Global Warming Fishtank Model
Fish

Ok, so go out and get yourself a big fishtank, pop a hole in the bottom and throw your garden hose in it (don’t really do this – just imagine doing it). Turn the water on so that the fishtank is only filling up slowly – meaning almost as much water is going out as coming in. What will happen? The tank will start to fill up, but will it fill up all the way? No. Why not? Well, because as the water in the tank increases, pressure increases. As pressure increases, the force pushing water through the hole increases and therefore the volume leaving the tank increases. Eventually an equilibrium point will be reached where volume in equals the volume out. Vin = Vout. Let’s say that that point is when the tank is half full. No system is perfect of course so there will be some variability in the water level but on average it should stay the same.
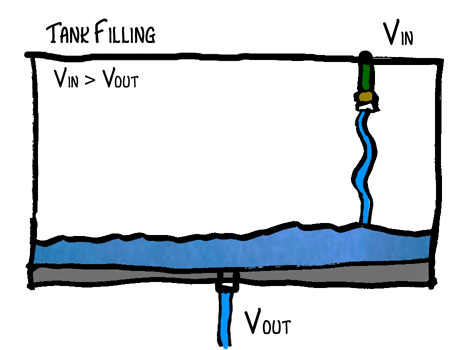
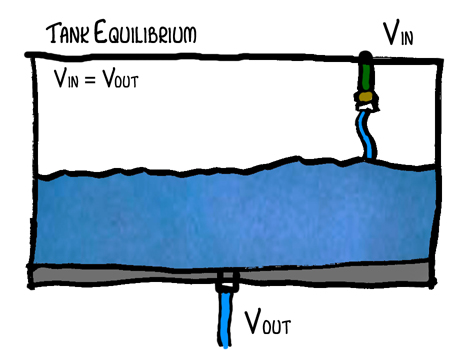
In this example, the tank represents the system of the Earth, the hose represents the energy from the sun and the hole represents the atmosphere (and its ability to allow heat to escape). The atmosphere’s capacity for allowing heat to escape is dependent upon its composition and especially on the greenhouse gases (CO2, methane, etc.).
So what does the theory of global warming say is happening? It says that as we increase greenhouse gases, we decrease the ability for heat to escape the system. In our fishtank example it is the equivalent of shrinking the hole in the bottom. So what will happen if we were to plug part of the hole so it was smaller? Well, the tank will once again start filling up with water because less water can escape through the smaller hole. But once again as the tank fills, pressure increases, and therefore the volume of water escaping increases until it once again reaches an equilibrium point.
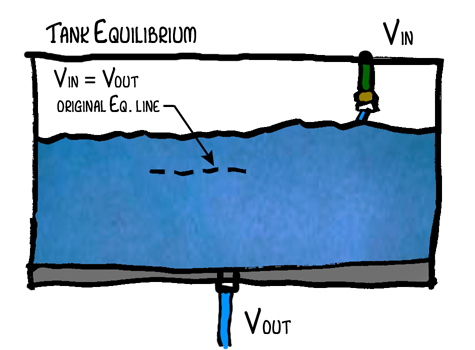
So what exactly does the water in the fishtank represent? It represents the energy stored in the system. In the case of the Earth, that largely means heat.
Some people make the erroneous claim that for any system energy in has to equal energy out. They say that if the energy from the sun is constant that the Earth can’t warm up. If there isn’t more energy coming in, how can energy increase. Their argument almost makes sense except that it is entirely WRONG! Energy in doesn’t have to equal energy out!! How can this be?
When you look at the tank at equilibrium, energy in does equal energy out, but look at what happens when the tank is filling with water. At that point the volume in is greater than the volume out. So where does that energy go? It can’t just disappear. That’s right. It doesn’t disappear. It is being absorbed by the system. When the tank is filling up the energy equation is Vin = Vout + Energyabsorbed.
The earth is of course far more complex than this, but fundamentally it is a worthy model for explaining how the energy stored in a system can change even when the energy coming in stays the same. It can do so by modifying the resistance presented to energy escaping.