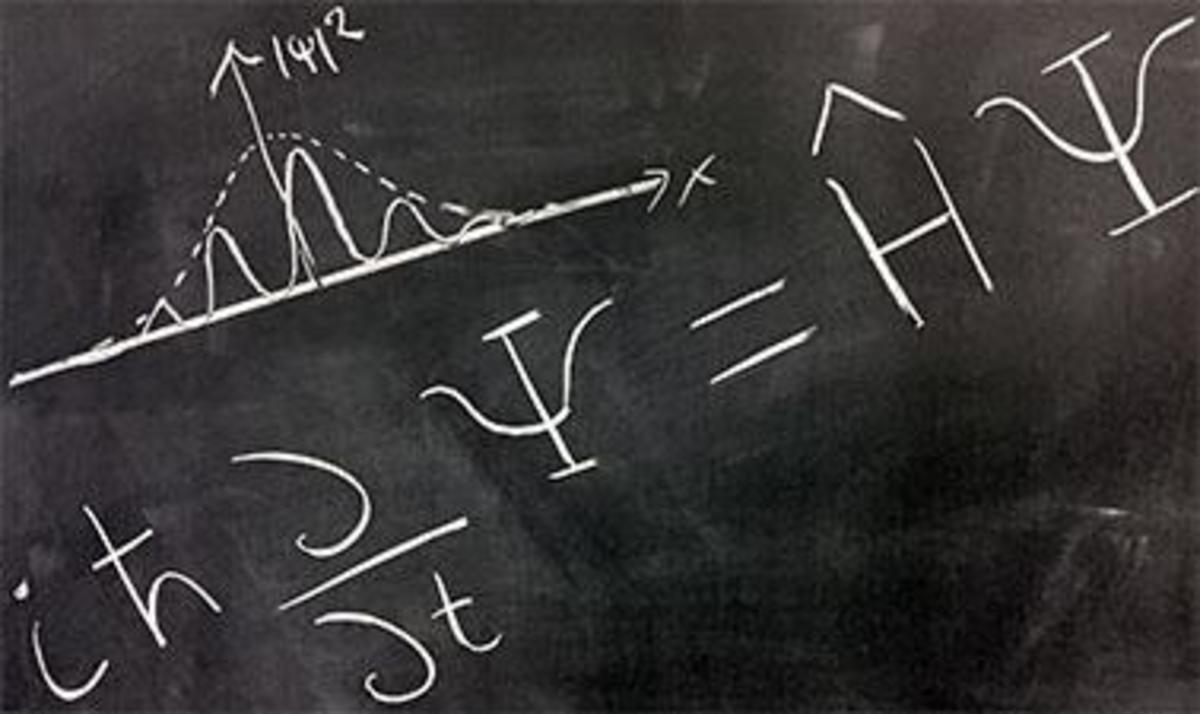The Photoelectric Effect Demonstrates The Particle Nature of Light
The Hertz Effect
The Photoelectric Effect was once referred to as the Hertz effect after Heinrich Hertz who first observed the phenomenon in 1887. Hertz observed that a metal plate illuminated with ultra violet light spontaneously emitted electrons from its surface.
Little did Hertz realise that the study of this phenomenon would one day lead to the idea of the wave-particle duality of matter and the quantum nature of light.
The Classical Theory
Up to thiseveryone had accepted James Clerk Maxwell's idea that light was made up of waves. His wave equations and all the experiments bore this fact out. Since Light could be reflected, refracted, diffracted and polarised so everyone just accepted the wave nature of light.
The Photoelectric Effect was going to change all of that but in the meantime scientists were having a hard time letting go of their old ideas even in the face of all the new evidence to the contrary.
The Terminology
Let me take you through this step by step so you can fully appreciate the impact that this effect was to have on how science was to view light and matter.
Let us first become familiar with a few terms in relation to waves which light was considered to be.
The energy of a wave depended on its frequency so the higher the frequency the more energetic the wave. Since the wavelength and frequency are inversely proportional then the higher the frequency the shorter the wavelength. The intensity of a wave or its brightness depends on its amplitude and for sound waves the louder the sound the higher the amplitude.
Visible light is just one narrow band of electromagnetic radiation and I will be using the terms light, electromagnetic radiation and radiation to refer to the energy radiating the metal surfaces in question.
Einstein's Photoelectric Equation
Einstein's beautiful Photoelectric Equation explains the phenomenon so eloquently:
The Kinetic Energy KE of the electrons being emitted from the surface of a metal with Work Function W is the difference between the energy it receives form the incident photon or quantum of light and the energy it uses to overcome the work function of the metal it is escaping from. Einstein gave the energy of the photon as hf where h is Planck's constant and f is the frequency of the light.
Work function of a metal would be similar to gravity holding us on the surface of our planet. In order for us to be emitted from the surface of our planet we would need to be given enough energy to escape gravity and any energy we had left over would be used to send us speeding off into space.
KE = hf - W
Einstein's explanation of the photoelectric effect won him the Nobel Prize in Physics in 1921.
Einstein v Maxwell
James Clerk Maxwell with his wave equations was the authority on waves at the time and though the results undermined Maxwell's theory Einstein's explanation of what was going on was so radical that it was fiercely resisted by those loyal to Maxwell and his ideas.
Einstein basically said that rather than consider electromagnetic radiation as continuous waves he asked that them to consider light to be packets of energy like tiny bullets. This idea implied the particle nature of light and this was just too weird for scientists to accept at this time.
When radiation is thought of as packets of energy called quanta then when a quantum of light hit an electron it gave the electron its energy.
If the energy of the incident quantum of light was large enough for the electron to overcome the Work Function of the metal then it jumped off the surface with the remaining energy.
