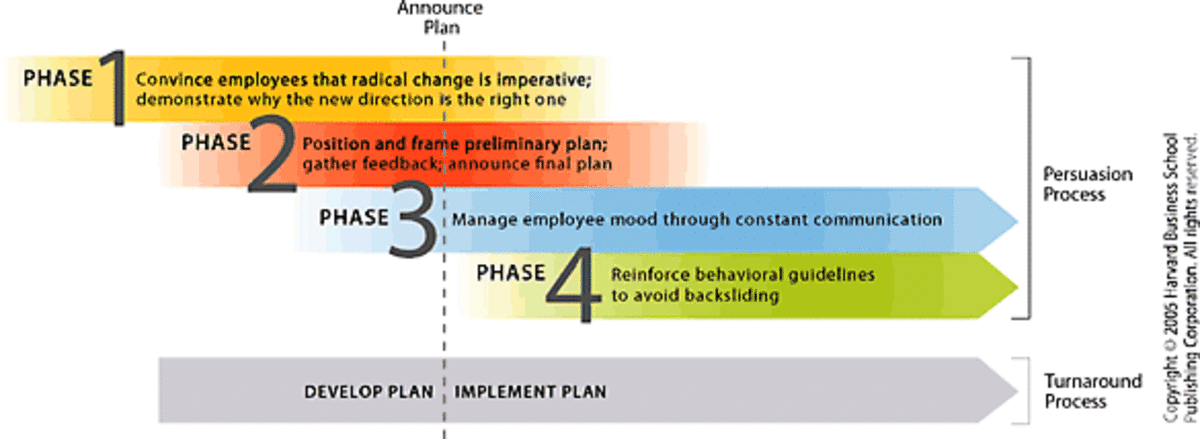
Acid Rain - Causes and Effects
What is Acid Rain
Acid Rain refers to a mixture of wet deposition (acidic rain, fog and snow) and dry deposition (acidic gases and particles) from the atmosphere that contains high levels of nitric and sulfuric acids. Acid rain has a pH of 5.5 or less. It was discovered by the scientists in 1852. The chemicals in acid rain can also destroy the fabrics and discolor them.
Causes of Acid Rain
Acid rain is the consequence of air pollution caused by various gaseous emissions from industries, vehicular emissions, and burning of fossil fuels like coal, petroleum, etc. The occurrence of acid rain is higher in places where there are more number of industries and vehicles, and more use of fossil fuels.
Gases like Sulfur dioxide (SO2) and nitrous oxides (NOx) are the primary causes of acid rain. These gases react with water, oxygen and other chemicals present in the atmosphere, thus resulting in acid rain.

Effects of Acid Rain
- Damage to Aquatic Life – Acid rain has a greater impact on water bodies like lakes, streams and other aquatic environments. It causes acidification of water in these water bodies which makes water toxic to the fishes and other aquatic animals.
- Damage to Forests – Acid rain causes damage to trees and plants. It releases aluminium into the soil, which makes it difficult for trees to take up water.
- Damage to Human Health – Acid rain also affects the human health, as it can lead to breathing and lung problems, especially in individuals with asthma.
- Damage to Buildings – Acid rain causes damage to the building materials and paints, including monuments, statues and sculptures.
Effects of Acid Rain on Monuments
Acid rain greatly affects the buildings and materials by causing corrosion. It can corrode limestone, marble, carbon-steel, zinc, nickel, paint and some plastics.
How Acid Rain affects the buildings
Acid rain is widely affecting famous monuments all over the world. When sulfur pollutants fall on to buildings made from limestone and sandstone they react with minerals in the stone to form a powdery substance that can be washed away by rain. Due to acid rain, the white marble of the monuments becomes pale. The reason for this is usually the local foundries and nearby oil refineries.
Heritage sites at high risk of damage due to acid rain includes one of the wonders of the world ‘The Taj Mahal’ and some other heritage sites under high risk are:
- Leshan Giant Buddha, Mount EMEI, China
- Longmen Grottoes, China
- Acropolis of Athens, Greece
- Dampier Rock Art Complex, Australia
Living as well as non-living bodies are becoming the
victim of pollution and managed to do what years of wars, invasions, and natural disasters have failed to do.