Do Pneumonia Vaccines Cause Super Bugs

How pneumonia vaccines may generate the evolution of super bugs.
A super bug is a bacteria that is resistant to the drugs administered against it such as for ear infections and pneumonia.
Prevnar PCV-7 and PCV-13
Approval for a vaccination containing seven common strains of pneumonia bacterium, pcv-7 manufactured by Pfizer, occurred in 2,000. It is a conjugated vaccine, meaning it contains bacteria (diphtheria) connected to a protein. There exist many more pneumonia-causing bacterial strains and they have developed a resistance to antibiotics and are flourishing.
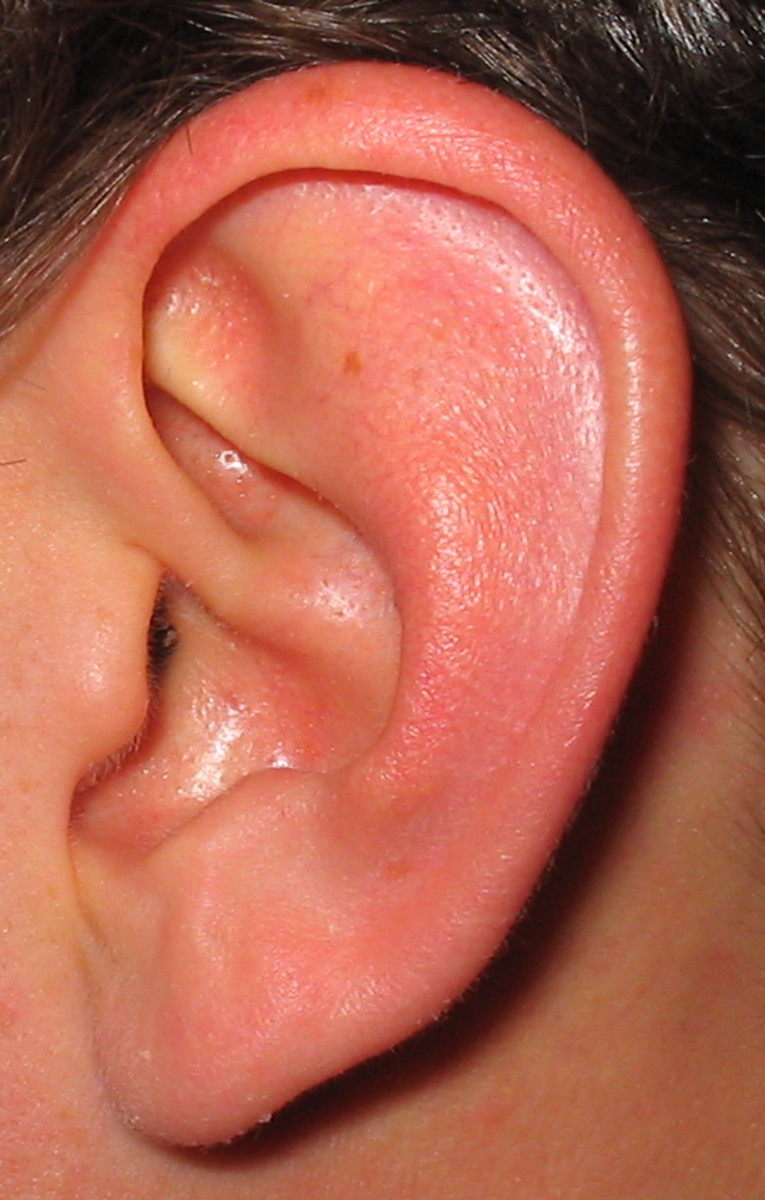
The biological niche that the more common strains had occupied, used in the pcv-7 vaccine, are getting filled by other strains resulting in complicated illness and disease. One particular strain of strep that causes pneumonia and meningitis is called 19A. It has a newly emerged subtype that causes ear infections in ages six to eighteen months and is antibiotic resistant.
