The paradox of the metabolism

Less you eat, less you are hungry...
How many of you have experienced the same paradoxical effect of eating less? In diets rich in calories, the more you eat, the more often you are hungry during the day; in diets poor in calories, the less you eat, the less frequently you are hungry during the day. How is this possible?
The metabolism adjusts its speed and selects its preferred routes based on the caloric energy ratio; the stomach is more or less elastic and able to readjust to its content.
Many factors contribute to this phenomenon, not only related to the metabolism.
Hunger and satiety
Hunger pains
When hunger contractions start to occur in the stomach, they are informally referred to as hunger pains. Hunger pains usually do not begin until 12 to 24 hours after the last ingestion of food. A single hunger contraction lasts about 30 seconds, and pains continue for around 30–45 minutes, then hunger subsides for around 30–150 minutes. Individual contractions are separated at first, but are almost continuous after a certain amount of time. Emotional states (anger, joy etc.) may inhibit hunger contractions. Levels of hunger are increased by lower blood sugar levels, and are higher in diabetics. They reach their greatest intensity in three to four days and may weaken in the succeeding days, although research suggests that hunger never disappears. Hunger contractions are most intense in young, healthy people who have high degrees of gastrointestinal tonus. Periods between contractions increase with old age.
Biological mechanisms
The fluctuation of leptin and ghrelin hormone levels results in the motivation of an organism to consume food. When an organism eats, adipocytes trigger the release of leptin into the body. Increasing levels of leptin result in a reduction of one's motivation to eat. After hours of non-consumption, leptin levels drop significantly. These low levels of leptin cause the release of a secondary hormone, ghrelin, which in turn reinitiates the feeling of hunger.
Some studies have suggested that an increased production of ghrelin may enhance appetite evoked by the sight of food, while an increase in stress may also influence the hormone's production. These findings may help to explain why hunger can prevail even in stressful situations.
The state achieved when the need for food has been satisfied is called satiety. The satiety center in animals is located in the ventromedial nucleus of the hypothalamus.
Short-term regulation of hunger and food intake
Short-term regulation of hunger and food intake involves neural signals from the GI tract, blood levels of nutrients, and GI tract hormones.
Neural signals from the GI tract
One method that the brain uses to evaluate the contents of the gut is through vagal nerve fibers that carry signals between the brain and the gastrointestinal tract (GI tract). Studies have shown that through these vagal nerve fibers, the brain can sense a difference between different macronutrients. Stretch receptors work to inhibit appetite upon distention of the GI tract by sending signals along the vagus nerve afferent pathway and inhibiting the hunger center.
Nutrient signals
Blood levels of glucose, amino acids, and fatty acids provide a constant flow of information to the brain that may be linked to regulating hunger and energy intake. Nutrient signals that indicate fullness, and therefore inhibit hunger include the following:
Rising blood glucose levels
Elevated blood levels of amino acids
Blood concentrations of fatty acids
Hormone signals
The hormones insulin and cholecystokinin (CCK) are released from the GI tract during food absorption and act to suppress feeling of hunger. CCK is a key in suppressing hunger because of its role in inhibiting NPY (Neuropeptide Y). Glucagon and epinephrine levels rise during fasting and stimulate hunger. Ghrelin, a hormone produced by the stomach, is a powerful hunger stimulant.
Long-term regulation of hunger and food intake
Leptin, a hormone secreted exclusively by adipose cells in response to an increase in body fat mass, is an important component in the regulation of long term hunger and food intake. Leptin serves as the brain's indicator of the body's total energy stores. When leptin levels rise in the bloodstream they bind to receptors in ARC. The functions of leptin are to:
- Suppress the release of NPY, which in turn prevents the release of appetite enhancing orexins from the lateral hypothalamus. This decreases appetite and food intake, promoting weight loss.
- Stimulate the expression of CART.
Though rising blood levels of leptin do promote weight loss to some extent, its main role is to protect the body against weight loss in times of nutritional deprivation. Other factors also have been shown to effect long-term hunger and food intake regulation including insulin.
Set-point theories of hunger and eating
The set-point theories of hunger and eating are a group of theories developed in the 1940s and 1950s that operate under the assumption that hunger is the result of an energy deficit and that eating is a means by which energy resources are returned to their optimal level, or energy set-point. According to this assumption, a person's energy resources are thought to be at or near their set-point soon after eating, and are thought to decline after that. Once the person's energy levels fall below a certain threshold, the sensation of hunger is experienced, which is the body's way of motivating the person to eat again. The set-point assumption is a negative feedback mechanism. Two popular set-point theories include the glucostatic set-point theory and the lipostatic set-point theory.
The set-point theories of hunger and eating present a number of weaknesses.
- The current epidemic of obesity and other eating disorders undermines these theories.
- The set-point theories of hunger and eating are inconsistent with basic evolutionary pressures related to hunger and eating as they are currently understood.
- Major predictions of the set-point theories of hunger and eating have not been confirmed.
- They fail to recognize other psychological and social influences on hunger and eating.
Satiety
For years, researchers have studied satiety. While many things are known to influence satiety - including individual differences in endocrine levels from one person to another - one of the biggest factors is the type of food that you eat. Some foods fill your stomach faster and/or remain in your stomach longer, and therefor do a better job of holding off hunger.
One of the most detailed studies that demonstrate this was conducted by Suzanna Holt and her fellow researchers at the University of Sydney. The results of their study, "The Satiety Index of Common Foods", were published in the European Journal of Clinical Nutrition, September 1995. In this study, the researchers fed human test subjects fixed-Calorie portions of thirty-eight different foods, and then recorded the subjects' perceived hunger following each feeding.
The results of Holt's study, like many similar studies, indicate that satiety is most strongly related to the weight of the food consumed. In other words, the foods that weigh the most satisfy our hunger best, regardless of the number of Calories they contain. However, higher amounts of certain nutrients, such as protein and dietary fiber, also appear to improve satiety.
Mechanism of satiety
Can Satiety Be Predicted?
If there was a way of predicting satiety, we'd be able to select foods that satisfied our hunger, but contained fewer Calories. These foods would greatly improve our ability to create meals that were effective for weight loss.
