Evolution and Thermodynamics
First off...
When I use the term "creationist" I am referring to someone who believes the universe had a divine creator/designer. I may spit out the term "theist", but I will use the two terms interchangeably, unless otherwise specified. I live in America where the dominant religion is Christianity and I may reference the bible and its dogma.
I am also aware that not every creationist rejects the Theory of Evolution, but I will be addressing an argument proposed by most creationists.
The Argument
The argument goes something like this:
"The Theory of Evolution is false because it violates the Second Law of Thermodynamics because entropy (disorder) increases over time. Therefore, more complex life forms cannot come from less complex life forms."
Every person proposing this argument will have different ways of saying it, but the idea remains static. And at face value it may make some sense, but only if you don't fully understand what the Theory of Evolution and/or the Second Law of Thermodynamics states. So before I get to addressing the argument, I want to take a moment to define those two terms.
What is a Theory?
Evolution hasn't been proven, it's 'just' a theory.
I'm sure we've all heard that one before, and I'm not sure about you but it makes me facepalm every time. So let me just get this out of the way real quick.
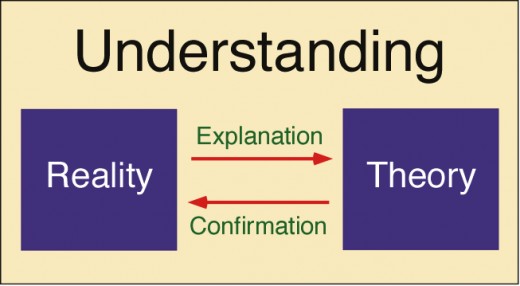
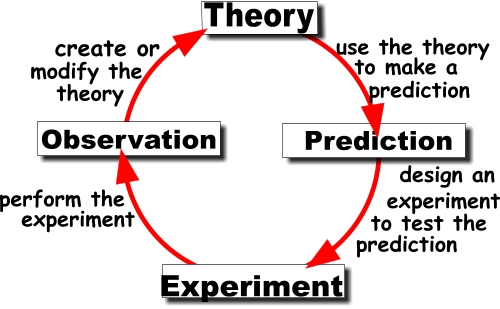
There are different types of theories, depending on context. When we laymen folk use 'theory' in everyday language it typically means hunch or guess. But that theory is different from a scientific theory. A scientific theory is the best explanation for what has been observed and tested. It remains falsifiable, but until something comes along to show that it's wrong, then the theory remains as the best possible explanation.
Sure, a scientific theory isn't fact and can still be disproven, but that wouldn't change the facts of evolution.
But wait, evolution is a theory, not a fact!
I think this is where most laymen folk, such as myself, get confused when talking about scientific theories. A theory in science is an explanation for observed facts and phenomenon in the natural world. Instead of saying 'theory', say 'explanation'. Certain facts were observed and a theory, or explanation, was developed to explain why those facts exists or were observed.
In short, if the Theory of Evolution was somehow shown to be false, the facts of evolution would still remain. We would just need a better explanation for those facts.
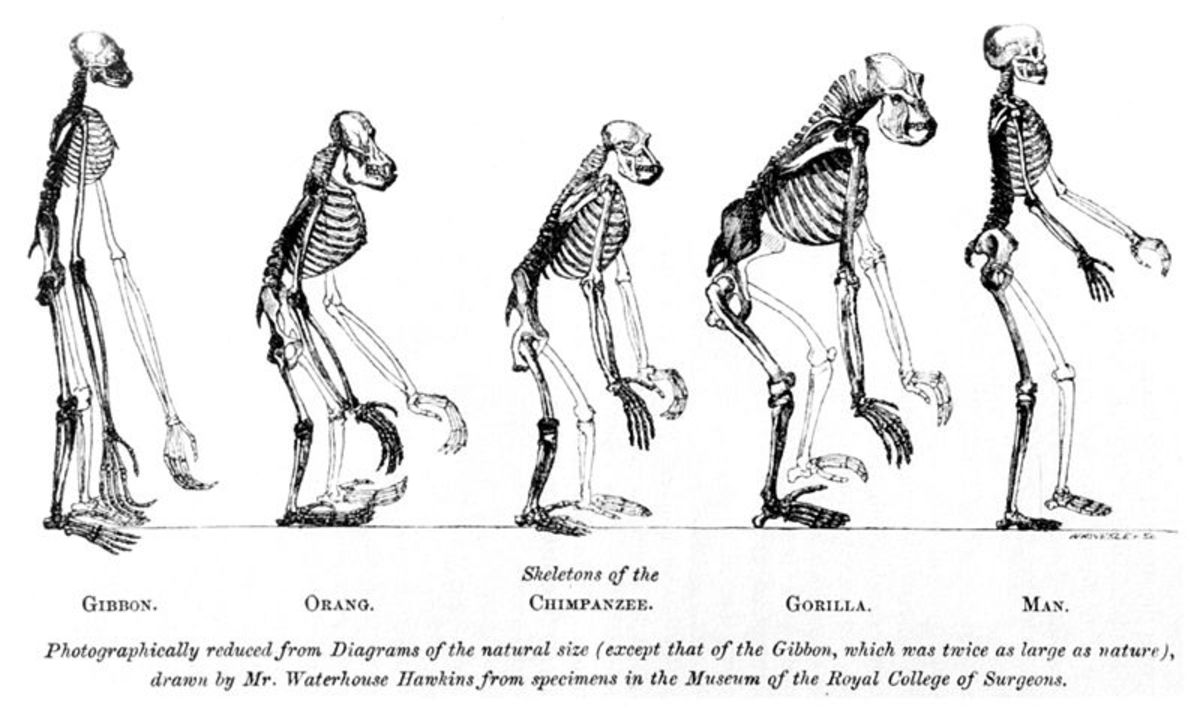
What is the Theory of Evolution?
One misconception, though common, is the idea that evolution explains how life started. But that's not exactly the purpose of the theory. The Theory of Evolution explains the diversity of life on earth and how it came to be through natural processes, not how life originated on earth or anywhere else in the universe.
A quick Google search will yield headlines about evolution being the explanation for the origins of life, but an in-depth reading will show otherwise. Currently, it's unknown how life started on Earth but abiogenesis (life coming from nonlife) has been demonstrated to occur naturally, however, scientists can't confirm that's how life started on our pale blue dot.
For more information on the Theory of Evolution, click here.
What is the Second Law of Thermodynamics?
"The Second Law of Thermodynamics is commonly known as the Law of Increased Entropy. While quantity remains the same (First Law), the quality of matter/energy deteriorates gradually over time. How so? Usable energy is inevitably used for productivity, growth and repair. In the process, usable energy is converted into unusable energy. Thus, usable energy is irretrievably lost in the form of unusable energy." (For source, click here.)
I suppose a simplified way of saying this is that order tends toward disorder because energy is being used but not replenished.

Does the Argument Work?
Again, at face value the argument against evolution may seem sound. How can simple organisms evolve into more complex organisms if there is a loss in energy? More energy would be required for things to become more orderly and complex. So, does the argument work?
No. Here's why. The second law of thermodynamics applies only to a closed system, a scenario in which there is a finite amount of energy. Remember the law talked about energy being "irretrievably lost"? That means no more energy is being put into the system. But the Earth is not a closed system because we have a more or less permanent energy source: the sun.
The sun pumps energy into us in various ways, typically in the form of food. Plants use the energy from the sun to grow and we, in turn, eat the plants (and animals who eat the plants) so we can grow. Energy is being transferred in usable forms. As long as that giant ball of gas keeps burning, we will have all the energy we will ever need to survive and evolve.