The Science behind Cooking

Ok here we are- the main dish. Now that I’ve covered the basics in part one of this series, we can get down to cooking. Cooking is an intricate process, most of us do it every day but do we stop to think what really goes on in there? What happens to the molecules and the bonds holding them together when we subject our foods to intense heat? Along for the ride in this second part, is my go-to Food Science guru, Alton Brown.
Related Hub
- The Science behind Food
Part one of a series looking into the science behind food and cooking. Discussion of essential nutrients in the human diet and their chemical composition gives way to an accompanying piece on cooking.
Let’s take a particular dish, chicken broccoli ziti, and see what really happens. The pasta and chicken will be cooked in a pot of boiling water and the broccoli will be steamed. We will take into consideration, biologically speaking, what is in these foods. We know pasta as a carbohydrate, chicken as a protein, and broccoli as a vegetable. Even though we think of pasta as carbs and our meats as proteins, we are simplifying things- these foods are complex. In the case of meats, fruits, and vegetables they were living organisms. They need the basic biologic building blocks- carbohydrates, proteins, lipids, and nucleic acid- just like us, in order to survive. Even though we associate a given food with one of the three major nutrients- carbohydrates, protein, or lipids- or one of the other three nutrients-vitamins, minerals, or water- this is for simplicity’s sake. In reality a piece of chicken or a bunch of broccoli contains representatives from most of the six nutrient classes.

Broccoli is known for being a good source of vitamin E; it also contains a very small amount of fat along with some fiber and sugar in the carbohydrate category and some protein. Additionally broccoli provides sodium, calcium, and iron. It contributes to the daily value of vitamin A and vitamin C as well. These estimates are based on a cup of chopped broccoli and the full report can be seen here. Chicken, of course, contains protein, it has no carbs to be noted in a one pound piece of chicken breast and a small amount of fats overall with about 7% of one’s daily value of cholesterol. Again this is based on the nutrition data website. For our last ingredient, one cooked cup of macaroni contains 14% of one’s daily value of carbohydrates. These carbs are largely in the form of starch. Macaroni also contains a good amount of protein, along with a few minerals-selenium, phosphorus, and manganese.
Alton Brown’s cookbook I’m Just Here for the Food introduces heat in the context of cooking. The major heating methods in the kitchen can be broken down to three heat mechanisms- conduction, convection, and radiation. Conduction is heat transfer via direct contact from a hot object, such as a pan, to a less hot object, in this case a piece of food. Heat will seek out an equilibrium, so the hotter item will transfer heat to the cooler item until a leveling off has occurred. Convection represents the transfer of heat energy in gas or liquid (and some solids) by movements of current. Heating a gas or liquid causes it to warm, expand, and becomes less dense. Then it cools and becomes denser- collectively this is a convection current. Radiation occurs when electromagnetic waves direct the transport of energy through space. Ultimately these waves transfer energy when they come into contact with an object.
Ok this video is a little cheesy and directed at middle schoolers but it sums up heat transfer in cooking nicely
Boiling an ingredient in water, like any of the three for this dish, gives a 70:30 ratio of conduction to convection. When water reaches its boiling point, 212° F, it produces vapor, turbulence, bubbles, and steam. For pasta, boiling water will get rid of excess starches on the surface of the food, while the heat and agitation will rehydrate and gelatinize the starch. This rehydration occurs when cooking dried pasta. The 30% convection in boiling water in a pot keeps the ingredient from sticking to the sides. Pasta as mentioned above, consists largely of starch, the storage form of glucose with its bonds and often complex configuration condensing the monomers into a tight area. The cooking process will begin to break down these glycosidic bonds, converting our starch into shorter, less complex carbohydrate chains. Mushier pasta means less starch is present and conversely indicates there are more simple carbohydrates. Pasta cooked al dente presents a healthier choice than mushy pasta since it retains more starch and thus less glucose is roaming free.
