Structure of Atoms

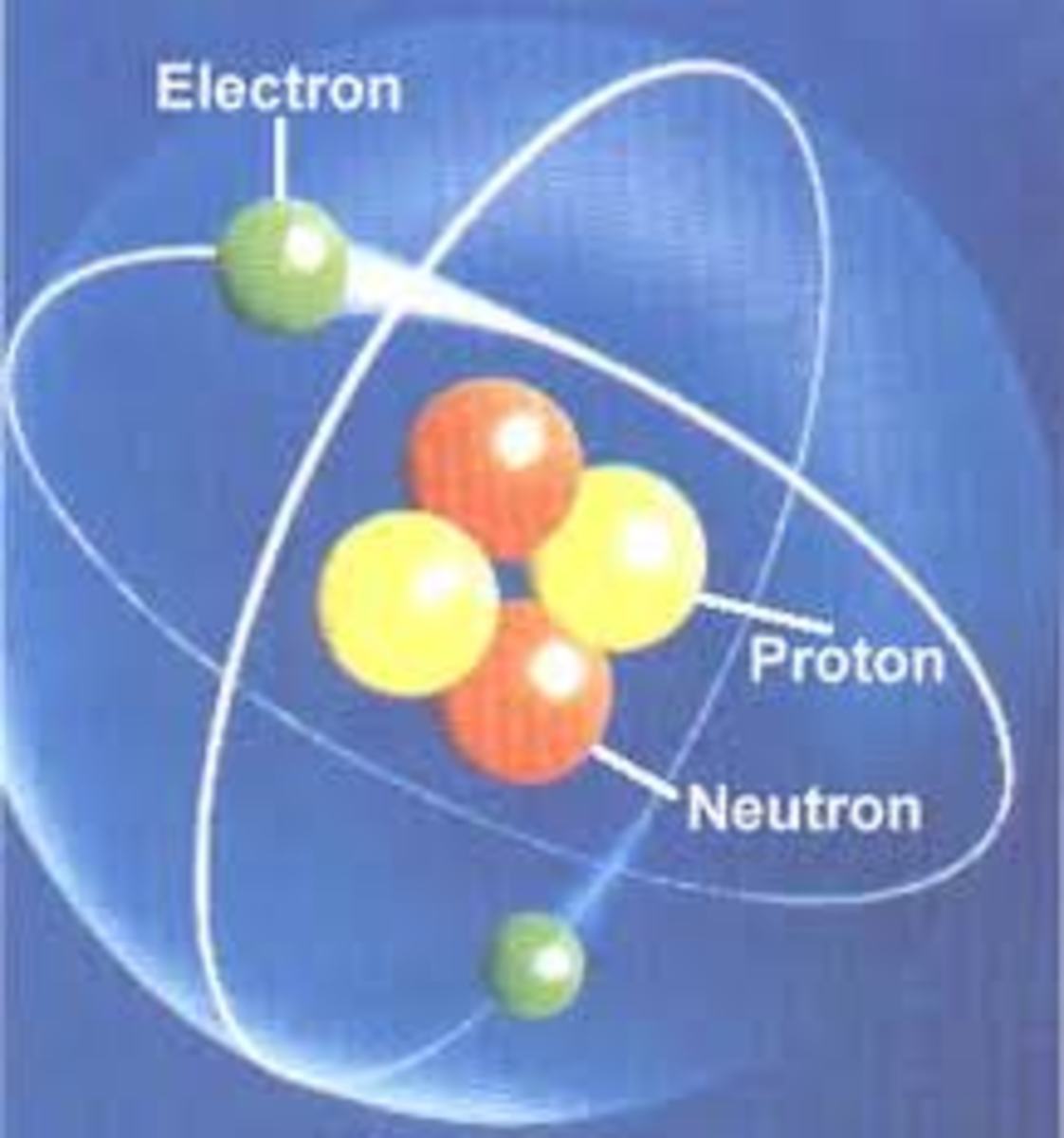
The Nucleus
The nucleus of an atom is made up of nucleons. Nucleons are positively charged protons and neutrons which have no charge. Protons have roughly the same mass as neutrons. Protons are held together by a very strong short range force called nuclear force that is strong enough to counteract the electrostatic repulsive force of the protons and that is why the nucleus stays together. There is also a gravitational force involved but this is too weak to be of any consequence at this level. 99.9% of an atom resides in the nucleus. The rest of the atom consists mainly of empty space because the nucleus is in a cloud of negatively charged particles called electrons each with a mass that is only a tiny fraction of the mass of the nucleus.
In a stable atom the number of electrons will be the same as the number of protons and in that way the positive charge exactly balances the negative charge.

