Do You Know What's In Your Toothpaste? Is Anything Safe Anymore?

Triclosan is an antibacterial and antifungal chemical that is added to many products to prevent or limit bacterial contamination. Along with toothpaste, mouthwash, deodorant, and soap, Triclosan can be found in hair products, cosmetics, shaving cream, household cleaners, clothing, toys, and a variety of other products most people use every day.
Canada, the European Union, and other countries have already banned the use of Triclosan, or limited its use, and the FDA is planning to release a report on the dangers Triclosan poses to both humans and the environment generally.
Animal studies so far have shown that Triclosan alters hormone regulation. The FDA calls Triclosan a powerful antibiotic, "too powerful." Triclosan, like many other antibiotics, is believed to contribute to making bacteria antibiotic resistant.
Edward Markey, D-Congressman from Massachusetts, has requested that the FDA conduct a scientific and regulatory review of Triclosan and the results are expected to be available next year. Markey wants Triclosan banned from use in products intended for children as well as any products that would come into contact with food.
Dr. David Brownstein is a board-certified family physician, and one of the leading practitioners of holistic medicine in the U.S. He says Triclosan is an endocrine disruptor, because it depresses thyroid hormones in the bodies of rats. The more Triclosan the rats were fed in the studies, the more negative symptoms they exhibited.
Rats, and humans share slightly more than 99% of the same genes. For that reason rats are considered a superior animal for studying and modeling human disease.
According to Newsmax Health, “In the Centers for Disease Control and Prevention’s National Health and Nutritional Examination Survey (2003-2004), Triclosan was detected in 75 percent of people studied. Detectable levels of Triclosan were also found in breast milk.
What Are The Advantages of Triclosan?
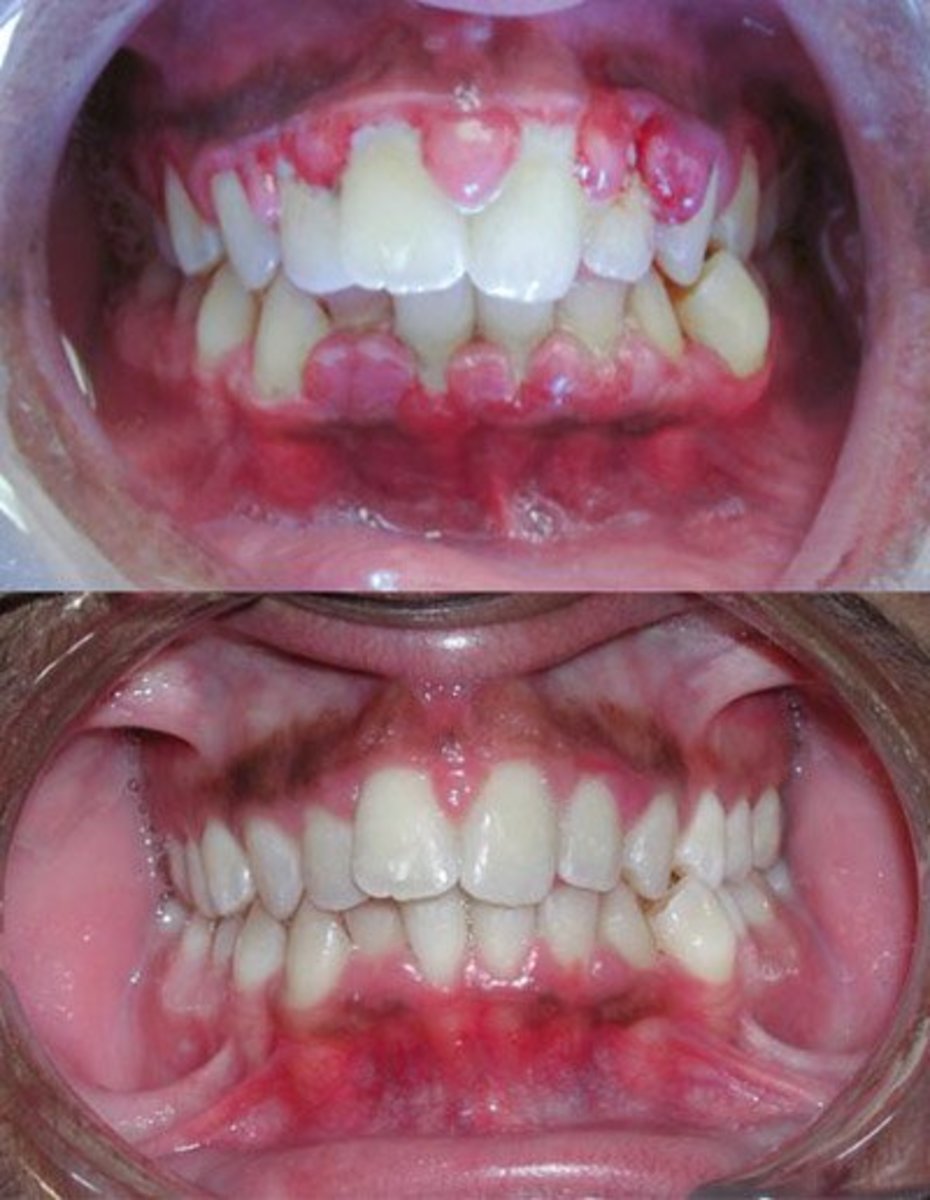
Triclosan has been shown to inhibit plaque formation and buildup on teeth, which causes cavities. Also, Triclosan has proven to be more effective than fluoride in preventing gingivitis, an inflammation of the gums causing them to be red, puffy, tender, and to bleed easily. In its late stages gingivitis causes tooth and bone loss.
