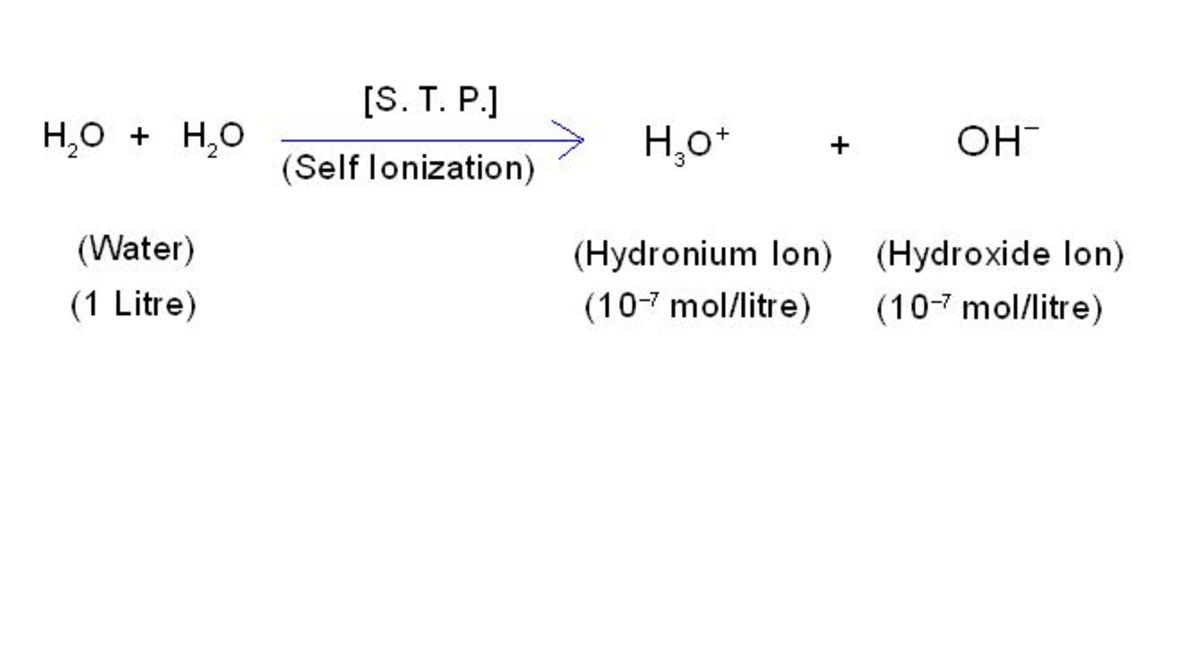
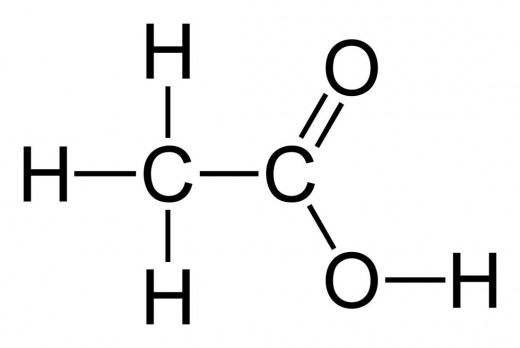
Acetic Acid
What is Acetic Acid?
Acetic acid, also known as ethanoic acid, is an organic acid that gives vinegar its sour taste and pungent smell. The name derives from acetum, the Latin word for vinegar. It is also the defining ingredient of vinegar. But vinegar is not the only use of acetic acid. The global demand for acetic acid is around 6.5 million tonnes per year. Industrially, acetic acid is used in a wide range of processes. Acetic acid is produced both synthetically and by bacterial fermentation. The bacterial fermentation accounts for 10% of the world production and is mostly used for vinegar.

Glacial Acetic Acid
Glacial Acetic Acid is 99% pure acetic acid, (water-free) and is dangerously corrosive. Pure, water-free acetic acid is a colourless liquid that absorbs water from the environment, and freezes at 16.5 °C (62 °F) to a colourless crystalline solid as seen on the picture right. Glacial acetic acid is also used in many different products such as paint, varnish, lacquer and other related products. It is also an important chemical reagent and industrial chemical, used in the production of soft drink bottles; cellulose acetate, mainly for photographic film; and polyvinyl acetate for wood glue, as well as synthetic fibres and fabrics. Dilute solutions of glacial acetic acid can be used in the clinical laboratory to lyse red blood cells in order to do manual white blood cell counts.

