism is present.
Laboratory findings.
Early in the disease we can find an increase in T4, a decrease in RAI uptake (often 0), leucocytosis and a high ESR. After a several weeks, the T4, is decreased and the RAI uptake remains low. Full recovery is the rule; rarely, patients may become hypothyroid.
Treatment
An acute phase lasts from 4-8 weeks, during which treatment is symptomatic (aspirin 600 mg q 3-4 h, prednisolone 10-20 mg orally tid; after 1 week prednisolone can be tapered by 5 mg every 2-3 days; thus glucocorticoids are usually not required for longer than several weeks. Symptoms of hyperthyroidism are effectively controlled by the use of beta-blockers).
Following the acute phase euthyroidism is restored, and the thyroid becomes depleted of stored hormone. Patients can either remain euthyroid or progress to hypothyroid phase. It rarely lasts longer than 2-3 months, and during this phase thyroid hormone replacement in the form of levothyroxine 0,10-0,15 mg/day should be given. After several months of treatment T4 can be discontinued.
Following the hypothyroid phase recovery occurs, and the normal histologic features and secretory capacity of the thyroid are restored.
Subacute lymphocytic thyroiditis (silent thyroiditis)
A subacute disorder occurring most commonly in women , often in the postpartum period, characterized by a variable, but mild degree of thyroid enlargement, absence of thyroid tenderness, and self-limited hyperthyroid phase of several weeks to several months, often followed by transient hypothyroidism but with eventual recovery to the euthyroid state.
Etiology
Recent evidence suggests on:
1) autoimmune component (because of autoantibodies observed);
2) genetic predisposition (this is a significant prevalence of HLA-DRW3 and HLA-DRW5 histocompatibility agents);
3) viral etiology (viral antibody titers are rarely elevated);
Clinical features.
Hyperthyroid symptoms are frequent and vary from mild to normal. (Postpartum thyroiditis occur 6 weeks to 3 months after delivery).
Physical examination usually discloses a mildly enlarged, diffuse, firm, nontender goiter it has been reported that up to 50 % of patients do not have goiter.
Laboratory findings
Serum total and free T4 and T3 are elevated. Biopsies reveal lymphocytic infiltration as seen in Hashimotos thyroiditis.
Thyroid autoantibodies are positive in greater than 50 % of patients.
Treatment
Hyperthyroid phase lasts from 6 weeks to 3 month. Treatment is conservative, usually requiring only B-adrenergic blockers with propranolol.
Euthyroid interval lasts for 3-6 weeks.
During hypothyroid period (it usually lasts no longer than 2-3 months thyroid hormone supplementation with T4 0,10-0,15 mg/day may be required. Following the hypothyroid phase patients usually remain clinically euthyroid.)
Chronic thyroiditis.
Hashimoto thyroiditis (chronic lymphocytic thyroiditis) HT
Etiology
HT is an organ - specific autoimmune disorder, a chronic inflammation of the thyroid with lymphocytic infiltration of the gland generally though to be caused by autoimmune factors.
It is mire prevalent (8:1) in woman than men and is most frequent between the ages of 30 and 50 . A family history of thyroid disorders is common, and incidence is increased in patients with chromosomal disorders, including Turners, Down and Klinefelters syndromes. Histologic studies reveal extensive infiltration of lymphocytes in the thyroid.
The basic defect underlying this disease suggests an abnormality in suppressor T lymphocytes that allows helper T lymphocytes to interact with specific antigens directed against the thyroid cell. A genetic predisposition is suggested because of the frequent occurrence of the HLA- DR5 histocompatibility antigen in patients with HT.
Clinical features
HT is characterized by a wide spectrum of clinical features, ranging from no symptoms and the presence of small goiter to frank myxedema.
Occasionally patients complain of a vague sensation of tightness in the area of the anterior neck or mild dysphagia. In general, however, thyroid enlargement is insidious and asymptomatic. Symptoms of hypothyroidism may or may not be present, depending on the presence or absence of biochemical hypothyroidism.
Physical examination usually discloses a symmetrically enlarged, very firm goiter, a smooth or knobby consistency is common. Occasionally patients present with a single thyroid nodule.
A small group of patients have a form of HT termed primary idiopathic hypothyroidism, goiter is usually absent in this group.(atrophic form of HT).
Yet a small subset of patients(probably 2-4%) present with hyperthyroidism and have so-called hashitoxicosis (hypertrophy from of HT).
Laboratory findings
1) early in the disease a normal T and high titers of antithyroid (antimicrosomal) antibodies, peroxidase antibodies can be detected. Late in the disease, the patient develops hypothyroidism with a decreased in T and antibodies in this stage are usually no longer detectable;
2) the thyroid scan typically shows a irregular pattern of iodine uptake;
3) fine-needle biopsy of the nodule or enlarging area should be done to rule out a coexistent neoplasm.
