Forward Osmosis is the Next Wave in Water Purification


Forward osmosis (FO) is a membrane process by which seawater may be desalinated and used for direct human consumption. A few companies have developed a unique FO process that uses low-grade heat at 85 degrees C to supply a large percentage of the system’s energy requirements.
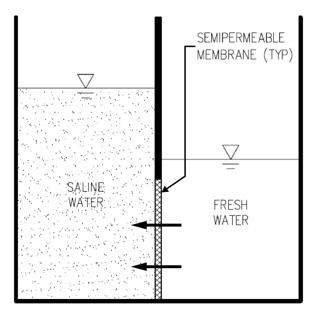
FO (or simply osmosis) is a process commonly encountered in nature. FO occurs naturally in the cells of all living things when liquids of differing concentrations are separated by a semi-porous membrane. FO differs from RO in that natural osmotic pressure that results from the difference in concentration in the two liquids, is the sole driving force for the transport of pure water across the membrane. In the RO process, pure water is driven through a semi-porous membrane using an applied pressure from a solution of high concentration to a solution of lower solute concentration. With FO, osmosis is allowed to occur naturally, without external force, and the pure water moves across the semi-porous membrane to dilute the more concentrated solution until both solutions have equal concentration, or another force acts to stop this movement of water.
In FO, two beakers of water separated by a semi-porous membrane and open to the atmosphere. The beaker on the left contains a water solution with a high concentration of dissolved mixture, and the beaker on the right contains pure water. Due to the differences in solution concentrations, pure water will be pulled through the membrane in order to mix more concentrated solution on the other side. Because this solution is open to atmosphere, rather than continuing until both solutions have equal concentration on both sides of the membrane, this process continues until a column of water exists on the left side that creates a pressure which stops pure water from moving through the membrane. This pressure difference is referred to as the osmotic pressure of the more concentrated solution.
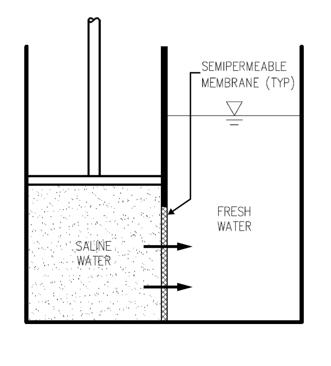
In RO, the process presented is reversed, by subjecting the surface of the concentrated solution on the left to a pressure greater than the osmotic pressure. The applied pressure forces pure water molecules from the concentrated solution across the membrane to the pure water side. In practice, dissolved solutes (salts) will move through the membrane as well. Since the rate of salt diffusion is governed by a concentration gradient, it is unaffected by an increase in applied pressure on the left hand side. If the pressure on the left hand side is high enough, pure water will pass through the membrane at a much higher rate than the dissolved salts. This is the underlying principle of RO. The osmotic pressure of the concentrated solution must be overcome before any water will pass through the membrane to the pure water side. Since the osmotic pressure of a solution is proportional to its concentration, higher pressures are required to treat waters of higher salinity. In the treatment of seawater using RO, feed pressures in the range of 600 to 1,000 psig are required to produce water of quality and at sufficient flow rates to be commercially viable. This high feed pressure requirement directly influences the energy demands of the RO process, and necessitates the use of equipment and piping designed to withstand such pressures.
In the FO process, the primary energy requirement is associated with separating pure water from the draw solution once it has passed through the FO membrane. Several methods can be used to accomplish this separation including RO, and thermal separation (i.e., liquid phase separation, precipitation, or volatization).
• Chemically inert and stable
• Neutral (or near neutral) pH
• Non-toxic
• Does not chemically or physically degrade the FO membrane
• Produces a high osmotic pressure gradient (i.e., very soluble)
• Easily and completely removed from water
For a thermal draw solution used in an FO separation process, it is desirable to choose an osmotic agent with a large change in solubility (the capacity to dissolve) over a relatively small temperature range. Candidates for osmotic agents include substances such as Boron, Diols (a diol is a chemical compound containing two hydroxyl groups), calcium salts of organic compounds, surfactants, water soluble polymers, nano and poly-electrolytes.
In order for FO to be commercially viable for the treatment of seawater for consumption, an osmotic agent must be selected. It is called the draw solution.
The primary energy requirement for using FO in desalination applications is associated with separating the osmotic agent from the diluted draw solution downstream of the FO process. In concept, a designer draw solution may be chosen so that the osmotic agent can be separated from the extracted seawater with low energy input requirement. That is the ideal.
Other possible advantages of FO with respect to RO include much reduced fouling potential of membranes, higher maximum feed water recoveries, and a reduced level of pretreatment.
RO is currently the state-of-the-art among desalination technologies. RO testing showed that seawater can produce water for an energy input of 5.98 to 8.90 kWh/kgal at a 50-mgd facility. Most of this cost is from pressure pumps and non-thermal heating. The theoretical minimum energy of desalination for seawater (free energy of mixing) at 35,000 PPM (35 g/L) salt and 50% recovery is 1.06 kWh/m3. This minimum is an ideal. Today's most energy-efficient commercial technology is two-stage RO with brine booster and runs at 3.17 kWh/m3.
These RO performance figures represent a difficult challenge for the competing FO desalination technology to overcome. This does not mean that there is no room for improvement within the desalination industry. The potential benefits provided by FO desalination in terms of energy savings, reduced pretreatment requirements, lower membrane fouling, and higher recoveries may demonstrate lower overall costs than RO.
Ideally, FO technology is most cost effective at locations where electrical energy costs are high, and fuel costs are low or low quality waste heat is readily available. Also, FO may prove especially competitive with RO at plant capacities between 1 and 10 mgd because of the limitations of feed pumping efficiency at a 10-mgd facility, the energy input to the RO process would increase to 6.6 to 9.8 kWh/kgal.
To separate water from the Draw solution requires heat of 85 degree C. This requires excessive energy. The ideal osmotic agent would have a lower separation temperature to reduce overall thermal losses from the system and would result in a lower thermal specific energy requirement. The reduction in separation temperature would also facilitate the use of lower quality heat sources to drive the separation process.
The boron in product water produced by RO presents a challenge to the membrane industry. Boron rejection ranged from 69 to 84 percent in demonstration tests, resulting in product water with boron concentrations ranging from 0.75 and 1.44 mg/L. The California Department of Public Health (CPDH) has set a drinking water notification level for boron at 1 mg/L, and the World Health Organization (WHO) has set a provisional guideline value for boron levels in drinking water at 0.5 mg/L. Using conventional RO , boron concentrations in the product water decreased as membrane flux was increased and recovery was decreased.
FO has other applications, NASA is designing a small forward osmosis device that could be incorporated into new long-exposure EVA suits in order to recycle wastewater (i.e., sweat and urine) into drinkable fluid. Determining the effect of mechanical mixing on membrane performance may help inform suit designers in the placement of a device to maximize permeate production. NASA also created the HydroPack, and will provide a clean, safe drink from any contaminated water source by simply dropping the product in water and leaving it to hydrate for ten hours. The Forward Osmosis membrane blocks all contaminants and provides an electrolyte enhanced drink that is beneficial to anyone in a water emergency situation. One helicopter filled with HydroPacks is equivalent to sending 14 helicopters of bottled water. It has been in use since 2010 at natural disaster sites and tested in Africa.
FO is the next wave for water purification!