Because of the strong relation between satiety and a food's weight, some researchers have recommended the consumption of foods with low Caloric densities - i.e. foods that have the fewest total Calories per gram. One of the most notable of these researchers is nutritionist Barbara Rolls, Ph.D., whose prior best-selling diet book, Volumetrics, explained her use of low Caloric density foods for weight loss. A more recent diet that makes use of low Caloric density foods is the Negative Calorie Diet. There are also many specialty diets that use a low Caloric density approach. Included among these are the cabbage soup diet and the grapefruit diet.
Unfortunately, Caloric density alone is not a reliable predictor of satiety, and it overlooks many enjoyable foods that would make wonderful additions to your diet. What you need is a better way to predict satiety.
GI and SF
A way to limit carbohydrate consumption is simply to limit the total number of Calories that you consume for each meal. This can be a very effective method for controlling blood sugar and losing body fat. Unfortunately, there's one BIG problem associated with this method - increased hunger! But what if you could eat less, and not be hungry? Is that possible?
A group of researchers from the University of Sydney in Sydney, Australia performed an interesting study in which they compared the satiating effects of different foods. These researchers, led by Suzanna Holt, include some of the same individuals that pioneered much of the work on the Glycemic Index. The result of their study, "The Satiety Index of Common Foods", was published in the European Journal of Clinical Nutrition, September 1995. In this study, the researchers fed human test subjects fixed-Calorie portions of thirty-eight different foods, and then recorded the subjects' perceived hunger following each feeding.
The results of this study clearly indicated that certain foods are much better than others for satisfying hunger. The researchers used white bread as their reference point, and arbitrarily assigned it a "Satiety Index" of 100. Foods that did a better job of satisfying hunger were given proportionately higher values, and foods that were less satisfying were assigned lower values. Among the most satisfying foods they tested were plain boiled potatoes, raw fruits, fish, and lean meats. Subjects that consumed the prescribed portion of these foods were less likely to feel hungry immediately afterward. Foods that did the poorest job of satisfying hunger included croissants, donuts, candy bars, and peanuts.
Because of the limited size of the Satiety Index study, there's some uncertainty in the accuracy of the values that were recorded for each food. However, one very important general observation was made by the Satiety Index researchers. They noted that a common feature was shared by the foods with the highest Satiety Index values. All of these foods had high weight-to-Calorie ratios. In other words, these foods contained a greater amount of bulk for each Calorie. They helped make you feel full, literally by filling your stomach.
This suspected relation between bulk and satiety may seem obvious and trivial, but it opens the door to a very powerful theory - that it may be possible to predict satiety by knowing the nutrient composition of the food! And if that is true, some form of the Satiety Index could prove to be a more flexible tool for assessing diet than the Glycemic Index.
Hedonic stimuli of food
“Over the last several years, there’s been emphasis on hedonic, rather than homeostatic mechanisms,” says Christopher Morrison of Pennington Biomedical Research Center in Baton Rouge, La. “The brain doesn’t just tell us whether we’re hungry or full. We eat for other reasons, including pleasure, and in obesity that may play an important role.”
Some of the hedonic or pleasure-oriented interest has focused on the mesolimbic dopamine system, a brain circuit that regulates incentive and reward. Activity here contributes to our motivations to seek sex, excitement and illicit drugs as well as food.
Several years ago, researchers found leptin-responsive cells in the ventral tegmental area (VTA), a major component of this reward circuit. But the effect of these cells on eating appears limited, says Martin Myers of the University of Michigan. “We think those neurons are doing something else—they’re not controlling food reward directly.”
But a new study led by Myers and reported in the journal Cell Metabolism strengthens the argument that leptin regulates appetite by making food more or less rewarding. The researchers identified a previously unrecognized group of leptin-sensitive neurons in the lateral hypothalamus (LH). Injecting leptin directly into these cells in mice, the researchers found, led to a rapid reduction in food intake and body weight—apparently by a mechanism other than controlling satiety.
The researchers found that this cluster of LH neurons connected directly to the VTA. What did they do there? Leptin in the LH, the researchers found, started a chain reaction that resulted in increased amounts of dopamine in the VTA.
This result baffled the scientists at first. “It’s different from what might have been expected,” Myers says. “There’s the general feeling that dopamine equals lust—for food, or drugs, or sex or whatever. But here, leptin increases the amount of dopamine in the reward system, while acting to decrease feeding.”
Metabolism response to Starvation
The magnitude and composition of the starvation response (i.e. metabolic adaptation) was estimated in a study of 8 individuals living in isolation in Biosphere 2 for two years. During their isolation, they gradually lost an average of 15% (range: 9–24%) of their body weight due to harsh conditions. On emerging from isolation, the eight isolated individuals were compared with a 152-person control group that initially had had similar physical characteristics. On average, the starvation response of the individuals after isolation was a 180 kcal reduction in daily total energy expenditure. 60 kcal of the starvation response was explained by a reduction in fat-free mass and fat mass. An additional 65 kcal was explained by a reduction in fidgeting, and the remaining 55 kcal was statistically insignificant.
The human starvation response is unique among animals in that human brains do not require the ingestion of glucose to function. During starvation, less than half the energy used by the brain comes from metabolized glucose. Because the human brain can use ketone bodies as major fuel sources, the body is not forced to break down skeletal muscles at a high rate, thereby maintaining both cognitive function and mobility for up to several weeks. This response is extremely important in human evolution and allowed for humans to continue to find food effectively even in the face of prolonged starvation.
Initially, the level of insulin in circulation drops and the levels of glucagon and epinephrine rise, degrading high amounts of glycogen and up regulating gluconeogenesis, lipolysis, and ketogenesis. The body’s glycogen stores are consumed in about 24 hours. In a normal 70 kg adult, only about 8,000 kilojoules of glycogen are stored in the body (mostly in the striated muscles).The body also engages in gluconeogenesis in order to convert glycerol and glucogenic amino acids into glucose for metabolism. Another adaptation is the Cori cycle, which involves shuttling lipid-derived energy in glucose to peripheral glycolytic tissues, which in turn send the lactate back to the liver for resynthesis to glucose. Because of these processes, blood glucose levels will remain relatively stable during prolonged starvation.