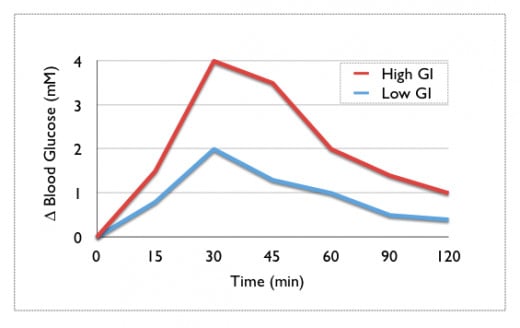
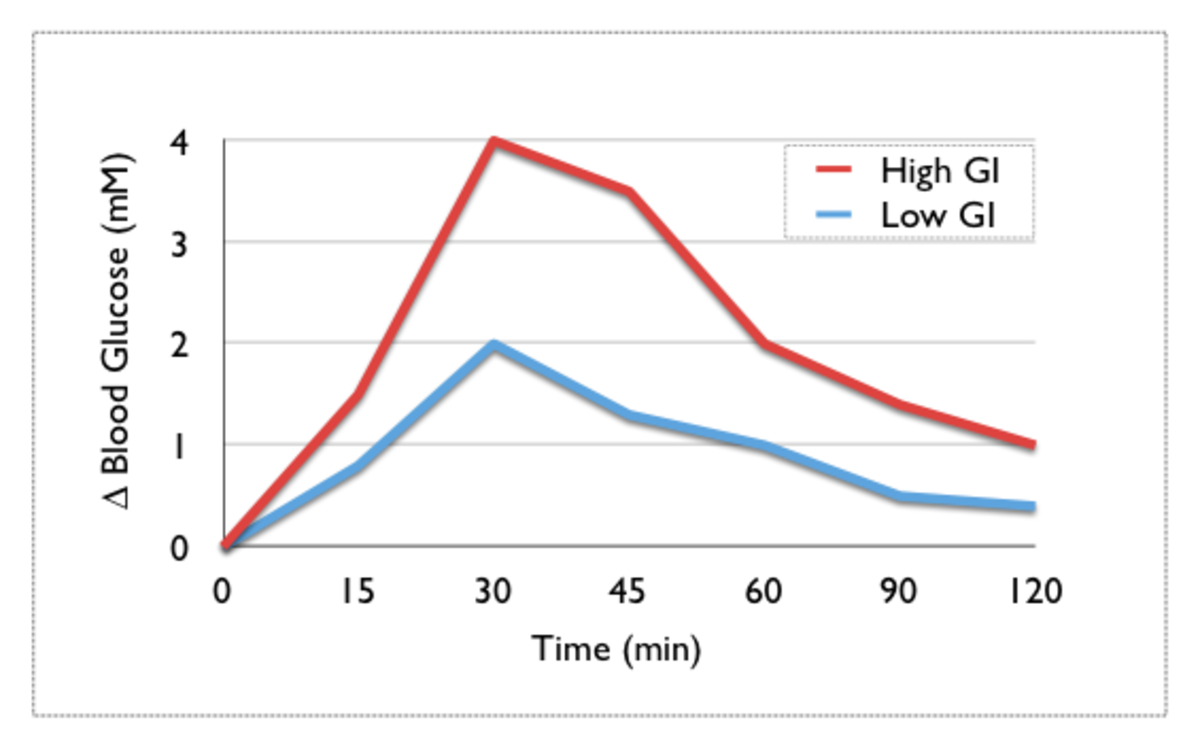
Starch is better for our bodies than simple sugars based on its glycemic index values. Starch rates low on the glycemic index (the scale runs from zero to one hundred) which means the body slowly absorbs it. This makes sense if we think back to the introduction article on this topic, the more complex a carbohydrate is when entering our digestion system, the longer it takes for the cascade of enzymes to break it down to the components our small intestine can absorb. For carbohydrates those components are simple monosaccharides- glucose, galactose, and fructose. When we ingest things that have many monosaccharides to start out with, a handful of gummy bears or in our discussion here overcooked pasta, the body doesn’t have to do as much, if any, work in terms of breaking down the components. It just needs them to make their way to the major absorption sites of the digestion system and cross over to the bloodstream. These types of foods are high glycemic index foods, foods those watching what they eat want to minimize and/or avoid. Of course a trade-off exists here; an ingredient with all of its starches intact would be tough for the body to break down and to most people wouldn’t taste as good. The more glucose monomers released from the complex structure of starch during cooking, the sweeter the food will taste as we consume it.
