The Once a Week Anti-Diabetic Drug
Semaglutide – The Once a Week Anti-Diabetic Drug
The new endorsement by the US Food and Drug Administration (FDA) to Novo Nordisk’s Semaglutide as once a week treatment for type 2 diabetes control was granted on October the 18th after the FDA advisory panel formed by 17 endocrinologist and Metabolic Drugs experts voted 16 in favor of its approval with one abstention.
After considering all the data and clinical trials conducted by the pharmaceutical company on more than 8000 adult patients with type II diabetes, including a two years long study of 3297 patient trials taking the drug for cardiovascular side effects. The FDA advisory panel found enough supporting evidence of the Semaglutide blood sugar lowering effect and its safety. The blood sugar lowering effect was measured by the record of the patients HbA1c or glycosylated hemoglobin, which is the gold-standard test for long-term sugar blood level measurement.
The HbA1c levels were 1.5 to 1.8% lower among the patients’ group receiving the drug compared with the others groups, who were either receiving a placebo or one of the competitors approved drugs. The 1.5 to 1.8% lower HbA1c on the patients treated with Novo Semaglutide was present even when compared with patients treated with Bydureon (exenatide) the AstraZeneca competing long-acting GLP-1 agonist extended release. Additionally, to the proven blood sugar lowering effect Semaglutide was directed associated with another beneficial effect on one clinical trial, the patient studied showed a 4.5- to 6.4-kg weight loss.
What Is Semaglutide?
Semaglutide is a long-acting glucagon-like peptide-1 (GLP-1) receptor, agonist. It imitates the normal hormone secreted by the intestinal cells called Glucagon, which produces to increase insulin secretion by the pancreas’ cells in the presence of elevated blood glucose; delays gastric emptying, which decreases the blood sugar after food or meals ingestion; also, the glucagon-like peptide 1 reduces the physiological glucagon secretion.
Concisely, Semaglutide mechanism of action is similar to the naturally secreted body hormone glucagon. The drug acts on the same receptor for the hormone and produces similar response reducing the blood sugar level.
How Is Semaglutide Prescribed?
Semaglutide prescription is one per week subcutaneous injection of 0.5 or 1 mg an adjunct to diet and exercise to get or improve blood sugar control in adults with type 2 Diabetes.
Is Semaglutide Safe for Your Heart?
Semaglutide is safe for your heart.
After several studies, there was not found any side effect for heart, arteries, or veins in patients receiving the Semaglutide during the two years clinical observation period. On the contrary, Semaglutide SUSTAIN-6 Top-line Data shows a cut on cardiovascular risk among those receiving it. Therefore, the FDA considered it cardiovascular safe, and with some protective or beneficial effects, additional observation and studies will continue as part of its post-marketing surveillance.