However, the main source of energy during prolonged starvation is derived from triglycerides. Compared to the 8,000 kilojoules of stored glycogen, lipid fuels are much richer in energy content, and a 70 kg adult will store over 400,000 kilojoules of triglycerides (mostly in adipose tissue). Triglycerides are broken down to fatty acids via lipolysis. Epinephrine precipitates lipolysis by activating protein kinase A, which phosphorylates hormone sensitive lipase (HSL) and perilipin. These enzymes, along with CGI-58 and adipose triglyceride lipase (ATGL), form complex at the surface of lipid droplets. The concerted action of ATGL and HSL liberates the first two fatty acids. Cellular monoacylglycerol lipase (MGL), liberates the final fatty acid. The remaining glycerol enters gluconeogenesis.
Fatty acids by themselves cannot be used as a direct fuel source. They must first undergo beta oxidation in the mitochondria (mostly of skeletal muscle, cardiac muscle, and liver cells). Fatty acids are transported into the mitochondria as an acyl-carnitine via the action of the enzyme CAT-1. This step controls the metabolic flux of beta oxidation. The resulting acetyl-CoA enters the TCA cycle and undergoes oxidative phosphorylation to produce ATP. Some of this ATP is invested in gluconeogenesis in order to produce more glucose.
Triglycerides and long-chain fatty acids are too hydrophobic to cross into brain cells, so the liver must convert them into short-chain fatty acids and ketone bodies through ketogenesis. The resulting ketone bodies, acetoacetate and β-hydroxybutyrate, are amphipathic and can be transported into the brain (and muscles) and broken down into acetyl-CoA for use in the TCA cycle. Acetoacetate breaks down spontaneously into acetone, and the acetone is released through the urine and lungs to produce the “acetone breath” that accompanies prolonged fasting. The brain also uses glucose during starvation, but most of the body’s glucose is allocated to the skeletal muscles and red blood cells. The cost of the brain using too much glucose is muscle loss. If the brain and muscles relied entirely on glucose, the body would lose 50% of its nitrogen content in 8–10 days.
After prolonged fasting, the body begins to degrade its own skeletal muscle. In order to keep the brain functioning, gluconeogenesis will continue to generate glucose, but glucogenic amino acids, primarily alanine, are required. These come from the skeletal muscle. Late in starvation, when blood ketone levels reach 5-7 mM, ketone use in the brain rises, while ketone use in muscles drops.
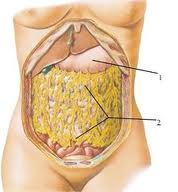
Fat distribution in the body
Remember! When you lose your weight you are getting rid of fats that are accumulated not only under your skin, but in your omentum, behind the perineum, and around the major organs in your body. The greater omentum has the potential to stock a large amount of fats and contributes enormously to the obesity state. Once you began a diet you should be aware that after cleaning your skin you must go on with your diet to eliminate the useless stocks of fats inside your body.
The Percent Daily Value (%DV)
The % Daily Values (%DVs) are based on the Daily Value recommendations for key nutrients but only for a 2,000 calorie daily diet--not 2,500 calories. You, like most people, may not know how many calories you consume in a day. But you can still use the %DV as a frame of reference whether or not you consume more or less than 2,000 calories.
The %DV helps you determine if a serving of food is high or low in a nutrient. Note: a few nutrients, like trans fat, do not have a %DV--they will be discussed later.
Each nutrient is based on 100% of the daily requirements for that nutrient (for a 2,000 calorie diet). This way you can tell high from low and know which nutrients contribute a lot, or a little, to your daily recommended allowance (upper or lower).
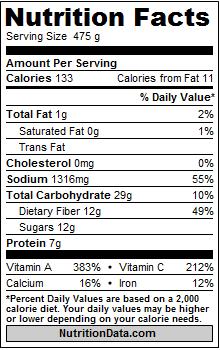
Nutrition facts label
The Nutrition Facts label is generated according to the nutrition labeling standard maintained by the FDA. You will find Nutrition Facts labels in the same format on all packaged foods sold. The panel gives information about the nutrients that the FDA has determined are most critical in the American diet. Changing the serving size in the Serving Size drop-down at the top of the page will automatically update the Nutrition Facts panel with values for the selected serving size.
New parameters for a diet
In a diet we have to know few, but extremely important parameters concerning the foods we are going to select.
We must establish the nutritional target (weight loss, gain or optimum health) and the caloric ratio of a diet.
Nutritional target
The nutritional target is based on 2 parameters: the satiating effect (Fullness Factor™) and the Nutrient density (ND Rating) of foods. Foods that are both nutritious and filling are considered better choices for weight loss. Foods that are nutritious without being filling are considered better choices for healthy weight gain. Foods that have more essential nutrients per calorie are considered better choices for optimum health.
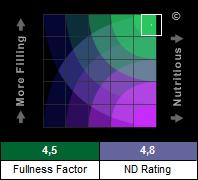
Introducing the Fullness Factor™
ND mathematically modeled the Satiety Index with a multivariate analysis that used nutrient profiles of the foods tested in the previously mentioned Satiety Index study. As anticipated, there was a good correlation between the Satiety Index values and each food's Caloric density. There were also significant but lesser correlations between the index and each food's levels of net carbohydrates, fat, dietary fiber, and protein. From the mathematical model developed, ND was able to create an equation to convert a food's nutrient profile into a predicted satiety index, which we call the Fullness Factor™.
The Fullness Factor has been normalized so that all resultant values fall into a range of 0 to 5. The calculated Fullness Factor for white bread is 1.8, so values above 1.8 indicate foods that are likely to be more satiating than white bread, and values less than 1.8 indicate foods that are likely to be less satiating. A food's Fullness Factor is independent of its serving size.
Potential Advantages of FF-based Diets over Low-Carb Diets
Diets based on the Fullness Factor have some advantages over Low-Carb diets:
- FF-based diets may better encourage the consumption of naturally healthy foods
- Because many fruits, vegetables, and less processed foods have high Fullness Factors, it may be easier to obtain essential nutrients when on FF-based diets.