Treatment
1) treatment of HT requires lifelong replacement with thyroid hormone to correct and prevent hypothyroidism. The average oral replacement dose with l-thyroxine is 100 to 150 mkg/day;
2) glucocorticoids have been reported to be effective in HT when true is a rapidly enlarging goiter associating with pressure symptoms;
3) symptomatic therapy
Ridel thyroiditis
Etiology
This extremely rare inflammatory disorder is of uncertain etiology, and earlier suggestions that it might be a fibroid variant of HT have not been substained.
Clinical features
Clinically, Ridel thyroiditis presents with pressure symptoms, and on examination an extremely hard , immobile thyroid gland is palpated The thyroid can be uniformly enlarged, or only one lobe might be affected. The disorder can be associated with other focal sclerosing symptoms, including retroperitoneal and mediastenal fibrosis and ascending cholecystitis.
Laboratory findings
1. Thyroid function tests show hypothyroidism in approximately 25 % of patients.
2. Thyroid antibodies are usually negative.
3. The thyroid scan shows decreased uptake in involved areas.
Treatment
- is surgical for those patients in whom symptoms of obstruction occur.
- Thyroid hormone is required for treatment of hypothyroidism, but thyroid hormone alone will not result in goiter shrinkage.
References
1. The Merck Manual of Diagnosis and Therapy (fourteenth Edition)/ Robert Berkow and others. – published by Merck Sharp & Donhme Research Laboratories, 1982. – P. , 997 – 1001. 1006 – 1010.
2. Manual of Endocrinology and Metabolism (Second Edition)/ Norman Lavin. – Little, Brown and Company.- Boston-New York-Toronto-London, 1994. - P. 357 - 380.
3. Endocrinology (A Logical Approach for Clinicians (Second Edition)). William Jubiz.-New York: WC Graw-Hill Book, 1985. - P. 68 – 100. 316 - 376.
4. Endocrine secrets/ (fourth Edition)Michael T. Mc Dermott/ P. 300 - 314.
Contributors







THYROID CANCER
The thyroid consists predominantly of follicular epithelial cells, which incorporate iodine into thyroid hormone to be stored in follicles, and of smaller numbers of parafollicular cells, which produce calcitonin (CT). Malignant transformation of either type of cell may occur, but the parafollicular malignancy (medullary carcinoma of the thyroid [MCT]) is much less common than cancers derived from follicular epithelial cells. Malignancies originating from follicular epithelial cells are designated according to their microscopic appearance and include papillary, follicular, and anaplasticcarcinomas
Papillary carcinoma and its variants comprise approximately 60-80% of thyroid cancers, whereasfollicular carcinoma makes up approximately 15-30% of primary thyroid malignancies. These two forms are frequently referred to as the differentiated thyroid carcinomas (DTCs). MCT accounts for 2-10% of thyroid carcinomas, whereas anaplastic forms of thyroid carcinoma account for 1-10%.
Papillary and follicular carcinomas are histologically distinct. Papillary carcinoma is generally an unencapsulated tumor marked by enlarged cells with dense cytoplasm and overlapping nuclei that have granular, powdery chromatin, nucleoli, and pseudonuclear inclusion bodies (often called "Orphan Annie eyes"), all arranged in papillary fronds. Follicular carcinoma is generally characterized by atypical-appearing thyroid cells with dense, uniform, overlapping nuclei, and a disorganized microfollicular architecture.
Papillary and follicular carcinomas behave as clinically distinct entities. Most endocrinologists consider follicular carcinoma to be the more aggressive of the differentiated cancers, with a higher rate of metastases, more frequent recurrence after therapy, and an exaggerated mortality rate compared with the relatively indolent papillary carcinoma. This view is not universal. Some authors believe that the sharp dichotomy between the clinical courses of papillary and follicular carcinomas is artificial and attribute the apparent aggressiveness of follicular carcinoma to its occurrence in an older population; they argue that when cases are controlled for age, outcomes of patients with either form of DTC are comparable,
Papillary carcinoma usually presents as a painless nodule within the thyroid gland or cervical lymphatics. The primary tumor is rarely encapsulated (4-22% in most series) but is less aggressive if a capsule is present. Papillary carcinoma is more commonly multifocal within the thyroid than is follicular carcinoma; 20-80% of glands have multiple lesions at resection. Extrathyroidal invasion through the capsule of the thyroid occurs in 5-16% of cases. Compared with other malignancies, papillary carcinoma is relatively indolent. Cancer-related death occurs in only 4-12% of patients during 20-year follow-up. Prognostic factors at the time of diagnosis that augur a poor outcome include male sex, age > 40 years, extrathyroidal invasion, distant metastases, and large primary tumor (> 1.5 cm diameter).