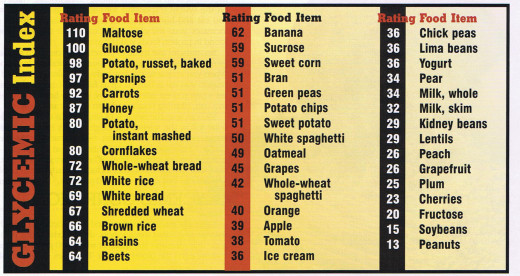

Levels of Protein Structure
Level
| Description
|
|---|---|
Primary
| Amino Acid Sequence
|
Secondary
| Coils and Folds of the primary structure, held together by hydrogen bonds. Alpha helices are coils made at every fourth amino acid, while pleated sheets consist of bonds between parallel polypeptides.
|
Tertiary
| Bonds between particular side chains (R group) of amino acids. The most common is the disulfide bridges between two cysteine molecules within sulfhydryl groups on side chains.
|
Quarternary
| This level is present when a given protein is made up of multiple subunits; it represents the bonding between these subunits.
|
Now we move on to cooking our chicken via boiling it in water. Chicken is predominantly known for its major component: protein. In the body proteins need to be handled with care, they possess an intricate structure-function relationship where changes in pH and temperature, as well as exposure to modifying agents, can cause destruction to the structure and thus function as well. In the cooking realm, exposure to heat most times represents a necessity for meats- to ensure safety for consumption by ridding them of micro-organisms. This heat denature the proteins within the meat as well. The four levels of protein structure are important to a discussion about cooking- a brief summary is seen in the table to the right. Proteins composed of multiple subunits display all four levels of structure, including quaternary structure. These subunits, which can be identical or unique, are held together by hydrogen bonds. These bonds are somewhat easy to break down, compared to peptide bonds, during application of pressure. In the case of cooking this pressure is heat. Heat will break down the hydrogen bonds holding together protein subunits but will not break down the peptide bonds holding the primary structure, the amino acids, together. Collagen is an example of a protein that has all four levels of protein structure, it contains three supercoiled subunits, wound together to form a triple helix. The bonding between these subunits contributes to the overall strength of the protein. Collagen is a major component of connective tissue found in animals. When cooking chicken the hydrogen bonds between the collagen subunits will break down, making the food much more edible since collagen is strong and tough to breakdown without the aid of heat.
Last up is our broccoli- one of the major things that happens to vegetables during cooking is a change in its physical appearance. While cooking broccoli it turns from a dull green color to a vibrant green and then, depending on how long you cook, it the color fades out again. Plants gain nutrients from a different mechanism than animals, a major difference between these two kingdoms. Photosynthesis allows plants to absorb energy from sunlight and convert it into fuel for all of its necessary processes. The driver behind photosynthesis is chloroplasts found within plants. These pigmented granules carry out photosynthesis. The pigment found in chloroplasts are chlorophyll giving the granule its green color, since chloroplasts are abundant throughout an entire plant these granules give the outward appearance of the plant. The chlorophyll itself absorbs light energy and the enzymes within the chloroplast convert it, along with water and carbon dioxide, to glucose and its byproducts.
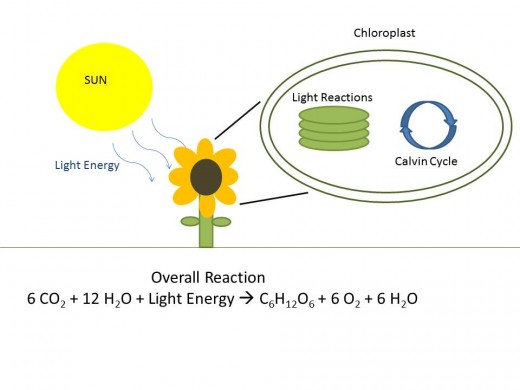
When a vegetable is initially heated up to cook, the gases around the chloroplasts disperse, making the chlorophyll look more pronounced. But chloroplasts are unstable, as time passes cooking damages them and the vegetable loses some of its color. As the broccoli continues to cook, acids within are released. Hydrogen will swap in for magnesium in the chlorophyll pigment, converting it to a grayish pigment- pheophytin. Broccoli contains a generous portion of Vitamin E, a fat-soluble vitamin. It is a good idea to know which key vitamins a vegetable contains in the context of how you are going to prepare it. Fat-soluble vitamins-A, D, E and K- will be lost to some extent if your cooking medium is fat-based. While the B vitamins and vitamin C are water-soluble and therefore some amounts of them can be lost in a water-based approach. Based on this idea, steaming veggies makes good sense as the best way to get the most nutrients out of them.
So there we have it- our chicken and broccoli with pasta made while exploring some of the scientific details involved in the cooking process. We used convection and conduction to sanitize our chicken; break down the collagen for edibility’s sake, began the starch breakdown process in our pasta and experienced a colorful journey with our vegetable. So the next time you are going through the routine of cooking, think of all the wonderful science occurring while you prep.