- FF-based diets offer a larger range of food selections
- No foods are off limits in FF-based diets. FF-based diets simply encourage you to select the foods that cause you to fill up faster without as many total Calories.
- FF-based diets can easily accommodate a vegetarian lifestyle
While many meats are good choices for a high-FF diet, it's also relatively easy to create a high-FF diet that doesn't contain animal-based foods.
The Fullness Factor™ - A Better Predictor of Satiety
After studying the results of numerous satiety studies, NutritionData used an advanced multivariate analysis of the existing data to create a new mathematical formula that predicts satiety from the nutrient content of a given food or recipe. This formula yields a value that we call the Fullness Factor (FF):
FF values fall within the range of 0 to 5. Foods with high FF's are more likely to satisfy your hunger with fewer Calories. Foods with low FF's are less likely to satisfy your hunger.
Comparing the Fullness Factor with Existing Satiety Data
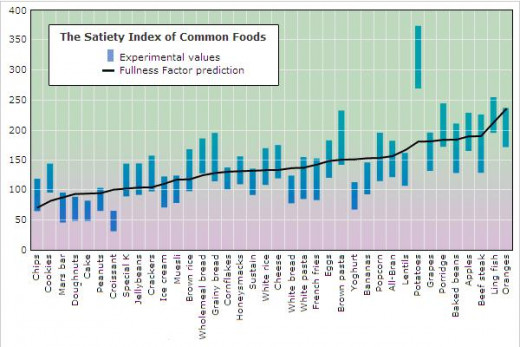
After creating this formula, ND plotted its predicted values against the experimental data taken from Holt's 1995 study. Below is a graph that shows this comparison, and following the graph is some further discussion.
Discussion
In the above graph, each blue bar represents the range of reported satiety values for each experimental food. (Approximately 12 human subjects participated in each food test, and results varied from one subject to another.) The black line represents the calculated Fullness Factors. As you can see, the Fullness Factor does a reasonably good job of predicting the satiety responses, with the possible exception of potatoes. Note, however, that the methodology of this particular study may have contributed to an artificially high satiety value for potatoes. Per the study's design, the participants were separately fed 220-Calorie servings of each food. A 220-Calorie serving of plain potatoes is larger and much less palatable than the other foods studied. The size of this serving may have influenced a repulsion to this test food that goes beyond the normal satiating response.
Limitations of the Fullness Factor
The Fullness Factor is calculated from the food's nutrient content, using values from those nutrients that have been shown experimentally to have the greatest impact on satiety. There are, however, other things that can influence a food's ability to satisfy our hunger. In particular, a food's specific taste and texture - i.e. its palatability - can encourage or discourage consumption. Palatability of a food is a highly individual and subjective value, though, that can't be accurately measured. The Fullness Factor, by design, merely provides an estimate of food satiety prior to consumption.
Benefits of the Fullness Factor
The benefits of the Fullness Factor far outweigh its limitations. The satiating effect of a food can now be more accurately predicted, solely from its nutrient content. This means that you can anticipate which foods and recipes will be most supportive of your diet.
Values of the Fullness Factor range from 0 to 5, with the Fullness Factor for white bread being 1.8. That means that for servings of equal Calories, those foods with FF's above 1.8 are more likely to fill you up than white bread, and foods with FF's below 1.8 are less likely to fill you up than white bread.
Comparing the Fullness Factor with the Glycemic Index
The Fullness Factor and the Glycemic Index are both nondimensional ratings which are used to predict your body's response to particular foods. While the Glycemic Index applies only to foods containing carbohydrates, the Fullness Factor can be used to evaluate all foods. A very detailed explanation of the Glycemic Index and Fullness Factor, as they relate to diet, can be found on ND's Glycemic Index page.

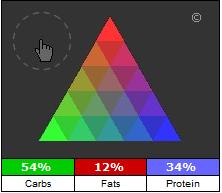
The caloric ratio
The caloric ratio is a percentage representation of the calories in a food that come from carbohydrates, fats, proteins, and alcohol.
Foods low in fat, for example, will cluster along the bottom edge of the pyramid, ranging from foods that are high in carbohydrates (at the left edge) to foods that are high in protein (at the right edge). Foods low in carbohydrates will cluster along the right edge of the pyramid, with foods that are high in fat at the upper edge and foods that are high in protein at the lower edge. Foods that have roughly the same number of calories from fats, calories, and protein will be found closer to the center of the pyramid.
How to use the Caloric Ratio Pyramid™Although there are differences of opinion over which ratios are most optimal for health and weight loss, many popular diets are based on a particular caloric ratio. For example, the USDA guidelines recommend that approximately 60% of the calories you consume should come from carbohydrates and approximately 30% from fat. Proponents of the Zone™ diet recommend a ratio (40%-30%-30%) much lower in carbohydrates but higher in protein. Advocates of the Atkins™ and South Beach™ diets recommend even lower consumption of carbohydrates, while many other diet philosophies focus on an ultralow consumption of fats.
Regardless of which diet philosophy you subscribe to, the Caloric Ratio Pyramid™ will show you how various foods and meal plans align with your dietary goals. Any recommended caloric ratio can be envisioned as a "target zone." (Please see examples below.)
Nutrients
The main nutrients in foods are three: carbohydrates, proteins and fats. Surrounding these three nutrients there are a bunch of other elements and molecules that contribute to our health: minerals, vitamins and non-vitamin antioxidants (between the most important elements we need), etc.
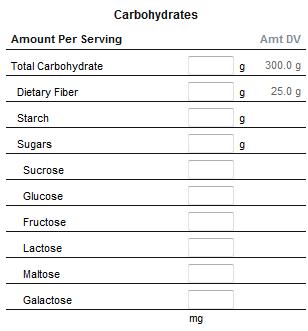
Carbohydrates
Carbohydrates are almost ubiquitous. They are macromolecule, consisting of carbon (C), hydrogen (H), and oxygen (O) atoms, with the empirical formula Cm(H2O)n (where m could be different from n). Carbohydrates are technically hydrates of carbon.