Studies have shown that re-vaccinations develop a state of immune tolerance where the body is no longer able to fight off infection from invasive bacteria. All subtypes of strain 19A are increasing in strength.
Bacterial serotype 19A rose from 4 percent between 2002 and 2004 to 15 percent in 2005 and 2006. Respiratory infections in children in the U.S. were tracked by a British lab who found that 40 percent of drug-resistant cases were serotype 19A (super bug). The results by researchers at the University of Iowa in 2004 and 2005 showed that 35 percent of resistant respiratory infections were also serotype 19A.
Average Rate of Pneumonia
Only about 10 in 100,000 a year catch pneumonia, and the yearly cases of children with pneumonia meningitis average seven in 100,000. The risk factor is higher for children who attend day care, have a history of ear infections or antibiotic use (antibiotics are in vaccines) or who have received a rotavirus vaccine.
Infant Vaccination Schedule for Pneumonia, pcv-7
The vaccine schedule for pcv-7 consists of five doses; one at 2 months, 3 months, 4 months, 12 months and 13 months.
Results of a study in the Netherlands on 1,003 infants:
Babies who were vaccinated with pcv-7 were twice as susceptible to a serotype 19A bacterial infection at the age of two. These pathogens are well known to cause middle ear infections as well as the potential to acquire a pneumonia respiratory disease caused by a stealth bacterium that are either immune to, or unaffected by, all drugs and antibiotic treatments.
What has caused the stealth bacterium serotypes 19A to proliferate?
Dutch scientists stated in a research paper that the increase in serotype 19A is associated with the widespread use of pneumonia vaccine in routine infant immunization programs.
How Effective are Pneumonia Shots
The Food and Drug Administration stated in a report that from a series of expert consultations, an agreeable opinion or consensus on the effectiveness of pneumonia vaccination has not been reached. There has been no consistent vaccine effect observed that prevents pneumonia.
The UK government has determined that the adult version of the vaccines, Prevnar pcv-13 by Pfizer and Pneumovax-14 and 23vPS by Merck, are not effective. It even appears to cause more harm than good. They plan to halt the pneumonia vaccine program for people over 65. After millions of injections were given, governmental advisers said the program had no discernible impact on the rate of pneumonia.
The cause for many strep bacteria and viruses to develop a resistance, or immunity, to drugs and antibiotics is the frequency of use over a lengthy period of time. They are used every day by staff in hospitals, prescribed by physicians and even added to some vaccines. From infancy to adulthood, we are not aware of taking in antibiotics through vaccinations and without having a bacterial infection at the time.