Spectral Lines
The electrons move very quickly about the nucleus and each electron has its own energy within the cloud it inhabits. If an electron is interfered with in any way then this energy is altered and things happen, e.g. if an electron is given more energy as when it is struck by another electron or particle or quantum of light then it gets excited and behaves in an excited manner until it releases this excess energy and goes back to being itself. The energy released is exactly the energy it gained from the collision and it can be seen using spectral analysis.
Isotopes
Two atoms with the same number of protons but different number of neutrons are called isotopes of each of other. The isotopes however would have the same number of electrons as the number of protons is the same. It then follows that the chemical properties as isotopes are the same. Interestingly scientists are still unable to separate isotopes by chemical methods and different elements have different numbers of isotopes. Gold and Cobalt both have only one naturally occurring isotope whereas Tin as 10.
Atomic Number v Atomic Mass
In the Periodic Table of Elements each element X appears with two numbers. A X Z
The larger number A is called the Mass Number and the smaller number Z is called the Atomic Number. These numbers allow us to determine the structure of each element
e.g. 7Li3
In 7Li3, the letters Li stands for the element Lithium. The number 3 tells us how many protons in the nucleus and the number 7 tells us the collective number of nucleons (protons and neutrons) in the nucleus. By subtracting 3 from 7 we can find the number of neutrons in the nucleus of 7Li3 which is the 4. Because the number of protons must be the same as the number of electrons in every stable atom it follows that 7Li3 has 3 electrons in the cloud around the nucleus.
1 H1 is hydrogen 2 H1 and 3 H1 are isotopes of Hydrogen called Deuterium and Tritium respectively and only differ in the number of neutrons in the core.
Isotopes of Hydrogen
Tritium (3 H1)is a radioactive isotope of Hydrogen(1 H1). It is rare for Tritium to occur naturally on earth. The radiation from Tritium is relatively weak but if ingested or inhaled into the lungs it is potential hazard.
Tritium is produced in nuclear reactors using Heavy Water moderators to slow neutrons down. Heavy Water is water with the hydrogen atoms replaced with Deuterium atoms. Heavy Water is not radioactive but is used in nuclear reactors to slow down neutrons enough to react with the core. These neutrons can be captured by the deuterium atoms in heavy water to form Tritium but not in any great numbers. The risk to the environment is however great enough to warrant the moderator being cleaned of Tritium every few years.
Intersetingly, plant seeds will not germinate in heavy water and animals given only heavy water to drink become ill and actually die when about 50% of their body-water has been replaced with heavy water.

Nature and our Core
Isn't amazing how such tiny changes at the atomic level can have such huge influences on other levels. Imagine that a Hydroden atom can become so different as a result of having one or two extra neutrons in the nucleus.
I wonder if in fact nature is teaching us a lesson in the very structure of the atoms that make up our physical existence.
The number of electrons in the cloud orbiting the nucleus of an atom does not have the same impact on the character of an atom as the structure of its core though it does determine its behaviour chemically. If we use this as an analogy for our own lives doesn't it make sense that we pay more attention to the core of our being rather to our external perceptions in our pursuit of truth? Ultimately it is what we believe we are at the core of our being that determines who we are and the activities we involve ourselves in are mere reflections of our core structure.
Ordinary hydrogen in water is life sustaining whereas isotopes of hydrogen in water are life threatening. The character of the core determines the ultimate contribution we each make to this world constructively or destructively.
Link to related articles by Spirit Whisperer
- The Cosmological Constant-Einstein's Biggest Blunder
After publishing his theories of relativity Albert Einstein still felt dissatisfied. The Theory of Special Relativity and General Relativity helped to give us the best understanding of the universe to date and... - Do What You Are Told
Nearly 100 years after the death of Copernicus, the great Mathematician and Scientist who challenged the worlds belief about our planetary system, Galileo Galilei (1564 1642) stood trial before the... - Stephen Hawkings Theory of No God
In his book The Grand Design Stephen Hawking comments on the ignorance of our ancestors who invented Gods to explain how nature behaved. He suggests that people developed a victim mentality as a result of... - Quantum Quandary