As previously stated, Triclosan can be found in some antibacterial soaps, but the FDA has said it has no proof that washing with soap that includes Triclosan is better than washing with soap that does not include it.
Triclosan Is Not The Only Health Threat In Toothpaste
According to USA Today, a company in Los Angeles has been charged with importing and distributing nearly 90,000 tubes of poisonous toothpaste from China. The toothpaste contained diethylene glycol, which is a chemical used as a solvent in antifreeze. Diethylene glycol, used by the Chinese instead of glycerin to thicken the toothpaste can cause kidney and liver damage over time.
The New York Times reports that this same Chinese toothpaste containing the diethylene glycol that was sold in Los Angeles was also sold in Miami and Puerto Rico.
The FDA is recommending that consumers discard any and all toothpaste they may have that was made in China. Most of the Chinese toothpaste is marketed at Dollar Plus stores and similar retail outlets.
The toothpaste in this case was ShiR Fresh Mint Fluoride Paste, but the FDA says it has identified at least 9 other Chinese toothpaste brands marketed in the U.S. containing the diethylene glycol, which was not listed in the ingredients on the package.
Keeping You and Your Family Safe
Reading the ingredients listed on the package is becoming more and more important. Triclosan, when it is included in a toothpaste, soap, or other hygiene product manufactured in the U.S., will be listed in the ingredients stated on the package.
Avoid products of dubious origin.
Sources
The New York Times: http://www.nytimes.com/2007/06/02/us/02toothpaste.html
USA Today: http://www.usatoday.com/money/industries/manufacturing/2008-03-06-toxic-toothpaste-charges_N.htm
Canada.com: http://www.canada.com/health/Common+toothpaste+additive+triclosan+deemed+toxic+environment/6361712/story.html
Medical Daily
http://www.medicaldaily.com/fda-reviews-antibacterial-hand-soap-why-triclosan-bad-you-and-planet-245474
ConsumerReports.org: http://news.consumerreports.org/safety/2010/05/should-triclosan-be-banned.html
U.S. Food and Drug Administration
https://www.fda.gov/ForConsumers/ConsumerUpdates/ucm205999.htm
© 2012 C E Clark