This would then explain why the effect happened or didn't without any time lag and when it did it happen it only did for a particular metal when light of a particular frequency was shone on it.
Important to note was that at no time was there a time lag observed so waiting did not affect the results. Either the electrons had enough energy to escape or they didn't and Einstein's idea of looking at light as packets of energy explained this perfectly.
Also by considering radiation to be packets of energy called photons or quanta the intensity of the incident radiation would now be defined as the number of such packets of energy or quanta striking the metal surface and that would account then for the intensity affecting the number of photoelectrons being emitted from a particular metal surface when light of a particular frequency was shone on it.
The Experiments
Following is a simplified version of the experiments conducted.
Light of a particular frequency was shone on a metal plate and the number of electrons emitted and their energy was recorded. For the same metal plate a range of frequencies of light were shone on the same plate and the readings for each were recorded.
The experiment was then repeated for different metals and the results recorded.
It was observed that for a given metal and a particular frequency of light either electrons were emitted or they were not. Increasing the intensity did not change this very important fact that the effect was spontaneous. This really puzzled the scientists who imagined that electrons would simply keep absorbing the energy from the light wave hitting it until they had enough to escape from the metal surface but this was not what was happening.
Shining increasingly higher frequency electromagnetic radiation on a specific metal plate eventually caused the effect. As the frequency was increased the energy andnotthe number of the electrons being emitted from the metal surface increased. They found that for a particular frequency of radiation it was the intensity and not the frequency of the radiation that actually affected the number of electron being emitted from the metal surface.
Attachment to Old Beliefs
Even with all the experimental evidence and Einstein's equation for the photoelectric effect supporters of Maxwell continued to oppose Einstein's idea of light exhibiting particle properties. Resistance to Einstein's idea of photons continued mainly because it appeared to contradict Maxwell's equations which had adequately explained reality up to this point and they weren't ready to throw all of that away.
Maxwell's classical theory would have the electrons accumulating enough energy to eventually jump off the metal surface and the energy of the emitted electron would depend on the intensity of the incident radiation but this simply was not the case.
Einstein himself fell prey to the same attachment that supporters of Maxwell's Theory felt for his classical theory when he came up with the Cosmological Constant to explain why the Universe does not collapse under gravity and I have written a separate article about that called:
“The Cosmological Constant-Einstein's Biggest Blunder”.
Applications
The Photoelectric Cell or Phototube generates a current when light shining on specially coated metal cathode emits electrons that are then attracted to the anode which is wire surrounded by the cathode. The current generated depends on the intensity of the incident radiation. The current from such a cell can be multiplied when a series of cells is arranged in an electron tube as the current is amplified by secondary electron emission. Phototubes arranged like this are capable of detecting radiation of very low intensity and the device is called a Photomultiplier which is used for radiation detection in nuclear research.
Another application of the phototube is the photo sensor motion detector which is used in burglar alarms, traffic light controls and automatic door openers.
When you shine the car headlights at your garage door and it opens the current to drive the motor that opens the garage door is generated by the photoelectric effect.
Sadly, the effect, like many other discoveries in science, has been used to hurt and kill people. Security forces checking abandoned cars in Northern Ireland would detonate a car bomb when they shone their torches into the car they were checking.
Video About The Photoelectric Effect and its Applications
Following is an animated video from YouTube to explain the Photoelectric Effect and its practical applications.
Richard Feynman
Following is a a really interesting video from YouTube with the famous Physicist Richard Feynman on Wave Particle Duality.
My Inspirational Teacher - Brother Mac
I first learned of the photoelectric effect when I was about 17 years old. The Principal of my school was also my Physics and Mathematics teacher and his name was Brother McDonnell but we fondly called him Brother Mac for short.
He was the man who would inspire me to study Physics and Mathematics and to become a teacher. It was his lesson about the photoelectric effect that captured my imagination and which started my love affair with Mathematics and Physics.
Brother Mc Donnell was more than just a Mathematics and Physics teacher, he was a man I respected and admired. He was humble and yet he could be firm but always fair and encouraging. He was a man who practised what he preached and was the role model I aspired to emulate throughout my teaching career.
He opened my eyes to the the way ideas and beliefs come and go but morality stays and grows!
He showed me how to appreciate the beauty of the ideas put forward by great scientists but always remembering to refrain from attachment to any of those same ideas.
Conclusion
It has been my personal experience that it is always the most intelligent among us who are always the ones who find it hard to accept change. I suppose if a person invests so much time and energy into an idea that seems to fit so well and explains everything so well then it is natural that the person would be inclined to defend his beliefs tooth and nail. It however seems like a kind of insanity to me for anyone to hold on so rigidly to beliefs that no longer fit the picture.
I have come to the conclusion that science will never fully explain reality because it is based on thinking. Thinking is an activity that serves us as we engage and interact with an illusory world we call reality. Thinking will simply give rise to more thoughts and beliefs but thinking will never explain what does not exist. Ironically it is not thinking but feeling that gives rise to discovery and it was only when these great thinkers had temporarily suspended their thinking that they made their wonderful discoveries. Newton was sitting daydreaming under an apple tree and not poring over his notes in a laboratory when he came up with his ideas about gravity. The German chemist Friedrich Kekule found the configuration for benzene in a dream while sleeping and not over his desk going over the hundreds of permutations of shapes he had thought up.
The more we balance thinking with feeling the happier and more accepting we will become of ourselves and the world and that is the only state of being conducive to learning anything of true value.
The ego will always present us with ideas to maintain the illusion we serve. Though Einstein's ideas fit the picture now they will not always do so. As long as we keep looking we will keep finding more and more ways of looking and the cycle is self propagating. Searching for explanations of what we believe to be true serves only to maintain our belief in illusion as truth. As long as we believe that what we see is real and can be explained by thought we continue to sleep.