Acetic Acid as Solvent
Acetic Acid can be used as a solvent for various applications. The chemical Acetophenone, which is obtained by the dry distillation of the calcium salts of acetic acid and other acids, is used for ethers in the making of alcohol-soluble resins. These in turn are used in a whole range of perfumes. The fragrances that could be created with acetophenone are strawberry, jasmine, cherry, almond and honeysuckle.
Acetphenone was also used in medicine. It was marketed as a hypnotic under the brand name Hypnone. Hypnone was commonly used in psychiatric hospitals to induce sleep with patients who suffered from delerium tremens.

Acetic Acid for the Chemist
Pure acetic acid is a very strong and corrosive poison. If ingested it causes vomitting with intense pain and is usually followed by convulsions and coma. If the acetic acid remains in the stomach for a while it even eats out the walls of your stomach. In case of a poisoning by acetic acid, milk or flour and water should be immediately taken.
Acetic Acid must be handled with special resistant gloves. Latex gloves offen no protection. Gloves made from nitrile rubber are usually worn when handling these compounds.
Books for the Chemist
Acetic Acid and its Derivatives
Summary of the book:
Striking a balance between basic chemistry and chemical engineering, this up-to-date reference discusses important aspects of acetic acid and its major derivatives, including chemistry, methods of preparation and manufacture, and synthesis, as well as current and emerging downstream technologies. The book provides comprehensive physical property data for compounds and their separation, including acetic acid-water separation. Describing five categories of techniques for the manufacture of acetic acid, it: examines thermophysical properties and aqueous solutions, with detailed explanations of mathematical models and correlations; supplies a critical analysis of property; outlines manufacturing costs and related economic factors; reviews the applications of acetic acid and derivatives; covers the chemistry and preparation of the derivatives; elucidates recent topics such as deicers, esters and new esterification technologies.

Acetic Acid in Vinegar
Vinegar derives from the old French words vine aigre which means "sour wine". The concentration of acetic acid in vinegar usually ranges from 4% to 8%. The concentration of acetic acid for pickling is a bit higher which is around 18%. Different sources are claiming that vinegar was invented during the Xia Dynasty, around 2000 BC. But traces have been found in Egyptian urns dating from around 3000 BC.
Vinegar is produced by fermentation through the oxidation of ethanol by acetic acid bacteria. The ethanol can be derived from wine, beer, fermented fruit juice, cider or even in a synthetical form from natural gas and petroleum derivatives. The fermentation process is either slow or fast. Slow fermentation processes are used in traditional vinegars and produce a vinegar of a higher quality like the Reggio Emilia Traditional Balsamic Vinega r.


History of Acetic Acid
Traces of acetic acid being used in alchemy extend into the third century. The Greek philosopher Theophrastus described how acetic acid can be used to produce pigments useful in art when acted on metals. The ancient Romans used to boil vinegar in pots made from lead to make Sapa, a highly sweet syrup. Sapa was rich in lead acetate which they called sugar of Saturn, a very sweet substance. This syrup caused also a lot of lead poisoning among the Roman aristocracy.
The preparation of glacial acetic acid through dry distillation of metal acetates was already known in the Renaissance, as the 16th century German alchemist Andreas Libavius described this procedure. (see picture below)

Acetic Acid in Medical Uses
Small amounts of acetic acid are being used as an application to cancer. The most common cancer where acetic acid is being used is cancer of the skin. It's also being used for growths in the nose, warts, ulcers, lupus and ringworm. Vinegar or dilute acetic acid is used for night sweats and to relieve diarrhea.
Ear infections of the outer ear caused by bacteria or fungus are well known to be treated with acetic acid as an antibiotic. It can help remove excessive earwax. Brand names are: Acetasol, Vosol and Vasotate.
Another well known use for acetic acid or plain white vinegar is treating the box jellyfish sting. Acetic acid or vinegar neutralizes the box jelly's nematocysts that have not yet discharged into the bloodstream.
Acetic Acid as a Natural Weed Killer
It's easy and cheap to buy chemical based weed killers. But if you care for your environment there are other options. Sure, the chemical based solutions work faster and longer but are a danger for your own health and the health of the environment.
Acetic acid can be used sucessfully, as it draws all the moisture out of the leafs. The higher the concentration of acetic acid the better it will operate. Plain vinegar's concentration of acetic acid is often too small for killing the weed. For eco-friendly landscaping projects and landscaping ideas there are different kinds of products on the market with higher concentrations of acetic acid that will do the job just fine.