Follicular carcinoma usually presents as an asymptomatic nodule within the thyroid, but unlike papillary carcinoma, it may present as an isolated metastatic pulmonary or osseous focus without a palpable thyroid lesion. Very rarely metastatic foci of follicular carcinoma retain hormonal synthetic capability and overproduce thyroid hormones, causing thyrotoxicosis. The tumor is nearly always encapsulated, and the degree of vascular or capsular invasiveness (minimal to extensive) is indicative of malignant potential. Follicular carcinoma is usually unifocal (< 10% multifocal). Death due to follicular carcinoma occurs in 13-59% of patients followed for 20 years. Prognostic factors at the time of initial therapy that portend a poor outcome include age greater than 50 years, male sex (in some settings), marked degree of vascular invasion, and distant metastases.
Treatment ofDTCs
Simply stated, therapy for DTC is based on surgical removal of the primary tumor and eradication of all metastatic disease with radioactive iodine (131I). Lifelong suppression of thyroid-stimulating hormone (TSH) with exogenous thyroid hormone subsequently reduces the risk of recurrence.
Opinions about the extent of initial surgical resection have been tempered by the possiblecomplications of thyroid surgery; recurrent laryngeal nerve damage with resultant hoarseness and/or iatrogenic hypoparathyroidism may occur in 1-5% of thyroid resections. Fear of complications, coupled with the relatively low mortality rate associated with DTC, has prompted some surgeons to remove only the thyroid lobe in which cancer is apparent at the time of exploration.
Patients with large or aggressive tumors, metastatic disease evident during surgery, or extrathyroidal lesions visible on postsurgical whole-body scans usually receive 100-200 mCi of 131I in an attempt to eradicate the malignancy. These "large" doses of radioiodine have traditionally been administered only in an approved inpatient facility under the auspices of the Nuclear Regulatory Commission (NRC). Patients remain isolated until ambient levels of radioactivity fall to acceptable levels. Radionuclide is excreted renally, but significant amounts are also present in saliva and sweat. Such wastes must be disposed of appropriately. Recently, the NRC has lifted the absolute requirement for inpatient administration of high-dose 131I, and it is now performed in some centers on an outpatient basis.
The early complications of 131I therapy.
Radioiodine is absorbed by the salivary glands, gastric mucosa, and thyroid tissue. Within 72 hours of oral administration of 131I, patients may experience radiation sialadenitis and transient nausea. Such symptoms are self-limited. Thyroid tissue may become edematous and tender but rarely requires corticosteroid therapy. Radioiodine, borne in the blood, causes transient, clinically insignificant suppression of the bone marrow. In some centers, dosimetry is used to determine the maximum dose of 131I that can be safely administered at one time to patients with invasive or metastatic disease.
Late complications of high-dose radioiodine therapy may include gonadal dysfunction and predisposition to nonthyroidal malignancies. Some studies have demonstrated reduced sperm counts in male patients proportional to the administered dose of 131I. Older women may experience temporary amenorrhea and reduced fertility. Two deaths from bladder cancer and three deaths from leukemia have been reported among patients treated with lifetime cumulative doses of radioiodine exceeding 1000 mCi. Most studies suggest that cumulative doses of 131I less than 700-800 mCi, given in increments of 100-200 mCi separated by 6-12 months, are not leukemogenic.
Following surgery and radioiodine therapy, all patients are placed on a large enough dose of exogenous thyroid hormone to render serum TSH levels low or undetectable. Most endocrinologists recommend detecting recurrent disease in the asymptomatic patient by annual neck palpation and serum thyroglobulin measurement. This protein, manufactured only by normal or malignant thyroid cells, should be undetectable in the serum of a patient who has undergone complete surgical and radioiodine ablation. Sensitivity of thyroglobulin measurement is enhanced if the patient is withdrawn from exogenous thyroid hormone or stimulated with recombinant TSH
Anaplastic carcinoma occurs more commonly in the elderly (peak age: 65-70 years) and affects equal numbers of males and females. These cancers may arise in preexisting DTCs (dedifferentiation), in benign nodules, or, most commonly, de novo. The minuscule number of anaplastic malignancies within large series of patients followed for decades with DTC may discredit the theory of dedifferentiation of established cancers.
KEY POINTS: THYROID CANCER
1. Papillary and follicular carcinomas comprise the DTCs. Mortality rates are low.
2. DTC is diagnosed by fine needle aspiration (FNA) of a thyroid nodule.
3. Therapy of DTC is based on surgical resection of the primary tumor and removal of all remaining thyroid tissue (bed).
4. Orally administered radioactive iodine is accumulated by thyroid tissue, ablating the thyroid bed and metastatic foci.
5. Thyroglobulin is the most sensitive tumor marker for DTC.
6. Elimination of TSH, a DTC growth factor, by suppressive doses of levothyroxine is the most important therapeutic intervention
The type of histologic variant does not appear to affect outcome; prognosis is dismal in most cases. Surgical extirpation has been combined with external beam irradiation (4500-6000 cGy) or chemotherapy (usually doxorubicin or paclitaxel) in an attempt to eradicate the malignancy. Despite vigorous therapy, average survival is approximately 6-8 months.