Ingredients: Streptococcus pneumoniae (7 common strains), diphtheria CRM protein, casamino acids, yeast extract, ammonium sulfate, aluminum.
Side Effects of pcv-7:
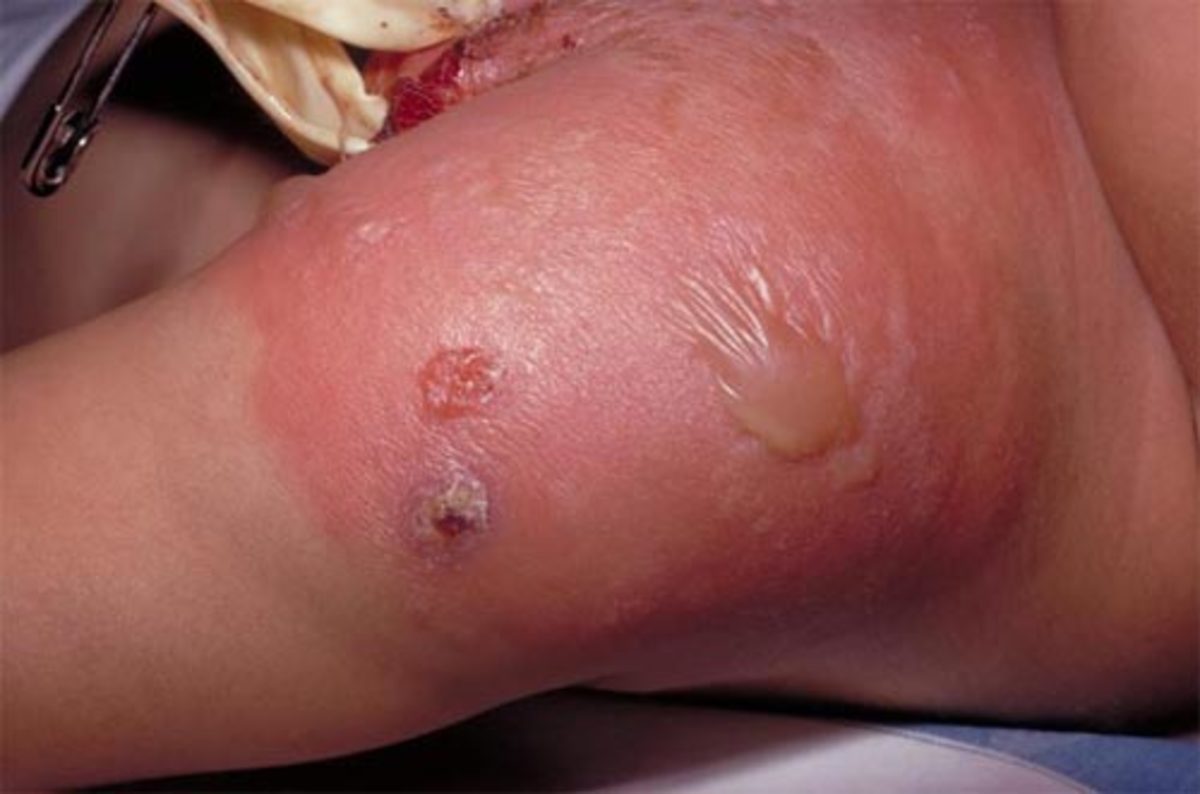
Redness and soreness at the injection site occurs in about 50% of newly vaccinated infants, a fever over 100.3 F in 21%, chills, general malaise, irritability, drowsiness, decreased appetite, and arthritis.
A serious reaction occurs in 1 in 10,000 that include breathing difficulty, hives, accelerated heartbeat and dizzy-like faintness. Seizures also occur in 1 in 10,000 that usually present within 4 days.
Pneumovax and Pneumovax II have been linked to 30 deaths and over 3,300 reported side effects; stated by the Medicines and Healthcare products Regulatory Agency of the UK.ll
Benefit to Risk Ratio
Pneumonia generally occurs at the rate of 10 in 100,000. The rate of incidence for serious vaccine side-effects is 1 in 10,000. The risks from vaccine side effects is the same, if not more than, the potential risks that may be associated with pneumonia meningitis itself.
Pneumovax by Merck and Co.:
A vaccine for pneumonia containing 14 strep strains. It is indicated for people over 65, and re-vaccination at 75. Although it becomes less effective over time, there is no evidence that re-vaccination improves long-term protection. It is made available for anyone over the age of two and people who have long-term illnesses in particular.
Pneumovax was first approved in 1977 based on studies in the 1940s on African gold miners. No randomized studies were ever done. In 1983 Merck released a 23 strain vaccine, 23vPS. By 1995, scientists had recognized 84 serotypes.
Neither are approved for under the age of two and re-vaccination with 23vPS is not included on the package insert as an approved use and is not recommended. Efficacy rate shows 56% against the more common serotypes included in the vaccine; 40% effective for those over 85.
Ages Baby to Adult
Whether or not one becomes more susceptible to stronger strains of strep bacterium, and therefore a super bug, after having received a pneumonia vaccine remains to be of considerable concern.
Whenever making major health care decisions for yourself and your children, especially one that involves a pharmaceutical product such as a vaccine, obtaining information from many different sources you trust is important. You may have to seek the consultation of other doctors to find one who is supportive of your health decisions; one who is willing to work with you in deciding what is best.
Lizolivia