Radiocarbon Dating

The Discovery
Radiocarbon Dating was found by American chemist Willard Libby in 1949. Since then, it has been used widely as a dating method I archaeology.
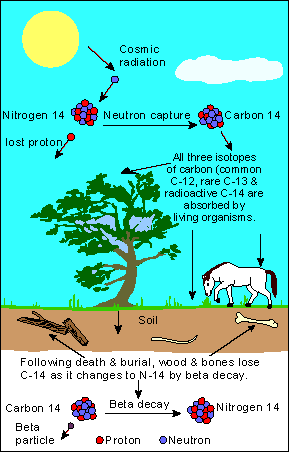
The Underlying Concept
The atmosphere contains a fixed ratio of Carbon-12 [C-12: Ordinary Carbon] and Carbon-14[C-14: Radioactive isotope of Carbon]. C-14 is formed by the effect of cosmic radiation on Nitrogen in the atmosphere.
Now plants absorb C-14 in the atmosphere through their intake of Carbon dioxide during photosynthesis. This C-14 passes on to animals as they feed off plants and other animals.
This ingestion of C-14 stops when the animal or plant dies after which the radio-isotope remaining in the physical structure if the organism begins to disintegrate at the rate of one half every 5730 years.
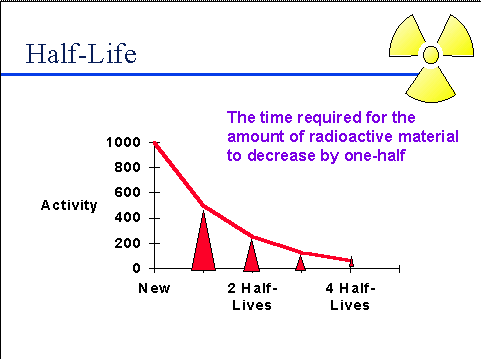
This implies that after 5730 years the concentration of C-14 left in the structure will become half of the original concentration at the time of death. This phenomenon is known as the half life of a radio-isotope.
By measuring the amount of C-14 left in the body, scientists can find out when it died or how old it is.
This method is used to date many organic materials like wood, charcoal, bones and shells.

An Approximate Method
The C-14 method provides only approximate results like all other dating methods and a standard error margin called STANDARD DEVIATION is calculated. This implies that the dates are accompanied by a +/- factor.
For example let’s have a look at the following date: 2500 +/- 100 BP
This means we have a range between 2400 BP to 2600 BP
Here “ BP” refers to BEFORE PRESENT and the year 1950 which introduced radiocarbon dating is taken as the base line or the first year or PRESENT for further calculations.

Archaeologists sometimes calculate the mean of multiple dates to lower the value of the standard deviation and reach a more precise figure.
Miscalculations
Sometimes the results which are found are way too wrong. This may happen from contamination of the sample or from procedural errors.

Calibration Discrepancies
Over the years the amount of C-14 produced in the atmosphere has not been constant. Also, there have been notable differences between the results obtained by C-14 dating and other methods like the Tree-rings Dating Method.
Due to these reasons, it has been made clear that some corrections are needed while converting Radiocarbon dates to Christian Calendar dates.
Some archaeologists prefer to publish uncalibrated dates due to the ongoing debates over calibration.
However, certain calibration tables have been more or less accepted by some scholars.
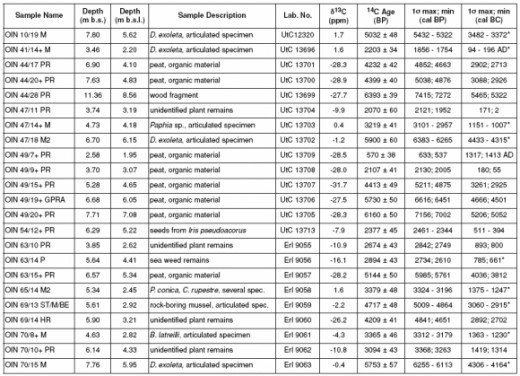
The Simultaneous Significance and Dilemma of the Method
Radiocarbon dates have made a dramatic difference to our understanding of Chronology of Ancient Cultures. But, different books often show different Radiocarbon Dates.
Why does this happen?
One answer could be the ongoing debates over calibration.
But the profound reason is the varied interpretation and judgment involved while using these dates. A doubt arises when a string of dates become available for one site. Therefore, confusion arises regarding which date to use.
Hence, there are choices to be made in the use of Radiocarbon dates.
How an archaeologist interprets and presents these dates depends on his/her understanding of the relative dating and history of various cultures.
An Introduction To Radiocarbon Dating