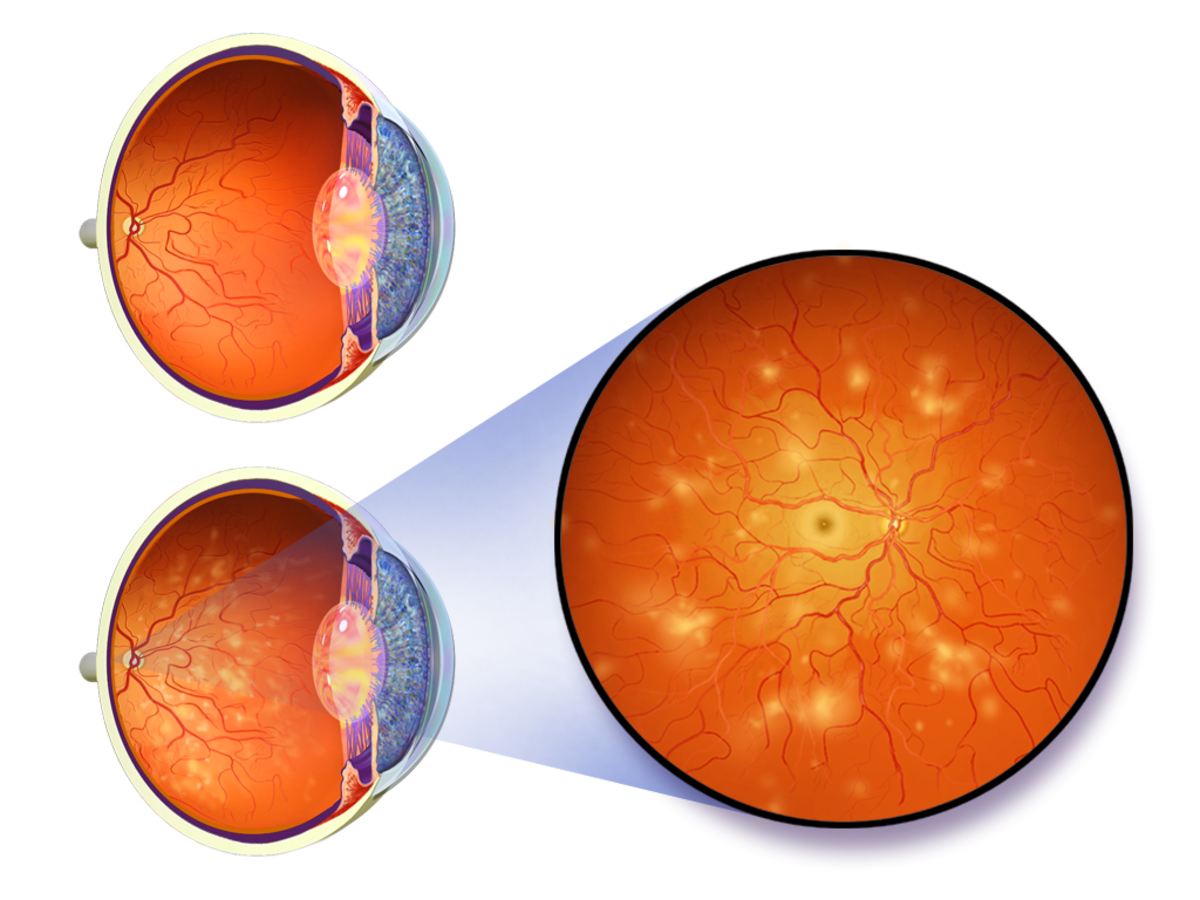
Can Semaglutide Produce Diabetic Retinopathy?
The clinical trials showed an increased report of retinopathy cases among the patients receiving Semaglutide. After a careful revision of the data from those tests, the retinopathy was found as a confounding element. The revision panel found that the patient had developed the retinopathy before they were receiving the Semaglutide, and the condition might or might not be worsening by the drug. However, the FDA demands that a safety warning must be included on the drug label indicating the risk of developing or worsening retinopathy.
The ophthalmologist members of the revision commission agreed about the primary outcomes of safety concern were met. They said that the side effects were the same as expected for similar drugs and not worrisome. The data were insufficient to determine if there is an adverse retinopathy effect o not associated with the Semaglutide treatment.
Consequently, further studies and epidemiologic vigilance are needed to establish any possible association. Other ophthalmologist consulted about it indicated that having a warning on the label about periodical eye exam on the patient receiving the drug was not only a good idea but also the best possible recommendation for the patients’ best interest.

Treatment of Type II Diabetes
The approval for patient use of the Semaglutide is welcome by the medical community and the patient with type II diabetes. Type II diabetes patients can have a drug that works helping them reducing their blood sugar level with only one single weekly injection or a single dose per week. The single dose makes it easier to comply with the medical treatment. It proved to work and lowered the blood sugar levels when used simultaneously with diet and exercise. The additional weight loss effect improves further the blood sugar level control.
Type II diabetes patients require a yearly eye examination to discard diabetic retinopathy as the standard of care. Patient receiving semaglutide needs to be warned about a possible development or worsening of diabetic retinopathy and maintain the yearly or more frequent eye examination if they already had developed a retinopathy.
This content is accurate and true to the best of the author’s knowledge and does not substitute for diagnosis, prognosis, treatment, prescription, and/or dietary advice from a licensed health professional. Drugs, supplements, and natural remedies may have dangerous side effects. If pregnant or nursing, consult with a qualified provider on an individual basis. Seek immediate help if you are experiencing a medical emergency.
© 2017 Dr Aron Mejias