The carbohydrates (saccharides) are divided into four chemical groups: monosaccharides, disaccharides, oligosaccharides, and polysaccharides. In general, the monosaccharides and disaccharides, which are smaller (lower molecular weight) carbohydrates, are commonly referred to as sugars.
The scientific nomenclature of carbohydrates is complex; the names of the monosaccharides and disaccharides very often end in the suffix –ose (e.g., gluc-ose).
Polysaccharides have many important roles in different organisms. They are reserves of energy (e.g., glycogen), they are structural molecules (e.g., cellulose, chitin and glycosaminoglycan or mucopolysaccharides), part of the genetic macromolecules and components of coenzymes (e.g., NAD, FAD, ATP). They have many other functions within the body.
The standard usage, however, is to classify carbohydrates chemically: simple if they are sugars (monosaccharides and disaccharides) and complex if they are polysaccharides (or oligosaccharides)
Pure digestible carbohydrate, on average, yields 4 Calories of energy per gram. Foods with high carbohydrate are often highly processed or refined foods made from plants; including sweets, cookies and candy, table sugar, honey, soft drinks, breads and crackers, jams and fruit products, pastas and breakfast cereals. Unrefined foods usually contain lower amounts of carbohydrate; including beans, tubers, rice, and unrefined fruit. Foods from animals have the lowest carbohydrate but milk does contain lactose.
Carbohydrates are a common source of energy in living organisms; however, no carbohydrate is an essential nutrient in humans. Carbohydrates are not essential for the synthesis of other molecules. Humans are able to obtain 100% of their energy requirement from protein and fats.
Following a diet consisting of very low amounts of daily carbohydrate for several days will usually result in higher levels of blood ketone bodies than an isocaloric diet with similar protein content.
Long-chain fatty acids cannot cross the blood–brain barrier, but the liver can break these down to produce ketones. However the medium-chain fatty acids octanoic and heptanoic acids can cross the barrier and be used by the brain, which normally relies upon glucose for its energy. Gluconeogenesis allows humans to synthesize some glucose from specific amino acids, from the glycerol backbone in triglycerides and in some cases from fatty acids.
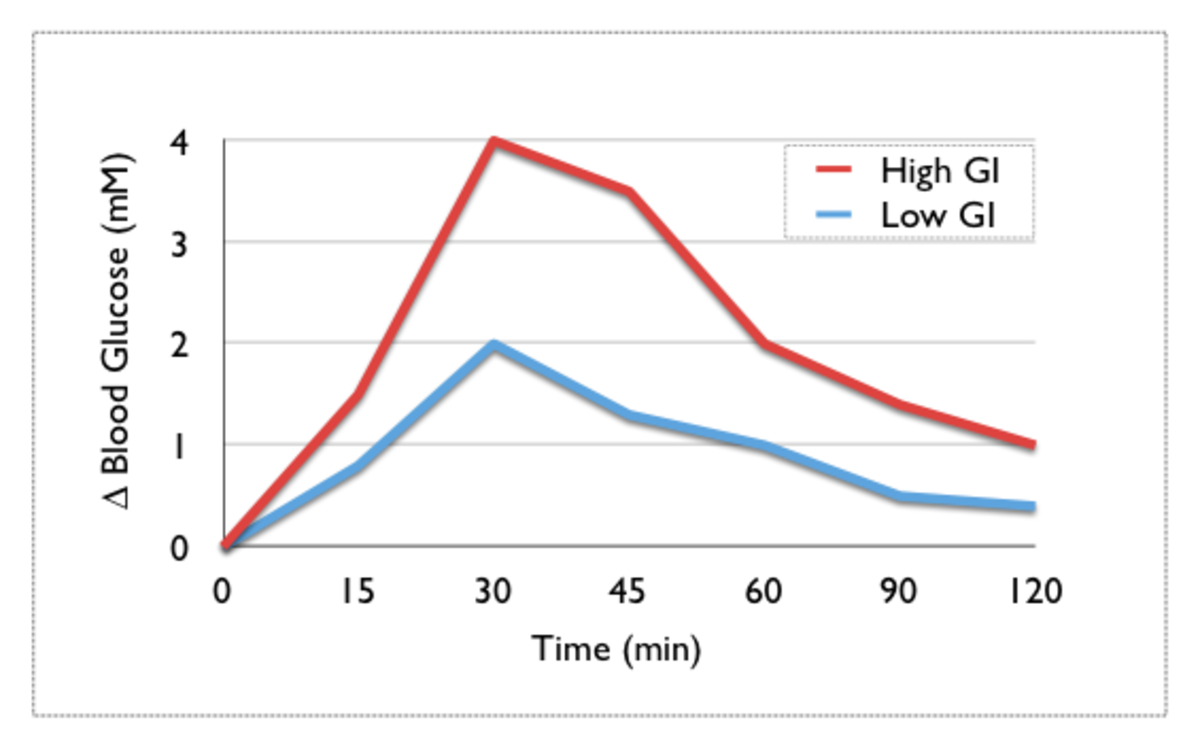
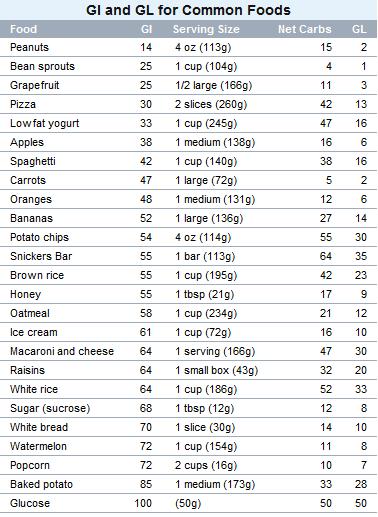
Glicemic Index
The Glycemic Index is a numerical Index that ranks carbohydrates based on their rate of glycemic response (i.e. their conversion to glucose within the human body). Glycemic Index uses a scale of 0 to 100, with higher values given to foods that cause the most rapid rise in blood sugar. Pure glucose serves as a reference point, and is given a Glycemic Index (GI) of 100.
