Secrets of Good Soil Fertility - Soil pH
The True Meaning of Soil pH
Most of us are aware of the term soil pH, but what does it actually mean and what to do with the info, because what I am about to tell you will turn most peoples understanding of soil pH on its head.
Soil pH is measuring of the concentration of Hydrogen ions (a cation) compared with Hydroxy ions (anions) within a measured solution on a scale of 1 to 14, 1 being very acidic and 14 being very alkaline. The ideal pH is around 6.5, which is slightly acidic and just below neutral, pH7. (This is where there are equal proportions of hyrogen and hydroxy ions).
So how do you find out the pH of your soil?
You can go to a local garden centre or hardware store and purchase a simple pH test kit or probe. The test kits come in two different forms, either a powder or liquid to which a soil sample is added and the probe is directly inserted into the soil.
For a powder test kit, simply fill the test tube with the required amount of soil and water (must be neutral, otherwise it will skew you results), followed by a capsule of powder which contains the active ingredient. Shake up and down, allow to settle and the colour of the solution will change accordingly, depending on the pH of your soil.
What does a pH actually signify?
The results of a pH test have traditionally been used to indicate whether there is a need to lime (alkalize) when a soil is acidic, below pH 7, or acidify a soil when a soil is alkaline, above pH 7. But according to some eminent soil scientists Prof William Albrecht, Dr Carey Reams and others, this is according to them, following much scientific work, the wrong interpretation of pH readings.
According to them, soil pH is princably determined by concentrations of calcium, magnesium, potassium and sodium ions and by working to get these into balance, soil pH will gradually be brought into line at 6.5 pH; pH readings can also be affected, by temperature, moisture and soil micro-organisms. Therefore, adding lime or sulphur to raise or lower soil pH is incorrect. The calcium in lime, and sulphur, should be viewed as nutrients in there own right as they are vital for healthy plant growth, cell formation and part of a plants defence mechanism.
This may all sound a bit complicated for the average gardener, but for a grower such information could mean the difference between an average crop or an excellent one. pH testing should be used as a rough guide, because the levels of correct soil nutrients are more important.
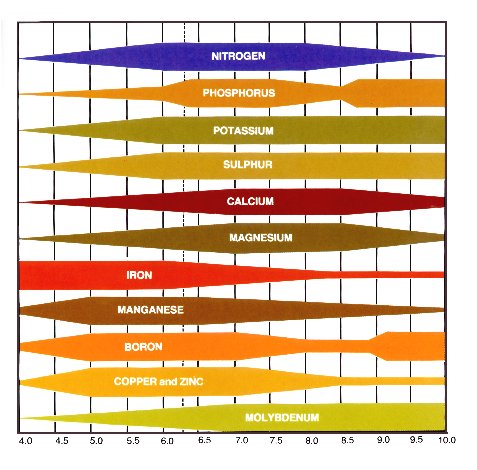
Why pH 6.5?
This the pH level where all the major plant nutrients are available, Nitrates, Potassium, Calcium, Phosphates, Iron, Magnesium, Sulphur, Manganese, Zinc, Copper and Boron. Either side of 6.5 pH, the nutrient availability will either increase or decrease.
by Alistair Olver
Want to get automatic updates when I write new hubs? Subscribe to my Hubpages via RSS.