Glycemic Index values are determined experimentally by feeding human test subjects a fixed portion of the food (after an overnight fast), and subsequently extracting and measuring samples of their blood at specific intervals of time. The earliest known work on the Glycemic Index was done by Dr. David Jenkins and associates at St. Michael's Hospital in Toronto, Canada. More recently, an effort to expand the Glycemic Index has been made by Jennie Brand-Miller and her associates at the Human Nutrition Unit of the University of Sydney in Sydney, Australia.
Nutritionists used to believe that all simple sugars digested quickly and caused a rapid rise in blood sugar, and that the opposite was true for "complex carbohydrates". But that's not always the case. While many sweet and sugary foods do have high GI's, some starchy foods like potatoes or white bread score even higher than honey or table sugar (sucrose)!
Although most candy has a relatively high Glycemic Index, eating a single piece of candy will result in a relatively small glycemic response. Why? Well, simply because your body's glycemic response is dependent on both the type and the amount of carbohydrate consumed. This concept, known as Glycemic Load, was first popularized in 1997 by Dr. Walter Willett and associates at the Harvard School of Public Health. Glycemic Load is calculated this way:
GL = GI/100 x Net Carbs
(Net Carbs are equal to the Total Carbohydrates minus Dietary Fiber)
Therefore, you can control your glycemic response by consuming low-GI foods and/or by restricting your intake of carbohydrates.
Proteins
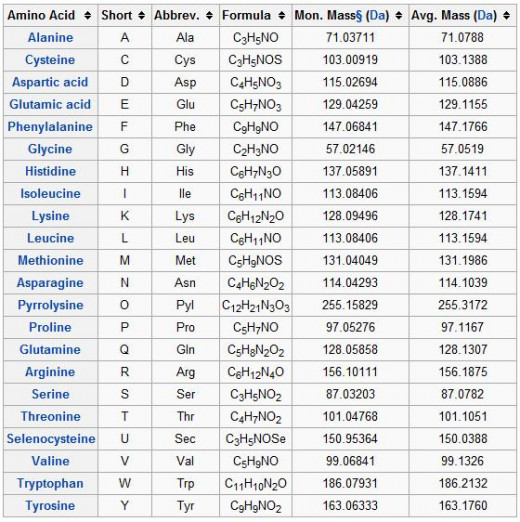
Proteins are macromolecules consisting of one or more chains of amino acid residues. Proteins perform many functions within living organisms, including catalyzing metabolic reactions, replicating DNA, responding to stimuli, and transporting molecules from one location to another. Proteins differ from one another primarily in their sequence of amino acids, which is dictated by the nucleotide sequence of their genes, and which usually results in folding of the protein into a specific three-dimensional structure that determines its activity.
Like other biological macromolecules such as polysaccharides, proteins are essential parts of organisms and participate in virtually every process within cells. Many proteins are enzymes that catalyze biochemical reactions and are vital to metabolism. Proteins also have structural or mechanical functions, such as actin and myosin in muscle and the proteins in the cytoskeleton, which form a system of scaffolding that maintains cell shape. Other proteins are important in cell signaling, immune responses, cell adhesion, and the cell cycle. Proteins are also necessary in animals' diets, since animals cannot synthesize all the amino acids they need and must obtain essential amino acids from food. Through the process of digestion, animals break down ingested protein into free amino acids that are then used in metabolism.
All proteinogenic amino acids possess common structural features, including an α-carbon to which an amino group, a carboxyl group, and a variable side chain are bonded. Only proline differs from this basic structure as it contains an unusual ring to the N-end amine group, which forces the CO–NH amide moiety into a fixed conformation. The side chains of the standard amino acids, detailed in the list of standard amino acids, have a great variety of chemical structures and properties; it is the combined effect of all of the amino acid side chains in a protein that ultimately determines its three-dimensional structure and its chemical reactivity. The amino acids in a polypeptide chain are linked by peptide bonds. Once linked in the protein chain, an individual amino acid is called a residue, and the linked series of carbon, nitrogen, and oxygen atoms are known as the main chain or protein backbone.
Protein Quality
As a fuel, proteins contain 4 kcal per gram, just like carbohydrates and unlike lipids, which contain 9 kcal per gram.
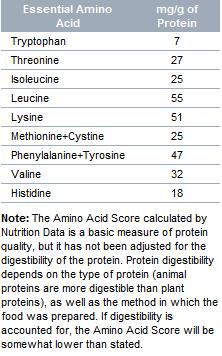
Protein Quality is dependent on having all the essential amino acids in the proper proportions. If one or more amino acids are not present in sufficient amounts, the protein in a food is considered incomplete. Diets that are very low in protein or that are very restrictive in the types of protein consumed (e.g., vegetarian diets) may not provide complete protein.
Each spoke on the Protein Quality Indicator™ represents one of the nine essential amino acids. The size of each spoke is proportionate to the percentage of the optimal level for that amino acid. The amino acid with the lowest level is considered the "limiting" amino acid for that food and determines the overall Amino Acid Score.
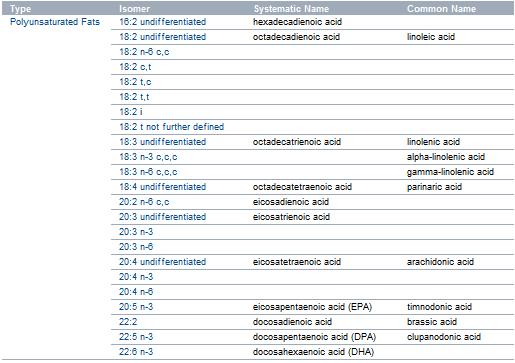
Fatty acids
Fats consist of a wide group of compounds that are generally soluble in organic solvents and generally insoluble in water. Chemically, fats are triglycerides: triesters of glycerol and any of several fatty acids. Fats may be either solid or liquid at room temperature, depending on their structure and composition. Although the words "oils", "fats", and "lipids" are all used to refer to fats, in reality, fat is a subset of lipid. "Oils" is usually used to refer to fats that are liquids at normal room temperature, while "fats" is usually used to refer to fats that are solids at normal room temperature. "Lipids" is used to refer to both liquid and solid fats, along with other related substances, usually in a medical or biochemical context, which are not soluble in water. The word "oil" is also used for any substance that does not mix with water and has a greasy feel, such as petroleum (or crude oil), heating oil, and essential oils, regardless of its chemical structure.
Fats form a category of lipid, distinguished from other lipids by their chemical structure and physical properties. This category of molecules is important for many forms of life, serving both structural and metabolic functions. They are an important part of the diet of most heterotrophs (including humans). Fats or lipids are broken down in the body by enzymes called lipases produced in the pancreas.
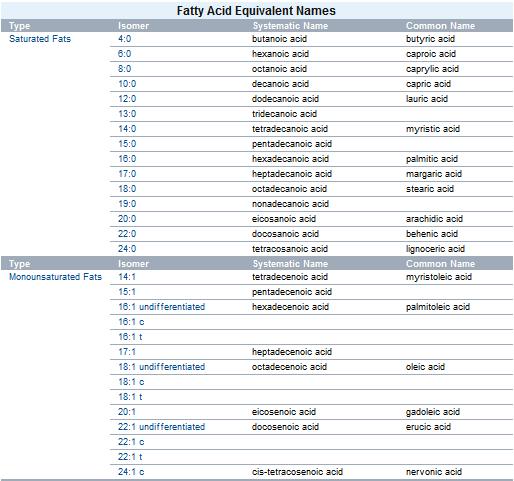
Fats can be categorized into saturated fats and unsaturated fats. Unsaturated fats can be further divided into cis fats, which are the most common in nature, and trans fats, which are rare in nature but present in partially hydrogenated vegetable oils.
There are many different kinds of fats, but each is a variation on the same chemical structure. All fats are derivatives of fatty acids and glycerol. The molecules are called triglycerides, which are triesters of glycerol (an ester being the molecule formed from the reaction of the carboxylic acid and an organic alcohol).
The properties of any specific fat molecule depend on the particular fatty acids that constitute it. Different fatty acids are composed of different numbers of carbon and hydrogen atoms. The carbon atoms, each bonded to two neighboring carbon atoms, form a zigzagging chain; the more carbon atoms there are in any fatty acid, the longer its chain will be. Fatty acids with long chains are more susceptible to intermolecular forces of attraction (in this case, van der Waals forces), rising their melting point. Long chains also yield more energy per molecule when metabolized.
A fat's constituent fatty acids may also differ in the C/H ratio. When all three fatty acids have the formula CnH(2n+1)CO2H, the resulting fat is called "saturated". Values of n usually range from 13 to 17. Each carbon atom in the chain is saturated with hydrogen, meaning they are bonded to as many hydrogens as possible. Unsaturated fats are derived from fatty acids with the formula CnH(2n-1)CO2H. These fatty acids contain double bonds within carbon chain. This results in an "unsaturated" fatty acid. More specifically, it would be a monounsaturated fatty acid. Polyunsaturated fatty acids would be fatty acids with more than one double bond; they have the formula, CnH(2n-3)CO2H and CnH(2n-5)CO2H. Unsaturated fats can be converted to saturated ones by the process of hydrogenation.
Trans fats
There are two ways the double bond may be arranged: the isomer with both parts of the chain on the same side of the double bond (the cis-isomer), or the isomer with the parts of the chain on opposite sides of the double bond (the trans-isomer). Most trans-isomer fats (commonly called trans fats) are commercially produced; trans fatty acids are rare in nature. The cis-isomer introduces a kink into the molecule that prevents the fats from stacking efficiently as in the case of fats with saturated chains. This decreases intermolecular forces between the fat molecules, making it more difficult for unsaturated cis-fats to freeze; they are typically liquid at room temperature. Trans fats may still stack like saturated fats, and are not as susceptible to metabolization as other fats. Trans fats may significantly increase the risk of coronary heart disease.
Importance
Vitamins A, D, E, and K are fat-soluble, meaning they can only be digested, absorbed, and transported in conjunction with fats. Fats are also sources of essential fatty acids, an important dietary requirement.
Fats play a vital role in maintaining healthy skin and hair, insulating body organs against shock, maintaining body temperature, and promoting healthy cell function.
Fats also serve as energy stores for the body, containing about 37 kilojoules per gram (8.8 kcal/g). They are broken down in the body to release glycerol and free fatty acids. The glycerol can be converted to glucose by the liver and thus used as a source of energy.
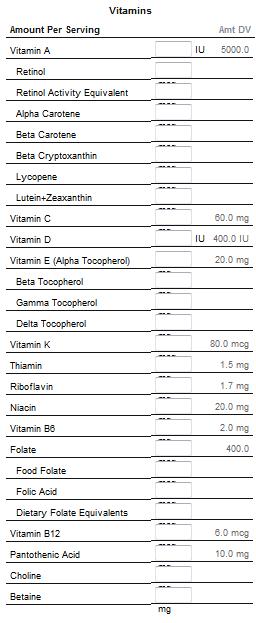
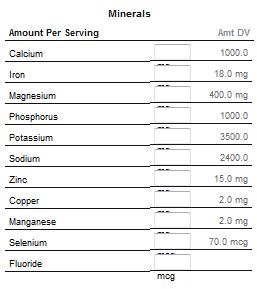
Vitamins
The term vitamin was derived from "vitamine," a compound word coined in 1912 by the Polish biochemist Kazimierz Funk when working at the Lister Institute of Preventive Medicine. The name is from vital and amine, meaning amine of life, because it was suggested in 1912 that the organic micronutrient food factors that prevent beriberi and perhaps other similar dietary-deficiency diseases might be chemical amines. This proved incorrect for the micronutrient class, and the word was shortened to vitamin.
A '''vitamin''' (US /ˈvaɪtəmɪn/ or UK /ˈvɪtəmɪn/) is an organic compound required by an organism as a vital nutrient in limited amounts.[1] An organic chemical compound (or related set of compounds) is called a vitamin when it cannot be synthesized in sufficient quantities by an organism, and must be obtained from the diet. Thus, the term is conditional both on the circumstances and on the particular organism.
Vitamins are classified by their biological and chemical activity, not their structure. Thus, each "vitamin" refers to a number of vitamer (any of a number of chemical compounds, generally having a similar molecular structure, each of which shows vitamin-activity in a vitamin-deficient biological system) compounds that all show the biological activity associated with a particular vitamin. Such a set of chemicals is grouped under an alphabetized vitamin "generic descriptor" title.
Vitamins have diverse biochemical functions. Some, such as vitamin D, have hormone-like functions as regulators of mineral metabolism, or regulators of cell and tissue growth and differentiation (such as some forms of vitamin A). Others function as antioxidants (e.g., vitamin E and sometimes vitamin C). The largest number of vitamins, the B complex vitamins, function as precursors for enzyme cofactors, that help enzymes in their work as catalysts in metabolism.
Vitamins are essential for the normal growth and development of a multicellular organism. Using the genetic blueprint inherited from its parents, a fetus begins to develop, at the moment of conception, from the nutrients it absorbs. It requires certain vitamins and minerals to be present at certain times. These nutrients facilitate the chemical reactions that produce among other things, skin, bone, and muscle. If there is serious deficiency in one or more of these nutrients, a child may develop a deficiency disease. Even minor deficiencies may cause permanent damage.
Vitamins are classified as either water-soluble or fat-soluble. In humans there are 13 vitamins: 4 fat-soluble (A, D, E, and K) and 9 water-soluble (8 B vitamins and vitamin C). Water-soluble vitamins dissolve easily in water and, in general, are readily excreted from the body, to the degree that urinary output is a strong predictor of vitamin consumption.[35] Because they are not as readily stored, more consistent intake is important.[36] Many types of water-soluble vitamins are synthesized by bacteria.[37] Fat-soluble vitamins are absorbed through the intestinal tract with the help of lipids (fats). Because they are more likely to accumulate in the body, they are more likely to lead to hypervitaminosis than are water-soluble vitamins.
Minerals
Dietary elements (commonly known as dietary minerals or mineral nutrients) are the chemical elements required by living organisms, other than the four elements carbon, hydrogen, nitrogen, and oxygen present in common organic molecules. The term "dietary mineral" is archaic, as it describes chemical elements rather than actual minerals.
Chemical elements in order of abundance in the human body include the seven major dietary elements calcium, phosphorus, potassium, sulfur, sodium, chlorine, and magnesium. Important "trace" or minor dietary elements, necessary for mammalian life, include iron, cobalt, copper, zinc, molybdenum, iodine, and selenium (see below for detailed discussion).
Over twenty dietary elements are necessary for mammals, and several more for various other types of life. The total number of chemical elements that are absolutely needed is not known for any organism. Ultratrace amounts of some elements (e.g., boron, chromium) are known to clearly have a role but the exact biochemical nature is unknown, and others (e.g. arsenic, silicon) are suspected to have a role in health, but without proof.
Non-vitamin antioxidants
An antioxidant is a molecule that inhibits the oxidation of other molecules. Oxidation is a chemical reaction that transfers electrons or hydrogen from a substance to an oxidizing agent. Oxidation reactions can produce free radicals. In turn, these radicals can start chain reactions. When the chain reaction occurs in a cell, it can cause damage or death to the cell. Antioxidants terminate these chain reactions by removing free radical intermediates, and inhibit other oxidation reactions. They do this by being oxidized themselves, so antioxidants are often reducing agents.
Although oxidation reactions are crucial for life, they can also be damaging; plants and animals maintain complex systems of multiple types of non-vitamin antioxidants, such as glutathione as well as enzymes such as catalase, superoxide dismutase and various peroxidases. Insufficient levels of antioxidants, or inhibition of the antioxidant enzymes, cause oxidative stress and may damage or kill cells. Oxidative stress is damage to cell structure and cell function by overly reactive oxygen-containing molecules and chronic excessive inflammation. Oxidative stress seems to play a significant role in many human diseases, including cancers. The use of antioxidants in pharmacology is intensively studied, particularly as treatments for stroke and neurodegenerative diseases.
Antioxidants also have many industrial uses, such as preservatives in food and cosmetics and to prevent the degradation of rubber and gasoline.
Antioxidants are classified into two broad divisions, depending on whether they are soluble in water (hydrophilic) or in lipids (lipophilic). In general, water-soluble antioxidants react with oxidants in the cell cytosol and the blood plasma, while lipid-soluble antioxidants protect cell membranes from lipid peroxidation. These compounds may be synthesized in the body or obtained from the diet. The different antioxidants are present at a wide range of concentrations in body fluids and tissues, with some such as glutathione or ubiquinone mostly present within cells, while others such as uric acid are more evenly distributed. Some antioxidants are only found in a few organisms and these compounds can be important in pathogens and can be virulence factors.
Some compounds contribute to antioxidant defense by chelating transition metals and preventing them from catalyzing the production of free radicals in the cell. Particularly important is the ability to sequester iron, which is the function of iron-binding proteins such as transferrin and ferritin. Selenium and zinc are commonly referred to as antioxidant nutrients, but these chemical elements have no antioxidant action themselves and are instead required for the activity of some antioxidant enzymes.
People who eat fruits and vegetables have a lower risk of heart disease and some neurological diseases, and there is evidence that some types of vegetables, and fruits in general, may lower risk against some cancers. Since fruits and vegetables happen to be good sources of nutrients and phytochemicals, this suggested that antioxidant compounds might lower risk against several diseases. This idea has been tested in a limited manner in clinical trials and does not seem to be true, as antioxidant supplements have no clear effect on the risk of chronic diseases such as cancer and heart disease. This suggests that these health benefits come from other substances in fruits and vegetables (possibly dietary fiber) or come from a complex mix of compounds. For example, the antioxidant effect of flavonoid-rich foods seems to be due to fructose-induced increases in the synthesis of the antioxidant uric acid and not to dietary antioxidants per se.
Links
- National Center for Biotechnology Information
- U S Food and Drug Administration Home Page
Home Page for the Food and Drug Administration (FDA) - Home | Food and Nutrition Information Center
- Home | Nutrition.gov
- Wikipedia, the free encyclopedia
- Nutrition facts, calories in food, labels, nutritional information and analysis – Nutritio
Find nutrition facts, including food labels, calories, nutritional information and analysis that helps promote healthy eating by telling you about the foods you eat.